Portable ASV for Arsenic Speciation in Aquatic Systems: A Field-Deployable Approach for Rapid, On-Site Analysis
This article provides a comprehensive overview of the application of portable Anodic Stripping Voltammetry (ASV) for the speciation of inorganic arsenic in aquatic environments.
Portable ASV for Arsenic Speciation in Aquatic Systems: A Field-Deployable Approach for Rapid, On-Site Analysis
Abstract
This article provides a comprehensive overview of the application of portable Anodic Stripping Voltammetry (ASV) for the speciation of inorganic arsenic in aquatic environments. Arsenic speciation, the differentiation between the more toxic arsenite (As(III)) and arsenate (As(V)), is critical for accurate risk assessment and remediation, yet reliable on-site analysis remains a challenge. We explore the foundational principles of ASV, detail methodological protocols for field application in diverse water matrices, and discuss strategies for troubleshooting and optimizing performance against common interferences. Furthermore, we validate the portable ASV technique against established laboratory methods, including ICP-MS and selective chemisorbent materials, demonstrating its reliability for rapid, cost-effective environmental monitoring. This resource is tailored for researchers, environmental scientists, and public health professionals seeking to implement robust, on-site arsenic speciation technologies.
Why Arsenic Speciation Matters: The Critical Role of ASV in Environmental and Health Risk Assessment
The Global Challenge of Arsenic Contamination in Aquatic Systems
Arsenic contamination of aquatic systems represents a critical global environmental and public health challenge. Arsenic is a toxic metalloid naturally present in the Earth's crust, which can be released into water bodies through geological processes and anthropogenic activities such as mining, industrial operations, and agricultural use of pesticides [1] [2]. It exists in multiple oxidation states (+III, +V, 0, -III) and various inorganic and organic chemical species, with over 100 different arsenic compounds identified in environmental systems [2].
The toxicity, mobility, and environmental fate of arsenic are critically dependent on its chemical species. Inorganic arsenic, particularly trivalent arsenite (As(III)), is significantly more toxic and mobile than pentavalent arsenate (As(V)), while organic forms such as arsenobetaine (AsB) and arsenocholine (AsC) are generally considered less toxic [3] [2]. Chronic exposure to inorganic arsenic through contaminated drinking water has been linked to serious health conditions including skin lesions, cardiovascular diseases, neurological disorders, and various cancers [1] [4]. The World Health Organization (WHO) has established a maximum permissible limit of 10 micrograms per liter (μg/L) for inorganic arsenic in drinking water, a threshold adopted by many regulatory agencies worldwide [1] [4].
This application note focuses on the development and implementation of portable anodic stripping voltammetry (ASV) methods for arsenic speciation analysis in aquatic systems. The ability to perform rapid, sensitive, and cost-effective on-site determination and speciation of inorganic arsenic is crucial for environmental monitoring, risk assessment, and remediation efforts.
Analytical Techniques for Arsenic Speciation
Traditional Laboratory Methods
Traditional techniques for arsenic speciation analysis include chromatographic, spectroscopic, and hyphenated methods. High-performance liquid chromatography coupled with inductively coupled plasma mass spectrometry (HPLC-ICP-MS) is widely regarded as the gold standard for arsenic speciation, offering excellent sensitivity and the ability to separate multiple arsenic species simultaneously [3] [1]. Other commonly used techniques include hydride generation atomic absorption spectroscopy (HG-AAS), atomic fluorescence spectrometry (AFS), and capillary electrophoresis (CE) [1] [2].
While these methods provide high sensitivity and selectivity, they present significant limitations for field deployment. They typically require complex sample preparation, sophisticated instrumentation, highly trained personnel, and laboratory infrastructure, making them time-consuming, costly, and unsuitable for on-site analysis [1] [4].
Electrochemical Methods
Electrochemical techniques, particularly anodic stripping voltammetry (ASV), have emerged as promising alternatives for arsenic detection and speciation. ASV offers high sensitivity, portability, rapid analysis, and cost-effectiveness, making it ideally suited for field deployment [5] [4] [6].
The fundamental principle of ASV involves two main steps: (1) a preconcentration step where arsenic species are electrochemically reduced and deposited onto the working electrode surface, and (2) a stripping step where the deposited arsenic is oxidized back into solution, generating a measurable current signal proportional to concentration [5] [6]. The selection of working electrode material is crucial, with gold electrodes being particularly effective for arsenic detection due to their favorable interaction with arsenic species [5] [6].
Table 1: Comparison of Analytical Techniques for Arsenic Speciation
| Technique | Detection Limit (μg/L) | Analysis Time | Portability | Cost | Speciation Capability |
|---|---|---|---|---|---|
| HPLC-ICP-MS | 0.005-0.05 | 10-30 min | No | Very High | Excellent |
| HG-AAS | 0.01-0.1 | 15-20 min | No | High | Good (with pretreatment) |
| CV-AFS | 0.005-0.02 | 10-15 min | No | High | Good (with pretreatment) |
| Portable ASV | 0.1-0.8 | 2-5 min | Yes | Moderate | Good (As(III)/As(V)) |
Portable ASV Method for Arsenic Speciation
Principles of ASV-based Arsenic Speciation
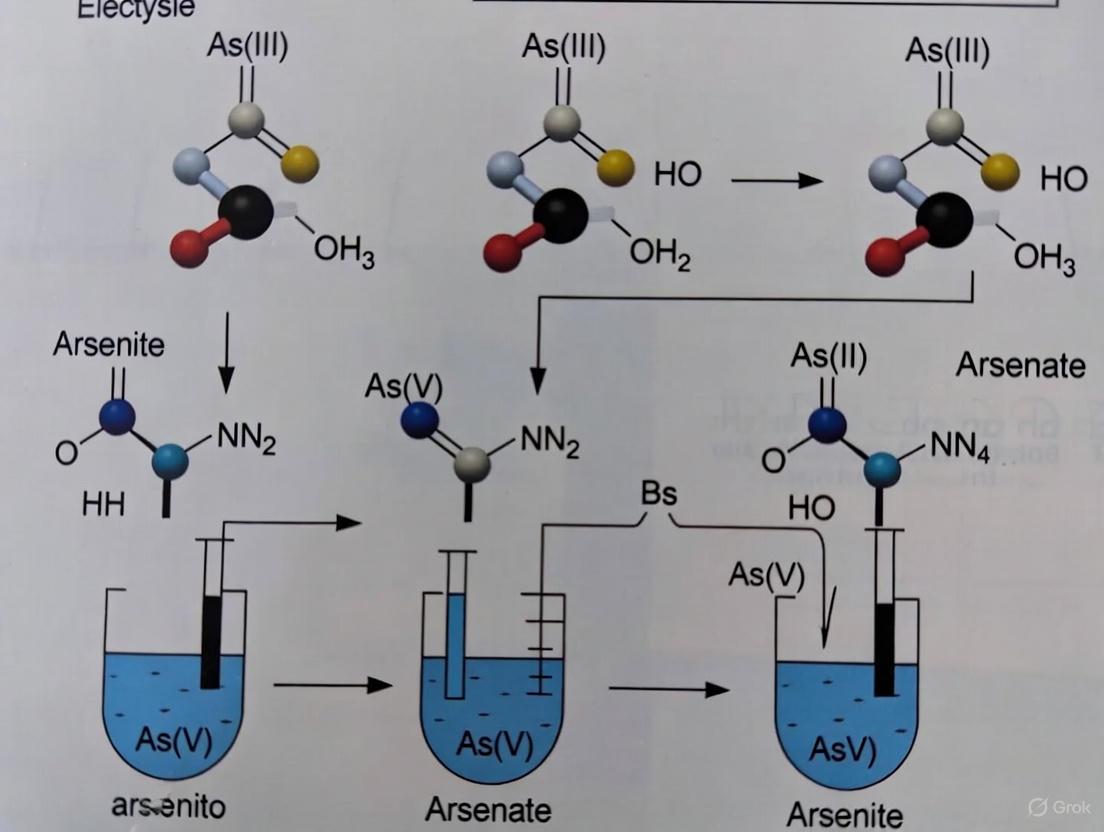
The portable ASV method for arsenic speciation leverages the different electrochemical behaviors of As(III) and As(V) species. As(III) can be directly determined at gold electrodes through electrodeposition and stripping, while As(V) requires prior reduction to As(III) or As(0) before detection [5] [6].
Two distinct operational approaches have been developed for arsenic speciation:
Selective deposition potential method: As(III) is selectively determined at a deposition potential of -0.3 V to -0.9 V, where As(V) is not electroactive. Total inorganic arsenic is then determined after electrochemical reduction of As(V) to As(0) at a more negative deposition potential (-1.2 V to -1.3 V). The As(V) concentration is calculated by difference [5] [6].
Underpotential deposition (UPD) method: This approach utilizes the UPD of As ad-atoms on gold electrodes, allowing for sensitive detection of both As(III) and total arsenic by modulating the deposition potential [6].
The following diagram illustrates the experimental workflow for portable ASV analysis of arsenic in water samples:
Detailed Experimental Protocol
Equipment and Reagents
Table 2: Research Reagent Solutions and Essential Materials
| Item | Specification | Function/Application |
|---|---|---|
| Portable Potentiostat | PalmSens4, EmStat4, or equivalent | Electrochemical measurements with USB/battery power |
| Gold Working Electrode | Rotating solid gold electrode (SGE), 2-3 mm diameter | Arsenic deposition and stripping |
| Reference Electrode | Ag/AgCl (3 M KCl) | Stable reference potential |
| Counter Electrode | Platinum wire or glassy carbon | Completes electrical circuit |
| Supporting Electrolyte | Hydrochloric acid (HCl) 0.1-1.0 M | Provides conducting medium, fixes pH |
| Standard Solutions | As(III) and As(V) stock solutions (1000 mg/L) | Calibration and quality control |
| Purified Water | Deionized water (18.2 MΩ·cm) | Solution preparation and dilution |
| Filtration System | 0.45 μm membrane filters | Sample pretreatment |
| pH Meter | Portable pH meter with combination electrode | Sample characterization |
Sample Collection and Pretreatment
- Sample Collection: Collect water samples in pre-cleaned polyethylene or polypropylene containers. Acid-wash all containers with 10% HNO₃ and rinse thoroughly with deionized water before use.
- Preservation: If immediate analysis is not possible, preserve samples by acidification to pH < 2 with high-purity HCl and store at 4°C.
- Filtration: Filter samples through 0.45 μm membrane filters to remove suspended particulates.
- pH Adjustment: Adjust sample pH to approximately 2 using concentrated HCl, if necessary, to match calibration conditions.
Instrument Setup and Calibration
Electrode Preparation:
- Polish the gold working electrode with 0.05 μm alumina slurry on a microcloth.
- Rinse thoroughly with deionized water.
- Electrochemically clean by cycling the potential between -0.3 V and +1.5 V in 0.1 M H₂SO₄ until a stable voltammogram is obtained.
Calibration Curve:
- Prepare standard solutions of As(III) in the concentration range of 1-50 μg/L in the supporting electrolyte (0.1 M HCl).
- For each standard, perform DPASV measurement using the parameters specified in Section 3.2.4.
- Plot peak current versus concentration to establish the calibration curve.
DPASV Measurement Procedure
As(III) Determination:
- Transfer 10-20 mL of sample or standard to the electrochemical cell.
- Deoxygenate by purging with high-purity nitrogen or argon for 5 minutes.
- Set the deposition potential to -0.3 V (for conventional ASV) or -0.9 V (for UPD-ASV).
- Apply the deposition potential for 60-180 seconds with solution stirring.
- After deposition, stop stirring and allow 15 seconds equilibration.
- Initiate the differential pulse stripping scan from -0.3 V to +0.3 V with the following parameters:
- Pulse amplitude: 25-50 mV
- Pulse width: 50 ms
- Step potential: 2-5 mV
- Scan rate: 10-20 mV/s
- Record the anodic stripping peak at approximately +0.1 V for As(0) to As(III) oxidation.
Total Inorganic Arsenic Determination:
- Using the same sample, set the deposition potential to -1.2 V (for conventional ASV) or -1.3 V (for UPD-ASV).
- Repeat the deposition and stripping steps as described above.
- The electrochemical reduction at this negative potential converts both As(III) and As(V) to As(0), enabling total inorganic arsenic measurement.
As(V) Calculation:
- Calculate As(V) concentration by subtracting the As(III) concentration from the total inorganic arsenic concentration.
Quality Control
- Method Blanks: Analyze reagent blanks with each batch of samples to monitor contamination.
- Quality Control Samples: Include certified reference materials or spiked samples with each analytical batch (recommended frequency: 5-10% of samples).
- Duplicate Analysis: Perform duplicate analysis on at least 10% of samples to assess precision.
- Detection Limit Verification: Regularly verify method detection limits using low-level standards.
Applications and Method Validation
Performance Characteristics
The portable ASV method for arsenic speciation has been rigorously validated against established reference methods. The table below summarizes the key performance characteristics:
Table 3: Performance Characteristics of Portable ASV Method for Arsenic Speciation
| Parameter | ASV Method | Reference Method (HG-ICP-OES) |
|---|---|---|
| Detection Limit for As(III) | 0.10 μg/L | 0.05-0.1 μg/L |
| Detection Limit for Total As | 0.10-0.80 μg/L | 0.05-0.1 μg/L |
| Linear Range | 0.5-50 μg/L | 0.1-100 μg/L |
| Precision (RSD) | 3-8% | 2-5% |
| Analysis Time | 2-5 minutes per species | 10-15 minutes per sample |
| Recovery (%) | 92-108% | 95-105% |
Studies have demonstrated excellent agreement between results obtained by portable ASV and reference methods such as hydride generation inductively coupled plasma optical emission spectroscopy (HG-ICP-OES) [5]. The method has been successfully applied to various water matrices including groundwater, surface water, and drinking water.
Advantages and Limitations
Advantages:
- Portability for field-based analysis
- Rapid analysis time (2-5 minutes per determination)
- Low detection limits (0.1-0.8 μg/L) meeting regulatory requirements
- Cost-effectiveness compared to laboratory-based techniques
- Minimal reagent consumption and waste generation
- Capability for As(III)/As(V) speciation
Limitations:
- Potential interference from other metal ions (Cu²⁺, Hg²⁺)
- Matrix effects in complex water samples
- Requires careful electrode maintenance and preparation
- Limited to inorganic arsenic speciation (does not distinguish organic arsenic species)
The development of portable ASV methods for arsenic speciation represents a significant advancement in environmental monitoring capabilities. These methods provide researchers and environmental professionals with a powerful tool for on-site determination and speciation of inorganic arsenic in aquatic systems, enabling rapid decision-making and more comprehensive environmental assessment.
The protocols outlined in this application note provide detailed methodologies for implementing portable ASV for arsenic speciation analysis. With proper validation and quality control, these methods can generate reliable data comparable to laboratory-based techniques while offering the advantages of portability, speed, and cost-effectiveness.
As arsenic contamination continues to pose global challenges, the availability of robust field-deployable analytical methods will be crucial for monitoring, risk assessment, and remediation efforts worldwide. Future developments in sensor technology and method optimization will further enhance the capabilities of portable electrochemical techniques for environmental analysis.
Within the context of developing a portable Anodic Stripping Voltammetry (ASV) method for arsenic speciation in aquatic systems, understanding the distinct chemical behaviors of inorganic arsenic species is paramount. Arsenic exists in several forms in water, primarily as arsenite (As(III)) and arsenate (As(V)). Their profound differences in toxicity and environmental mobility directly influence public health risk assessment and the design of effective remediation and monitoring strategies [7]. Accurate speciation—the differentiation and quantification of these individual forms—is therefore critical, as measuring total arsenic alone provides an incomplete and potentially misleading picture of both risk and treatability [8]. This application note details the essential characteristics of As(III) and As(V) and provides validated protocols for their analysis using a portable ASV workflow, enabling precise, on-site speciation.
Core Chemical Properties and Toxicity
The toxicity, mobility, and chemical reactivity of arsenic are fundamentally governed by its oxidation state.
- Arsenite (As(III)): This trivalent form (+3 oxidation state) is generally considered the more toxic and mobile species [7] [9]. It is highly soluble in water and has a high affinity for thiol (-SH) groups in proteins and enzymes. By binding to these groups, As(III) can inhibit critical cellular functions, such as those facilitated by pyruvate dehydrogenase, leading to impaired cellular respiration and ATP formation [9].
- Arsenate (As(V)): The pentavalent form (+5 oxidation state) is less toxic than As(III) but still poses significant health risks through long-term exposure [8] [9]. Its toxicity mechanism is primarily based on its chemical similarity to phosphate. As(V) can substitute for phosphate in biochemical reactions, for example forming unstable glucose-6-arsenate during glycolysis, which leads to a depletion of cellular energy reserves [9].
Table 1: Comparative Properties of Arsenite (As(III)) and Arsenate (As(V))
| Property | Arsenite (As(III)) | Arsenate (As(V)) |
|---|---|---|
| Oxidation State | +3 | +5 |
| Relative Toxicity | More toxic | Less toxic than As(III) |
| Primary Toxicity Mechanism | Binding to protein thiol groups, enzyme inhibition | Uncoupled oxidative phosphorylation by substituting for phosphate |
| Solubility in Water | Highly soluble [8] | Less soluble than As(III) [8] |
| Typical Environmental Context | Predominant in anoxic (low-oxygen) groundwater (e.g., deep aquifers) | Predominant in oxic (oxygen-rich) surface waters [10] |
Environmental Mobility and Behavior
The mobility of arsenic in aquatic and soil environments is strongly influenced by its speciation, which in turn is controlled by local redox conditions.
- Sorption Behavior: The binding of arsenic to mineral surfaces, particularly iron oxide minerals (e.g., amorphous iron oxide, goethite), is a key process controlling its mobility. The affinity of As(III) and As(V) for these minerals is highly pH-dependent [10].
- As(V):
- High Affinity: Below pH 5-6, As(V) is more strongly sorbed to iron oxides than As(III).
- Low Mobility: In this acidic to slightly acidic range, its mobility is limited.
- As(III):
- High Affinity: Above pH 7-8, As(III) has a higher affinity for the sorption sites.
- High Mobility: In neutral to alkaline, anoxic conditions (common in many groundwater systems), As(III) is both highly soluble and poorly sorbed, making it the primary driver of arsenic contamination in groundwater [10].
- As(V):
- Impact of Plant Life: Recent research highlights a previously underestimated pathway for arsenic release. Submerged aquatic plants can initially immobilize arsenic in sediments by forming iron plaques around their roots. However, when these plants die and decay, the sediment becomes anaerobic, promoting the dissolution of the iron plaques and the subsequent release of trapped arsenic back into the water column [11]. This reversal turns a protective sink into a significant contamination source.
Table 2: Factors Influencing Environmental Mobility of Arsenic Species
| Factor | Impact on As(III) | Impact on As(V) |
|---|---|---|
| pH | Higher mobility in neutral to alkaline conditions (pH >7) | Higher mobility in acidic conditions (pH <6) |
| Redox Potential | Stable and mobile under anoxic (reducing) conditions | Stable and mobile under oxic (oxidizing) conditions |
| Presence of Iron Oxides | Weak sorption at neutral pH; mobility can be high | Strong sorption across a wide pH range; mobility is typically lower |
| Competing Anions (e.g., Phosphate) | Moderate competition for sorption sites | Strong competition for sorption sites, can significantly increase mobility |
Analytical Method: Portable Anodic Stripping Voltammetry (ASV) for Speciation
The need for on-site, rapid arsenic speciation has led to the development of portable electrochemical methods like Anodic Stripping Voltammetry (ASV). A recently developed DPASV (Differential Pulse ASV) method provides a sensitive and cost-effective solution for field-based speciation [5].
Principle of Operation
The method leverages the different electrochemical behaviors of As(III) and As(V) on a solid gold electrode (SGE):
- Selective Detection of As(III): As(III) is electroactive and can be directly determined by ASV. It is first deposited onto the gold electrode at a potential of -0.3 V and then stripped (oxidized) during an anodic scan, producing a characteristic current peak at about +0.1 V [5].
- Indirect Determination of As(V): As(V) is not electroactive under these conditions and must first be reduced to As(0). The method uses an electrochemical reduction step at -1.2 V, powered by nascent hydrogen, to convert As(V) to As(0). This is followed by the stripping step to measure the total inorganic arsenic content (As(III) + As(V)). The As(V) concentration is then calculated by subtracting the direct As(III) measurement from the total arsenic measurement [5].
Workflow for Arsenic Speciation in Water
The following diagram illustrates the step-by-step protocol for determining As(III) and As(V) concentrations in a water sample using portable ASV.
Detailed Experimental Protocol
Method: Differential Pulse Anodic Stripping Voltammetry (DPASV) with a Solid Gold Electrode [5].
Objective: To determine the concentration of As(III) and As(V) in natural water samples.
I. Materials and Reagents
- Instrumentation: Portable potentiostat capable of DPASV.
- Working Electrode: Rotating solid gold electrode (SGE).
- Reference Electrode: Ag/AgCl (or similar).
- Counter Electrode: Platinum wire.
- Supporting Electrolyte: 1 M HCl (prepared with ultrapure water).
- Sample Vessels: Clean, polyethylene or glass vials.
- Pipettes: For precise sample handling.
Table 3: Research Reagent Solutions and Essential Materials
| Item | Function / Description |
|---|---|
| Portable Potentiostat | Core instrument for applying potentials and measuring current; enables on-site analysis. |
| Solid Gold Electrode (SGE) | Working electrode; provides a highly sensitive surface for arsenic deposition and stripping. |
| Hydrochloric Acid (HCl) | Supporting electrolyte; provides a consistent ionic strength and acidic medium for the electrochemical reaction. |
| Standard Solutions | Certified reference materials of As(III) (e.g., NaAsO₂) and As(V) (e.g., Na₂HAsO₄·7H₂O) for calibration. |
| Portable Syringe System | For sample introduction and handling in the field, compatible with methods like the As(V)-selective chemisorbent [12]. |
II. Safety Precautions
- Arsenic standards are highly toxic. Wear appropriate personal protective equipment (PPE) including gloves and safety glasses.
- Handle acids with care.
- Follow local regulations for waste disposal.
III. Procedure
Step 1: Instrument and Electrode Preparation
- Initialize the portable potentiostat according to the manufacturer's instructions.
- Clean the solid gold electrode by polishing with a 0.05 μm alumina slurry (if required) and rinse thoroughly with ultrapure water.
- Mount the electrode in the cell and set the rotation speed to a constant value (e.g., 2000 rpm).
Step 2: Calibration Curve for As(III)
- Prepare a series of standard solutions of As(III) (e.g., 0, 5, 10, 25, 50 μg L⁻¹) in the supporting electrolyte (1 M HCl).
- For each standard:
- Transfer 10 mL to the electrochemical cell.
- Deposition: Apply a deposition potential of -0.3 V vs. Ag/AgCl for a fixed time (e.g., 60-120 s) with electrode rotation.
- Equilibrium: Stop rotation and wait for 15 s.
- Stripping: Perform a differential pulse anodic scan from -0.3 V to +0.5 V.
- Record the stripping peak current at approximately +0.1 V.
- Plot a calibration curve of peak current versus As(III) concentration.
Step 3: Analysis of Water Sample for As(III)
- Filter the water sample if necessary (0.45 μm filter).
- Mix 9 mL of sample with 1 mL of concentrated HCl to achieve a final concentration of ~1 M HCl.
- Analyze this acidified sample following the exact procedure in Step 2.
- Determine the concentration of As(III) in the sample from the calibration curve.
Step 4: Analysis of Water Sample for Total Inorganic Arsenic
- Take a new aliquot of the same acidified sample.
- Electrochemical Reduction: Apply a reduction potential of -1.2 V vs. Ag/AgCl for a fixed time (e.g., 60-120 s) to reduce As(V) to As(0).
- Deposition & Stripping: Immediately after reduction, proceed with the standard DPASV sequence (deposition at -0.3 V, equilibrium, and anodic scan).
- The peak current obtained at +0.1 V now corresponds to the total inorganic arsenic (As(III) + As(V)).
Step 5: Speciation Calculation
- Calculate the concentration of As(V) by difference:
[As(V)] = [Total Inorganic As] - [As(III)]
IV. Performance Characteristics
- Limit of Detection (LOD): The method can achieve an LOD of 0.10 μg L⁻¹ for total arsenic [5].
- Selectivity: The method is highly selective for inorganic arsenic species. Validation against reference techniques like HG-ICP-OES shows satisfactory agreement [5].
Health Implications and Regulatory Context
The stark difference in toxicity between arsenic species underpins its profound public health impact. Chronic exposure to arsenic, primarily through contaminated drinking water, is a global issue affecting millions and is strongly linked to cancers, cardiovascular disease, and skin lesions [13] [9]. A landmark 20-year study in Bangladesh provided the strongest evidence to date that reducing arsenic exposure leads to a dramatic drop in death rates from these chronic diseases. The research showed that individuals who switched to water with low arsenic levels saw their mortality risk fall to match that of people who were never heavily exposed [13]. This highlights the critical importance of speciation and remediation.
Global regulations, such as the World Health Organization's guideline of 10 μg L⁻¹ for total arsenic in drinking water, are based on the combined risk posed by all inorganic species [13]. Given that As(III) is both more toxic and more mobile in typical groundwater, its presence often constitutes the primary health risk. Effective water treatment must therefore target both species, which often requires an oxidation step to convert As(III) to the more easily removable As(V) [8] [10]. The ability to perform on-site speciation with methods like portable ASV is invaluable for identifying hotspots, guiding treatment choices, and monitoring compliance.
The Limitations of Total Arsenic Analysis and the Imperative for Species-Specific Data
In environmental chemistry and toxicology, the measurement of total arsenic concentration has historically been the standard approach for risk assessment and regulatory compliance. However, this method provides fundamentally incomplete information for accurate risk evaluation, as arsenic exists in multiple chemical species with dramatically different toxicological profiles, mobilities, and biogeochemical behaviors. The limitations of total arsenic analysis become particularly critical in the context of developing portable analytical methods for arsenic speciation in aquatic systems, where understanding the precise distribution of arsenic species is essential for both public health protection and water treatment optimization.
Arsenic's toxicity is highly dependent on its chemical form. Inorganic arsenic species, particularly trivalent arsenite (As(III)), are significantly more toxic than pentavalent arsenate (As(V)) or organic forms such as dimethylarsinic acid (DMA) and arsenobetaine (AsB). [14] states that "As(III) is up to 60 times more toxic than As(V) in humans," while organoarsenicals like arsenobetaine are generally considered to have low toxicity. [15] This dramatic variation in toxicity means that measuring total arsenic alone provides insufficient information for meaningful health risk assessments, as two samples with identical total arsenic concentrations may present vastly different toxicological risks depending on their speciation profiles.
Critical Limitations of Total Arsenic Analysis
Toxicological Inadequacy
The fundamental limitation of total arsenic analysis lies in its inability to distinguish between arsenic species of differing toxicities. Regulatory agencies recognize this distinction by setting limits specifically for inorganic arsenic in food and water. [16] For instance, the International Agency for Research on Cancer (IARC) classifies inorganic arsenic as "carcinogenic to humans" (Group 1), while methylated forms like MMA and DMA are categorized as "possibly carcinogenic to humans" (Group 2B). [16] This classification underscores the critical importance of species-specific data for accurate risk assessment.
Water Treatment Optimization Challenges
The effectiveness of arsenic removal technologies in water treatment depends heavily on the specific arsenic species present. [15] explains that "since the form of arsenic species in natural waters changes with water chemistry and these species may require different technologies to facilitate their removal, understanding the arsenic species distribution can be critical to the development of effective treatment processes." Specifically, As(V) is more readily removed by conventional treatment processes such as coagulation, while As(III) often requires pre-oxidation for efficient removal. [17] Without speciation data, treatment system design becomes largely guesswork, potentially leading to inadequate arsenic removal and continued public health risks.
Environmental Behavior and Bioavailability
Arsenic species exhibit markedly different mobilities and environmental behaviors. In sediments, arsenic can exist in multiple fractions with varying bioavailability, including strongly adsorbed (AsPO₄), acid-volatile sulfide/carbonate-bound (AsHCl), Fe oxide coprecipitated (AsRe), and organic matter/sulfide-bound (AsOX) forms. [18] Research has shown that "bioavailable As (B-As) constituted 72.92 ± 4.15% of the T-As in sediments" in polluted estuarine areas, highlighting the potential ecological impact that would be masked by total arsenic measurement alone. [18] These species transformations are mediated by microbial communities whose composition and function are themselves influenced by arsenic contamination, creating complex biogeochemical cycling that cannot be understood through total arsenic concentrations alone.
Advanced Methodologies for Arsenic Speciation
Laboratory-Based Speciation Techniques
Traditional laboratory methods for arsenic speciation have relied on sophisticated instrumentation, primarily high-performance liquid chromatography coupled with inductively coupled plasma mass spectrometry (HPLC-ICP-MS). This technique provides excellent sensitivity and has been successfully applied to various matrices, including food products like rice. [16] describes an optimized HPLC-ICP-MS method that reduces "overall retention time to less than 4 minutes while enhancing peak separation" for arsenic species in rice, demonstrating the ongoing refinement of laboratory-based speciation methods.
Other established techniques include hydride generation atomic absorption spectrometry (HG-AAS) and atomic fluorescence spectrometry (HG-AFS). These methods typically require sample pretreatment to convert arsenic species to forms amenable to analysis, which may involve digestion, preconcentration, or derivatization steps. [19] details various sample preparation approaches, noting that "if organic arsenic compounds are to be determined, the species in question must be isolated," while for total arsenic determination, "the arsenic must be brought into solution and, if necessary, converted to inorganic form."
Portable Methods for Field-Based Speciation
The development of portable, field-deployable speciation methods addresses critical limitations of laboratory-based analysis, particularly the preservation of species integrity during sample transport and storage. [17] highlights that "speciation of arsenic by electrochemical means does not suffer from phosphate interference," which is a significant advantage over colorimetric methods in phosphate-rich waters.
Anodic Stripping Voltammetry (ASV) has emerged as a promising technique for portable arsenic speciation. [5] describes a differential pulse ASV (DPASV) method using a rotating solid gold electrode for the "rapid, sensitive and cost-effective determination and speciation of inorganic arsenic in aquatic environments." This approach enables direct measurement of As(III) at +0.1V after deposition at -0.3V, while total inorganic arsenic is determined after electrochemical reduction of As(V) to elemental arsenic at -1.2V. The method achieves a detection limit of 0.10 μg L⁻¹ for total arsenic, making it suitable for monitoring at regulatory levels.
Solid-Phase Extraction (SPE) techniques using selective sorbents offer another approach to portable speciation. [17] demonstrates the use of an As(V)-selective chemisorbent (ImpAs) that efficiently removes As(V) from solution while allowing As(III) to pass through unaffected. This enables speciation through measurement of total arsenic before and after treatment, with the difference representing the As(V) fraction. The method utilizes "handheld syringes, enabling high sample throughput with minimal set-up costs" and has been validated against ASV in natural waters.
Table 1: Comparison of Arsenic Speciation Methods
| Method | Detection Limit | Key Advantages | Limitations | Applications |
|---|---|---|---|---|
| HPLC-ICP-MS | <0.1 μg L⁻¹ [16] | High sensitivity, multi-species detection | Laboratory-based, expensive instrumentation | Food analysis, regulatory testing |
| Anodic Stripping Voltammetry (ASV) | 0.10 μg L⁻¹ (total As) [5] | Portable, cost-effective, no phosphate interference | Requires electrode maintenance | Field monitoring, aquatic systems |
| Selective Chemisorption (ImpAs) | Not specified | Simple, portable, high throughput | Limited to As(III)/As(V) separation | Field speciation, treatment monitoring |
| HG-AAS/AFS | ~1-10 μg L⁻¹ [19] | Established methodology, good sensitivity | Limited to hydride-forming species | Laboratory analysis |
Detailed Experimental Protocols
Protocol for Arsenic Speciation by Anodic Striammetry
Principle: This method leverages the different electrochemical behaviors of As(III) and As(V) on a gold electrode surface. As(III) is directly determined, while As(V) requires preliminary reduction before analysis. [5]
Equipment and Reagents:
- Portable potentiostat with capability for differential pulse measurements
- Rotating solid gold electrode (working electrode)
- Reference electrode (Ag/AgCl recommended)
- Counter electrode (platinum wire)
- Supporting electrolyte: 1 M HCl or appropriate buffer
- Sodium borohydride (analytical grade, low arsenic content)
- Nitrogen gas for deaeration
- Standard solutions: As(III) and As(V) at 1000 mg L⁻¹
Procedure:
- Electrode Preparation: Polish the gold electrode with 0.05 μm alumina slurry, rinse thoroughly with deionized water, and activate by cycling the potential between -0.3V and +1.5V in 0.1 M H₂SO₄ until a stable voltammogram is obtained.
As(III) Determination:
- Transfer 10 mL of sample or standard to the electrochemical cell.
- Add supporting electrolyte to maintain consistent ionic strength.
- Purge with nitrogen for 300 seconds to remove dissolved oxygen.
- Set deposition potential to -0.3V and rotate electrode at 2000 rpm for 60-180 seconds.
- After deposition, stop rotation and wait 15 seconds.
- Apply differential pulse waveform from -0.3V to +0.1V with pulse amplitude of 50 mV and step height of 4 mV.
- Measure peak current at approximately +0.1V, which corresponds to As(0) to As(III) oxidation.
Total Inorganic Arsenic Determination:
- To the same cell, apply a reduction potential of -1.2V for 60-120 seconds to electrochemically reduce As(V) to As(0).
- Follow the same stripping procedure as for As(III) determination.
- The measured signal corresponds to total inorganic arsenic (As(III) + As(V)).
Calculation:
- As(III) concentration is determined directly from step 2.
- As(V) concentration is calculated by subtracting As(III) from total inorganic arsenic.
Quality Control:
- Analyze certified reference materials with known arsenic speciation.
- Perform standard additions for matrix-matched calibration.
- Monitor electrode performance with daily calibration standards.
Protocol for Speciation Using Selective Solid-Phase Extraction
Principle: This method utilizes a selective chemisorbent (ImpAs) that retains As(V) while allowing As(III) to pass through, enabling speciation through measurement of arsenic before and after treatment. [17]
Equipment and Reagents:
- ImpAs chemisorbent material (Zn(II)-based receptor on polymeric beads)
- Handheld syringe barrels (1-10 mL) with frits
- Appropriate analytical instrument for total arsenic measurement (e.g., portable XRF, field kit)
- pH meter and buffers
- Standard solutions of As(III) and As(V)
Procedure:
- Column Preparation:
- Place a small frit at the bottom of the syringe barrel.
- Slurry the ImpAs material in deionized water and transfer to the syringe to create a bed volume of approximately 1 mL.
- Place another frit on top of the resin bed.
- Condition the column with 5 bed volumes of deionized water at pH 7.
Sample Treatment:
- Measure total arsenic concentration in the untreated sample (C_total).
- Pass 5-10 mL of sample through the ImpAs column at a flow rate of 1-2 mL min⁻¹.
- Collect the effluent and measure its arsenic concentration (C_effluent).
Calculation:
- As(V) concentration = Ctotal - Ceffluent
- As(III) concentration = C_effluent
Quality Control:
- Verify column efficiency using standard solutions of known As(III)/As(V) ratios.
- Monitor breakthrough capacity for high-arsenic samples.
- Test for As(III) oxidation during the process using spiked samples.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Essential Research Reagents for Arsenic Speciation Analysis
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Solid Gold Electrode | Working electrode for ASV measurements | Provides sensitive arsenic detection; requires regular polishing and activation [5] |
| Sodium Borohydride (NaBH₄) | Reducing agent for hydride generation | Critical for HG-based methods; arsenic contamination in reagent can affect blanks [19] |
| ImpAs Chemisorbent | Selective As(V) sorbent for SPE | Zn(II)-based receptor immobilized on polymeric beads; operates at pH 6-8 [17] |
| C18 Reverse-Phase Column | Chromatographic separation | Used with ion-pairing agents for HPLC-ICP-MS speciation [16] |
| Ammonium Pyrrolidine Dithiocarbamate | Chelating agent for extraction | Enables selective extraction of trivalent arsenic at pH 2-6 [19] |
| Nitric Acid (High Purity) | Sample digestion and preservation | Essential for preventing arsenic loss during storage; prevents volatilization of trivalent arsenic [19] |
Analytical Considerations and Quality Assurance
Sample Collection and Preservation
Proper sample handling is critical for accurate arsenic speciation, as species interconversion can occur rapidly after collection. [19] emphasizes that "the investigator must always be aware of the possibility of losing some of the species of interest through adsorption on vessel walls or on suspended matter or through volatilization." For aqueous samples, acidification with high-purity nitric acid to pH <2 is recommended to preserve species integrity, though this may not prevent all transformations. [17] notes the particular challenge of "the progressive oxidation of As(III) on an hours-to-days timescale" in field samples, highlighting the advantage of on-site speciation methods.
Method Validation
Validation of arsenic speciation methods should include:
- Analysis of Certified Reference Materials (CRMs) with certified species concentrations [16]
- Recovery studies using samples spiked with known concentrations of As(III) and As(V)
- Comparison with reference methods such as HPLC-ICP-MS [17]
- Assessment of method detection limits and quantification limits for each species
- Evaluation of precision through replicate analyses
For portable methods, validation under field conditions is particularly important, as environmental matrices may contain interfering substances that affect analytical performance.
The limitations of total arsenic analysis are significant and multifaceted, spanning toxicological, treatment, and biogeochemical considerations. The imperative for species-specific data drives the development and application of sophisticated analytical methods, including emerging portable techniques like anodic stripping voltammetry and selective solid-phase extraction. These advancements enable more accurate risk assessments, optimized treatment strategies, and improved understanding of arsenic cycling in aquatic systems. As analytical technologies continue to evolve, particularly in the realm of field-deployable speciation methods, our ability to address the global challenge of arsenic contamination will be greatly enhanced through access to species-specific data that reflects the true environmental behavior and health impacts of this complex contaminant.
Anodic Stripping Voltammetry (ASV) is a highly sensitive electrochemical technique renowned for its capability to detect heavy metals and metalloids at trace levels (parts-per-billion, ppb, or lower) [20]. Its application is crucial in environmental monitoring, food safety, and pharmaceutical development, where precise speciation—differentiating between chemical forms of an element—is essential for accurate toxicity and bioavailability assessment [21]. The core principle of ASV involves a two-stage process: the electrochemical reduction and pre-concentration of metal ions onto an electrode surface, followed by their subsequent oxidative stripping back into solution, which generates the analytical signal [20]. The selectivity and sensitivity of ASV make it particularly suitable for speciation analysis, enabling the differentiation of inorganic arsenic species, such as the highly toxic arsenite (As(III)) and the less toxic arsenate (As(V)) [5] [22]. This document details the core principles, protocols, and applications of ASV, framed within research on portable methods for arsenic speciation in aquatic systems.
Core Principles of ASV
The exceptional sensitivity of ASV, which often surpasses other analytical techniques for metal analysis, is achieved through its distinctive two-step methodology: a pre-concentration step followed by a stripping step [20].
The Two-Step ASV Process
The fundamental process of ASV is illustrated in the diagram below.
- Pre-concentration (Electrodeposition): The first step involves immersing the working electrode in the sample solution and applying a constant potential that is sufficiently negative to reduce the target metal ions (Mⁿ⁺) to their elemental state (M⁰). During this period, which can last from seconds to minutes, the reduced metal atoms are pre-concentrated onto or into the electrode surface. This step effectively enriches the analyte at the electrode, which is the key to achieving very low detection limits [20].
- Stripping (Anodic Dissolution): Following the deposition period, the potential is swept in an anodic (positive) direction. As the potential reaches the oxidation potential of the deposited metal, the metal atoms are stripped from the electrode surface and re-oxidized back into solution as ions. This oxidation process generates a measurable current. The resulting peak current (or the charge under the peak) is directly proportional to the concentration of the metal in the original sample solution [20].
Key Parameters Influencing ASV Performance
- Deposition Potential (
E_dep): Must be negative enough to reduce the target metal ion but not so negative as to cause interference from other species or hydrogen evolution [20]. - Deposition Time (
t_dep): Longer deposition times increase the amount of metal deposited, lowering the detection limit but increasing analysis time and potential for surface fouling [20]. - Mass Transport: Stirring or rotating the electrode during deposition is crucial to enhance the flux of metal ions to the electrode surface, improving reproducibility and sensitivity [5] [23].
- Electrode Material: The choice of working electrode is critical, as it must provide a favorable environment for the deposition and stripping of the target analyte (see Section 2.3).
- Solution Composition: The pH, electrolyte, and buffer can affect metal speciation, stability, and the electrochemical window available for analysis [20].
Electrode Materials for ASV and Speciation
The choice of working electrode is paramount in ASV. While mercury electrodes were historically preferred for their wide cathodic window and formation of homogenous amalgams, toxicity concerns have driven the development of solid electrodes [20]. The table below compares common electrode materials used in ASV, with a focus on arsenic speciation.
Table 1: Comparison of Electrode Materials for ASV in Metal and Metalloid Speciation
| Electrode Material | Key Advantages | Limitations / Challenges | Exemplary Application in Speciation |
|---|---|---|---|
| Gold (Au) Electrode (Solid or microwire) | - High sensitivity and electrocatalytic activity for arsenic [5] [22].- Forms well-defined intermetallic compounds with As(0), facilitating detection [22].- Suitable for portable systems. | - Surface oxide formation can interfere [20].- Requires careful potential control and surface renewal. | - Direct determination of As(III) at +0.1 V (vs. ref.) after deposition at -0.3 V [5].- Determination of total inorganic As after electrochemical reduction of As(V) at -1.2 V [5] [23]. |
| Gold Nanoparticle (AuNP) Modified Electrodes | - Enhanced surface area and sensitivity [24].- Can be fabricated on disposable screen-printed electrodes (SPEs). | - Fabrication complexity and stability over time. | - Used with L-cysteine modification for sensitive As(III) detection in complex matrices like rice [24]. |
| Thoria Nanoparticles-Carbon Paste Electrode | - Rapid procedure with high selectivity and sensitivity for As(III) [25].- Low detection limit (0.1 μg L⁻¹). | - Material preparation and electrode reproducibility. | - Direct determination of arsenite and total inorganic arsenic in water samples [25]. |
| Bismuth (Bi) Film Electrodes | - Environmentally friendly [20].- Wide operational potential window and well-defined stripping signals for many metals. | - Performance for arsenic speciation is less established compared to gold. | - Not prominently featured in the provided arsenic speciation literature, but a common Hg replacement for other metals. |
ASV for Arsenic Speciation in Aquatic Systems
The toxicity of arsenic is highly dependent on its chemical form, with inorganic arsenite (As(III)) being significantly more toxic than arsenate (As(V)) [17] [21]. Therefore, simply measuring total arsenic content is insufficient for an accurate risk assessment. ASV provides a powerful tool for inorganic arsenic speciation.
Speciation Workflow for Inorganic Arsenic
The general methodology for differentiating As(III) and As(V) using ASV is outlined below.
Detailed Experimental Protocol for Arsenic Speciation by ASV
This protocol is adapted from recent research on portable methods for arsenic determination in aquatic environments [5] [22].
Objective: To determine the concentration of As(III) and As(V) in a natural water sample using a solid gold electrode and differential pulse anodic stripping voltammetry (DPASV).
I. Materials and Reagents Table 2: Research Reagent Solutions and Essential Materials
| Item | Function / Explanation |
|---|---|
| Portable or Benchtop Potentiostat | Instrument for applying potentials and measuring currents. |
| Solid Gold Working Electrode (e.g., rotating gold disk or gold microwire) | High electrocatalytic activity for arsenic deposition/stripping [5] [22]. |
| Platinum Counter Electrode | Completes the electrical circuit in the electrochemical cell. |
| Reference Electrode (e.g., Ag/AgCl) | Provides a stable, known potential reference. |
| Supporting Electrolyte (e.g., HCl 1-5 M, or acetate buffer with chloride) [22] [23] | Provides ionic conductivity and can define the medium for electrochemical reactions. Acidic conditions or presence of chloride facilitate As(V) reduction [22]. |
| Potassium Permanganate (KMnO₄) Solution (optional, for alternative method) | Acts as a chemical oxidant to convert As(III) to As(V) for indirect speciation [22]. |
| Standard Solutions of As(III) and As(V) (e.g., 1000 mg L⁻¹) | Used for calibration and standard addition methods. |
| Ultrapure Water (>18 MΩ·cm) | Prevents contamination from impurities in blanks and solutions. |
II. Procedure
Sample Preparation:
- Collect water samples in clean, acid-washed containers. If possible, analyze on-site to avoid species transformation during storage [17].
- Filter the sample if necessary to remove suspended solids.
- For the supporting electrolyte, either acidify the sample to a consistent pH with concentrated HCl (e.g., to 1 M HCl) or add an appropriate buffer (e.g., acetate buffer to pH 4.7 with 0.25 M chloride) [22] [23].
Instrumental Setup:
- Set up the three-electrode system in the sample solution.
- Select the Differential Pulse (DP) mode for the stripping step to enhance sensitivity and resolution.
- Optimized parameters for a gold electrode may include [5]:
- Deposition Potential (Edep): -0.3 V (for As(III)) or -1.2 V (for total inorganic As)
- Deposition Time (tdep): 60-120 seconds (with stirring/rotation)
- Equilibration Time: 10 seconds (no stirring)
- DP Pulse Parameters: Pulse amplitude 50 mV, pulse time 50 ms, step height 2 mV.
Determination of As(III):
- Transfer an aliquot of the prepared sample to the electrochemical cell.
- Apply a deposition potential of -0.3 V (vs. Ag/AgCl) for a fixed time (e.g., 120 s) while stirring. At this potential, As(III) is selectively reduced to As(0) and deposited on the gold electrode.
- After a quiet period, initiate the DP anodic scan from -0.3 V to a more positive potential (e.g., +0.4 V).
- Record the voltammogram. The stripping peak for As(III) typically appears around +0.1 V to +0.3 V (depending on the electrode and medium) [5].
- Measure the peak current (ip).
Determination of Total Inorganic Arsenic:
- Using the same or a new aliquot of the sample, apply a deposition potential of -1.2 V for a fixed time. This more negative potential generates "nascent hydrogen" at the electrode surface, which chemically reduces As(V) to As(0), allowing it to be co-deposited with As(III) [5] [23].
- Perform the anodic stripping scan as before. The resulting peak corresponds to the total inorganic arsenic content (As(III) + As(V)).
Quantification and Data Analysis:
- Calibration: Construct a calibration curve by analyzing standard solutions of As(III) with known concentrations under the same conditions as step 3. Alternatively, use the method of standard additions by spiking the sample with known amounts of As(III) standard. This method is preferred for complex matrices like natural waters as it compensates for matrix effects [20].
- Calculation:
- As(III) concentration is determined directly from the analysis in step 3.
- Total Inorganic As concentration is determined from the analysis in step 4.
- As(V) concentration is calculated by subtraction:
[As(V)] = [Total Inorganic As] - [As(III)].
Validation and Comparison with Other Techniques
ASV methods for arsenic have been rigorously validated against established spectroscopic techniques. For instance, results for arsenic speciation in real water samples obtained by DPASV using a gold electrode showed "satisfactory agreement" with those from hydride generation coupled with inductively coupled plasma atomic emission spectroscopy (HG-ICP-OES) [5]. Another study reported "very good agreement" (slope = +1.029, R² = 0.99) between a voltammetric method using a gold microwire and inductively coupled plasma-mass spectrometry (ICP-MS) for groundwater analysis [22].
The primary advantages of ASV over these laboratory-based techniques are its portability, which enables on-site analysis and avoids species changes during transport; its low cost and minimal power requirements; and its ability to provide information on the labile fraction of metals, which is often more relevant for toxicity assessments than total acid-digested concentration [20].
Portable Anodic Stripping Voltammetry (ASV) represents a significant advancement in environmental monitoring, enabling rapid, sensitive, and cost-effective determination of toxic elements such as arsenic in aquatic systems [5]. This technique is particularly valuable for speciation analysis, distinguishing between highly toxic arsenite (As(III)) and less toxic arsenate (As(V)) forms, which is crucial for accurate risk assessment [5]. The method's portability facilitates real-time, on-site analysis, eliminating the need for sample transportation and preservation while providing immediate data for decision-making in field research and public health initiatives [5] [26].
Key Advantages of Portable ASV
Operational Speed and Rapid Analysis
Portable ASV significantly reduces analysis time compared to traditional laboratory methods. The electrochemical reduction approach for arsenic speciation minimizes procedural steps, enabling faster determination of arsenic species in field conditions [5]. Researchers can obtain quantitative speciation data directly on-site without the delays associated with transporting samples to centralized laboratories.
Cost-Effectiveness and Economic Efficiency
The portable ASV method offers substantial cost advantages over traditional analytical techniques:
Table 1: Cost Comparison of Arsenic Speciation Methods
| Method Component | Portable ASV | Traditional Laboratory Methods |
|---|---|---|
| Instrumentation | Portable potentiostat [5] | ICP-OES, HPLC-ICP-MS |
| Per-sample Cost | Minimal reagent consumption [5] | High reagent and gas consumption |
| Infrastructure | Field-deployable; no lab facility needed | Requires specialized laboratory facilities |
| Personnel Costs | Single operator for field analysis | Multiple specialized technicians |
| Sample Transport | Eliminated | Significant cost for proper sample preservation and transport |
The economic benefits extend beyond instrumentation. Portable ASV minimizes chemical reagent consumption through electrochemical reduction instead of chemical reduction, substantially reducing ongoing operational expenses [5]. Laser-derived graphene electrodes, used in similar heavy metal detection applications, can be produced for less than $0.01 per electrode, demonstrating the remarkable cost efficiency achievable with advanced field-deployable sensor technologies [27].
Superior On-Site Capability for Field Deployment
Portable ASV systems are specifically engineered for field deployment, offering robust performance in diverse environmental conditions. The method has been successfully validated for direct quantitative determination and speciation of inorganic arsenic in real water samples, showing satisfactory agreement with laboratory-based reference methods like hydride generation technique coupled with inductively coupled plasma atomic emission spectroscopy (HG-ICP-OES) [5]. This capability enables researchers to conduct high-frequency spatial and temporal monitoring campaigns that would be prohibitively expensive and time-consuming using conventional laboratory-based approaches.
Experimental Protocols for Arsenic Speciation Using Portable ASV
Principle of Operation
The portable ASV method for arsenic speciation utilizes a solid gold electrode (SGE) as the working electrode. The fundamental principle involves the electrochemical reduction and subsequent oxidation of arsenic species at controlled potentials:
- As(III) Determination: As(III) is selectively determined at +0.1 V by ASV after deposition at −0.3 V [5]
- Total Arsenic Determination: Total As content (As(V) + As(III)) is determined after electrochemical reduction at −1.2 V by nascent hydrogen to As⁰ [5]
- As(V) Quantification: As(V) concentration is evaluated indirectly by subtracting As(III) from total As [5]
Detailed Analytical Protocol
Equipment and Reagents Setup
- Portable potentiostat with capability for differential pulse anodic stripping voltammetry (DPASV) [5]
- Solid gold electrode (SGE) as working electrode [5]
- Reference electrode and counter electrode
- Acetate-buffered solution (0.1 M, pH 4) as supporting electrolyte [5]
- Standard solutions of As(III) and As(V) for calibration
Sample Preparation Protocol
- Collect water samples following standard environmental sampling procedures
- Filter samples through 0.45 μm membrane filters if particulate matter is present
- Adjust sample pH to 4.0 using acetic acid/sodium acetate buffer
- For total arsenic determination, acidify a separate aliquot to pH <2 with high-purity HCl
Instrumental Analysis Procedure
- Electrode Conditioning: Clean the gold electrode by applying a conditioning potential in supporting electrolyte
- As(III) Determination:
- Set deposition potential to −0.3 V with deposition time of 60-180 seconds
- Apply differential pulse waveform with pulse amplitude of 50 mV and pulse width of 50 ms
- Record stripping peak at approximately +0.1 V corresponding to As(III)
- Total Arsenic Determination:
- Set electrochemical reduction potential to −1.2 V for 60-120 seconds
- Apply the same DPASV parameters as for As(III) determination
- Record combined stripping peak for total arsenic
- Data Analysis:
- Calculate As(III) concentration from direct measurement
- Determine total arsenic from combined peak after electrochemical reduction
- Compute As(V) by difference: As(V) = Total As - As(III)
Figure 1: ASV Arsenic Speciation Workflow
Quality Control and Validation
- Perform triplicate measurements for each sample
- Use standard addition method for quantification in complex matrices
- Validate method performance with certified reference materials
- Maintain detection limit of 0.10 μg L⁻¹ for As(tot) [5]
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Essential Research Reagents and Materials for Portable ASV
| Reagent/Material | Function/Application | Specifications/Notes |
|---|---|---|
| Solid Gold Electrode (SGE) | Working electrode for arsenic detection | Enables selective As(III) determination at +0.1 V [5] |
| Acetate Buffer Solution | Supporting electrolyte (pH 4.0) | 0.1 M concentration for optimal arsenic response [5] |
| Arsenic Standard Solutions | Calibration and quantification | Separate As(III) and As(V) standards for speciation [5] |
| Portable Potentiostat | Instrumentation for voltammetric measurements | Enables DPASV measurements in field conditions [5] |
| Laser-Derived Graphene Electrodes | Alternative electrode material | Cost-effective option (<$0.01 per electrode) [27] |
| Nascent Hydrogen | Electrochemical reduction agent | Reduces As(V) to As⁰ at −1.2 V for total arsenic determination [5] |
Method Performance and Validation
The portable ASV method demonstrates excellent analytical performance for environmental monitoring applications:
Table 3: Performance Characteristics of Portable ASV for Arsenic Speciation
| Performance Parameter | Specification/Value | Significance |
|---|---|---|
| Detection Limit (As(tot)) | 0.10 μg L⁻¹ [5] | Below WHO guideline values for drinking water |
| Linear Range | Up to 1000 ppb for As species [5] | Suitable for both environmental and contaminated samples |
| Method Correlation | Agreement with HG-ICP-OES [5] | Validated against standard laboratory methods |
| Analysis Time | Rapid determination [5] | Enables high-throughput field screening |
| Selectivity | Distinguishes As(III) and As(V) [5] | Provides toxicologically relevant speciation data |
Figure 2: Portable ASV Advantage Framework
Applications in Environmental Research and Public Health
Portable ASV for arsenic speciation addresses critical needs in environmental monitoring and public health protection. The method enables researchers to conduct comprehensive arsenic contamination assessments in diverse aquatic systems, from groundwater to surface waters [5]. This capability is particularly valuable for identifying arsenic hotspots and monitoring remediation efforts in real-time. The technology supports public health initiatives by providing rapid assessment of drinking water sources in arsenic-affected regions, enabling timely interventions to reduce exposure risks [26]. Furthermore, the method's cost-effectiveness allows for expanded monitoring networks in resource-limited settings where arsenic contamination poses significant health threats.
Deploying Portable ASV in the Field: A Step-by-Step Protocol for Aquatic System Analysis
Within the context of a broader thesis on portable analytical methods for arsenic speciation in aquatic systems, this application note details the configuration and operation of a portable Anodic Stripping Voltammetry (ASV) system. The toxicity of arsenic is highly dependent on its chemical form, with inorganic arsenite (As(III)) being significantly more toxic than arsenate (As(V)) [28] [17]. This makes speciation—the differentiation and quantification of individual arsenic species—critically important for accurate environmental and health risk assessments [2]. While laboratory-based techniques like HPLC-ICP-MS are the gold standard for speciation [28], portable ASV offers a robust, cost-effective, and sensitive alternative for rapid, on-site determination and speciation of inorganic arsenic in water samples [5].
This protocol provides a detailed methodology for researchers and scientists to perform differential pulse anodic stripping voltammetry (DPASV) for the direct determination of As(III) and the indirect quantification of As(V) in natural waters.
Principle of ASV for Arsenic Speciation
Anodic Stripping Voltammetry for arsenic speciation is a two-step electrochemical process. First, a deposition potential is applied to a solid working electrode, reducing and concentrating dissolved arsenite (As(III)) from the solution onto the electrode surface as elemental arsenic (As(0)). Following this preconcentration step, the potential is scanned in an anodic (positive) direction, re-oxidizing the deposited metal back into solution. The current generated during this stripping step is measured, with the peak current being proportional to the concentration of As(III) in the original sample [5].
The speciation of total inorganic arsenic is achieved by exploiting the different electrochemical behaviors of As(III) and As(V). As(III) is selectively determined directly. The total inorganic arsenic content (As(III) + As(V)) is then measured after an in-situ electrochemical reduction of As(V) to As(0) at a highly negative potential, utilizing nascent hydrogen. The As(V) concentration is subsequently calculated by subtracting the As(III) concentration from the total inorganic arsenic concentration [5]. This approach minimizes chemical reagent consumption and is well-suited for field analysis.
Workflow for Arsenic Speciation using Portable ASV
System Configuration and Essential Equipment
Configuring a portable ASV system for arsenic analysis requires careful selection of components to ensure sensitivity, reproducibility, and field-portability.
System Architecture of a Portable ASV Analyzer
Core Instrumentation and Research Reagents
Table 1: Essential Equipment and Reagents for Portable ASV Arsenic Speciation
| Item Name | Specification/Type | Critical Function in the Protocol |
|---|---|---|
| Portable Potentiostat | With capacity for DPASV and software control | Applies precise deposition potentials and scans stripping potentials; measures resultant current [5]. |
| Solid Gold Working Electrode | Rotating disk electrode (RDE) preferred | Working electrode for As deposition/stripping; gold surface provides excellent electrocatalytic activity for arsenic [5] [17]. |
| Reference Electrode | Ag/AgCl (with KCl electrolyte) | Provides a stable, known reference potential for the electrochemical cell [5]. |
| Counter Electrode | Platinum wire or coil | Completes the electrical circuit in the three-electrode setup [5]. |
| Supporting Electrolyte | High-purity HCl or HNO₃ | Provides conductive medium and defines solution pH for optimal arsenic response [5]. |
| Standard Solutions | 1000 mg L⁻¹ As(III) and As(V) | Used for instrument calibration and quality control [5] [17]. |
| Purified Water | Type I (18.2 MΩ·cm) | Preparation of all solutions and rinsing to prevent contamination [5]. |
| Inert Gas Supply | High-purity Nitrogen or Argon | Deaeration of samples to remove dissolved oxygen, which causes interfering electrochemical signals [5]. |
Experimental Protocols
Reagent Preparation and Safety
- Supporting Electrolyte (1 M HCl): Carefully add 83 mL of concentrated hydrochloric acid (ACS grade) to approximately 800 mL of purified water in a fume hood. Allow to cool, then dilute to 1 L with water.
- Arsenic Stock Solutions (1000 mg L⁻¹): Commercially available certified standard solutions are recommended. Alternatively, prepare As(III) stock by dissolving 1.320 g of As₂O₃ (arsenic trioxide, highly toxic) in a minimal volume of 1 M NaOH, then acidify with HCl and dilute to 1 L with 1% HCl. As(V) stock can be prepared from Na₂HAsO₄·7H₂O.
- Working Standards: Prepare fresh working standards daily in the supporting electrolyte (e.g., 1 M HCl) by serial dilution of the stock solutions. The typical calibration range is 1–50 µg L⁻¹.
Safety Note: Arsenic standards are highly toxic. Personal protective equipment (PPE) including gloves and lab coats must be worn. All waste should be collected and disposed of as hazardous chemical waste.
Detailed DPASV Measurement Procedure for Arsenic Speciation
- System Setup: Power on the portable potentiostat and connect the computer. Install the solid gold working electrode, Ag/AgCl reference electrode, and platinum counter electrode in the cell. Set the electrode rotation speed to 2000 rpm (if using an RDE).
- Sample Preparation: Pipette 10 mL of filtered water sample or standard into the electrochemical cell. Add an equal volume of 2x supporting electrolyte (e.g., 2 M HCl) to achieve a final concentration of 1 M HCl. Place the cell in the stand and introduce the electrodes.
- Solution Deaeration: Bubble high-purity nitrogen or argon through the solution for a minimum of 10 minutes to remove dissolved oxygen. Maintain a blanket of inert gas over the solution during analysis.
- Direct Determination of As(III):
- Conditioning: Apply a conditioning potential of +0.5 V for 30 s without rotation to clean the electrode surface.
- Deposition: Rotate the electrode and apply a deposition potential of -0.3 V for 60-180 s (optimize for sensitivity). This step reduces As(III) to As(0) and deposits it onto the gold electrode.
- Equilibration: Stop rotation and gas flow. Allow the solution to become quiescent for 15 s.
- Stripping: Initiate the differential pulse anodic stripping scan from -0.3 V to +0.4 V. The peak for As(0) → As(III) oxidation will appear at approximately +0.1 V [5].
- Cleaning: After measurement, hold the potential at +0.5 V for 30 s with rotation to strip off any residual deposited material.
- Determination of Total Inorganic Arsenic:
- Use the same sample solution from step 4.
- Electrochemical Reduction: Apply a potential of -1.2 V for 60-120 s without rotation. This step uses nascent hydrogen to electrochemically reduce As(V) to As(0) [5].
- Deposition & Stripping: Immediately proceed with the deposition (at -0.3 V, with rotation) and stripping scan as described in Step 4. The measured peak current now corresponds to the total inorganic arsenic (As(III) + As(V)).
- Calculation of As(V): The concentration of As(V) is determined by difference.
- As(V) Concentration = Total Inorganic As Concentration - As(III) Concentration
Optimized DPASV Parameters
Table 2: Optimized Operational Parameters for Arsenic Speciation by DPASV [5]
| Parameter | For As(III) Determination | For Total Inorganic As |
|---|---|---|
| Deposition Potential (E_dep) | -0.3 V | -0.3 V (post-reduction) |
| Deposition Time (t_dep) | 60 - 180 s | 60 - 180 s |
| Electrode Rotation | 2000 rpm | 2000 rpm |
| Reduction Potential | Not Applied | -1.2 V |
| Reduction Time | Not Applied | 60 - 120 s |
| Stripping Scan Range | -0.3 V to +0.4 V | -0.3 V to +0.4 V |
| Peak Potential (As) | ~ +0.1 V | ~ +0.1 V |
| Supporting Electrolyte | 1 M HCl | 1 M HCl |
Performance and Validation
The described DPASV method has been validated against standard spectroscopic techniques. A limit of detection (LOD) of 0.10 µg L⁻¹ for total arsenic has been achieved, which is well below the WHO guideline value of 10 µg L⁻¹ for drinking water [5] [29]. Analysis of real water samples showed satisfactory agreement with results from hydride generation coupled with inductively coupled plasma atomic emission spectroscopy (HG-ICP-OES), confirming the method's accuracy for environmental application [5]. The precision, expressed as relative standard deviation (RSD), is typically below 10% [5] [30].
This protocol provides a reliable foundation for the on-site speciation of inorganic arsenic, enabling researchers to generate high-quality data for environmental monitoring and risk assessment directly in the field.
Within the development of portable anodic stripping voltammetry (ASV) methods for arsenic speciation in aquatic systems, electrode selection and preparation are paramount. Gold-based electrodes have emerged as the benchmark substrate for the detection of arsenic, particularly the more toxic arsenite (As(III)) species, due to their excellent electrocatalytic properties, high sensitivity, and suitability for field-portable instrumentation [5] [31]. Their superior performance is attributed to a favorable interaction with arsenic atoms, which facilitates the pre-concentration and stripping steps central to ASV. This application note provides a detailed protocol for the selection, pre-treatment, and application of gold electrodes to achieve reliable, sensitive, and speciated detection of inorganic arsenic in water samples.
Gold Electrodes as the Benchmark: Performance and Comparison
The efficacy of gold electrodes for arsenic detection is well-documented across various configurations and modifications. Table 1 summarizes key performance metrics from recent studies, demonstrating that gold-based electrodes consistently achieve detection limits well below the World Health Organization (WHO) guideline of 10 µg L⁻¹ for arsenic in drinking water [32].
Table 1: Performance Comparison of Gold-Based Electrodes for As(III) Detection
| Electrode Type | Technique | Linear Range (µg L⁻¹) | Limit of Detection (µg L⁻¹) | Key Features | Source |
|---|---|---|---|---|---|
| Solid Gold Electrode | DPASV | N/R | 0.10 (As(total)) | Applicable for speciation of As(III) and As(V); portable potentiostat compatible. [5] | |
| Gold Nanotextured Electrode (Au/GNE) | SWASV | 0.1 - 9 | 0.08 - 0.1 | High sensitivity (39.54 µA ppb⁻¹ cm⁻²); excellent selectivity against interfering ions. [32] | |
| Au Macroelectrode with UPD | ASV | 0.4 - 7.5 | ~0.4 | Utilizes underpotential deposition (UPD); no interference from Cu(II) or Cl⁻. [33] | |
| Rotating Gold Microwire Electrode | SWASV | 1.0 - 100 | 0.21 | Enhanced mass transport; suitable for complex matrices like shellfish digests. [34] | |
| scTRACE Gold (Commercial) | SWASV | 0.9 - 10 | 0.9 | Standardized method for mineral water; optimized for portable VA analyzer. [35] |
The fundamental advantage of gold lies in its ability to form an intermetallic compound or an adlayer with deposited arsenic, which results in a well-defined and sensitive stripping peak [31]. Furthermore, the surface properties of gold can be engineered through simple electrochemical pre-treatments to create nanostructures, significantly increasing the electroactive surface area and enhancing the signal. For instance, one study developed a nanotextured gold electrode (Au/GNE) via electrochemical oxidation-reduction cycles, which achieved an exceptional sensitivity of 39.54 µA ppb⁻¹ cm⁻² [32].
Experimental Protocols for Electrode Preparation and Measurement
Electrochemical Activation of Solid Gold Electrodes
A critical step to ensure reproducible and sensitive results is the electrochemical activation of the gold electrode surface. The following protocol is adapted for a standard three-electrode system (Gold Working Electrode, Platinum Counter Electrode, Ag/AgCl Reference Electrode).
Procedure:
- Initial Cleaning: Polish the solid gold electrode surface with an aqueous alumina slurry (e.g., 0.05 µm) on a microcloth pad. Rinse thoroughly with deionized water.
- Electrochemical Cleaning: Place the electrode in a 0.5 M H₂SO₄ solution. Perform cyclic voltammetry (CV) by scanning the potential between -0.2 V and +1.5 V (vs. Ag/AgCl) at a scan rate of 100 mV s⁻¹ for 20-50 cycles until a stable, characteristic gold CV profile is obtained [5] [32].
- Rinsing: After activation, rinse the electrode thoroughly with deionized water to remove any residual acid.
Fabrication of a Gold Nanotextured Electrode (Au/GNE)
For researchers requiring ultra-low detection limits, fabricating a nanotextured surface is highly effective.
Procedure:
- Substrate Preparation: Begin with a gold foil or a gold disk electrode and clean it as described in section 3.1.
- Nanotexturing: Immerse the electrode in a metal-ion-free electrolyte, such as 0.5 M H₂SO₄. Apply repeated electrochemical oxidation-reduction sweeps (e.g., CV) over a suitable potential window. This process generates a nanotextured surface with a high density of active sites, as confirmed by scanning electron microscopy (SEM) [32].
- Characterization: Validate the increased surface area by measuring the capacitance via CV in a non-Faradaic potential region.
Speciated Arsenic Detection via Anodic Stripping Voltammetry
This protocol outlines the determination of As(III) and total inorganic arsenic using a gold electrode, allowing for the calculation of As(V) by difference [5].
Reagents:
- Supporting electrolyte: 1 M HCl or 0.1 M HNO₃ is commonly used.
- Standard solutions of As(III) (from NaAsO₂) and As(V) (from Na₂HAsO₄·7H₂O).
- High-purity deionized water (18.2 MΩ·cm).
Instrumentation:
- Potentiostat (portable or benchtop).
- Three-electrode system: Gold Working Electrode, Ag/AgCl Reference Electrode, Platinum Counter Electrode.
- If using a rotating electrode, a modulated speed rotator.
Procedure: A. Direct Determination of As(III)
- Transfer a known volume of the water sample and supporting electrolyte to the electrochemical cell. Deoxygenate the solution by purging with high-purity nitrogen or argon for 300 seconds.
- Deposition: Hold the working electrode at a deposition potential of -0.3 V to +0.1 V (vs. Ag/AgCl) for 60-90 seconds with stirring or rotation. This step selectively reduces and deposits As(III) as As(0) onto the gold surface [5] [33].
- Stripping: After an equilibrium period (10-15 seconds), initiate an anodic potential scan (e.g., Differential Pulse or Square Wave mode) from the deposition potential to a more positive potential (~+0.4 V). The oxidation of deposited As(0) back to As(III) produces a characteristic current peak.
- Quantification: Measure the peak current, which is proportional to the concentration of As(III) in the sample. Use the standard addition method for quantification.
B. Determination of Total Inorganic Arsenic
- To the same sample, apply a more negative pre-reduction potential (e.g., -1.2 V) for a short duration. This electrochemically reduces As(V) to As(0) on the electrode surface via nascent hydrogen [5].
- Repeat the stripping step (A3). The resulting peak current corresponds to the total inorganic arsenic content (As(III) + As(V)).
- Speciation Calculation: The As(V) concentration is determined by subtracting the As(III) concentration from the total inorganic arsenic concentration.
Workflow Visualization
The following diagram illustrates the logical workflow for arsenic speciation using a gold electrode, from sample preparation to final quantification.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of this protocol relies on the use of specific, high-quality materials. Table 2 lists the essential reagents and their functions in the analysis.
Table 2: Key Research Reagent Solutions and Materials
| Item | Specifications / Example | Primary Function in Protocol |
|---|---|---|
| Gold Working Electrode | Solid gold electrode (SGE), gold microwire, or gold foil. | Primary sensing substrate; provides electrocatalytic surface for As deposition and stripping. [5] [34] |
| Supporting Electrolyte | 1 M HCl or 0.1 M HNO₃. | Provides conductive medium and defines chemical environment for the electrochemical reaction. [32] |
| Arsenic Standard Solutions | As(III) (from NaAsO₂) and As(V) (from Na₂HAsO₄·7H₂O). | Used for calibration and quantification via the standard addition method. [5] [36] |
| Polishing Supplies | Alumina slurry (0.05 µm) and microcloth pad. | For mechanical pre-treatment to create a fresh, reproducible electrode surface. [32] |
| Electrochemical Activator | 0.5 M H₂SO₄ solution. | For electrochemical cleaning and activation of the gold surface via cyclic voltammetry. [32] |
| Inert Gas | High-purity Nitrogen or Argon. | For deoxygenation of the sample solution to prevent interference from dissolved oxygen. [5] |
The accurate determination of arsenic species in aquatic systems is critically dependent on the stability of the sample from the moment of collection until laboratory analysis. Arsenic toxicity is highly species-dependent, with inorganic arsenite (As(III)) being approximately 60 times more toxic than inorganic arsenate (As(V)) [37]. This significant difference in toxicity underscores the importance of preserving the original distribution of species. Without appropriate preservation, rapid oxidation of As(III) to As(V) or microbial-mediated transformations can occur, leading to inaccurate risk assessments and flawed scientific conclusions [37].
This application note provides detailed protocols for preserving inorganic arsenic speciation in water samples, with particular emphasis on procedures compatible with portable anodic stripping voltammetry (ASV) methods. The guidelines presented here are essential for ensuring data quality in field-based environmental monitoring and research.
The Challenge of Arsenic Species Stability
Stability of Aqueous Arsenic Species
The inherent instability of arsenic species between sampling and analysis presents a significant analytical challenge. Published data show considerable variation in the stability of As(III) in groundwater, ranging from as little as 2 days to up to 3 months under certain conditions [37]. This variability highlights the necessity of implementing effective preservation strategies for all water samples intended for speciation analysis.
The primary instability arises from the oxidation of As(III) to As(V), although reduction of As(V) to As(III) has also been reported [37]. These transformations are facilitated by several factors present in the sample matrix:
- Dissolved oxygen interacting with catalytic metal ions like iron [37]
- Microbial activity that metabolizes arsenic species [37]
- Changes in pH and temperature that accelerate redox reactions [37]
Implications for Analytical Techniques
The choice of preservation method must be compatible with the intended analytical technique. For example, hydrochloric acid (HCl) preservation is unsuitable when using inductively coupled plasma mass spectrometry (ICP-MS) due to the formation of 40Ar35Cl+ molecular interference on monoisotopic 75As+ [37]. Conversely, approaches effective for chromatographic techniques may not be optimal for voltammetric analysis.
Research Reagent Solutions for Speciation Preservation
Table 1: Key reagents for arsenic species preservation in water samples
| Reagent | Concentration | Function | Compatibility Notes |
|---|---|---|---|
| Potassium Sodium Tartrate | 2 mmol L⁻¹ | Complexes metal ions that catalyze As(III) oxidation | Compatible with ASV; effective in both model and natural waters [37] |
| Citric Acid | 2 mmol L⁻¹ | Acts as metal chelator and mild acidifying agent | Preserves As(III) for 7 days in model solutions [37] |
| Citric Acid + Acetic Acid | 2 mmol L⁻¹ + 5% | Enhanced chelation with acidification | Improves preservation in natural waters; compatible with ASV [37] |
| Sodium Oxalate | 2 mmol L⁻¹ | Strong metal complexation, particularly for iron | Effective in model solutions [37] |
| Sodium Citrate | 2 mmol L⁻¹ | Buffering and metal chelation | Similar efficacy to citric acid [37] |
| Disodium EDTA | 0.25 mmol L⁻¹ | Strong chelation of Fe, Mn, Al cations | Preferred for IC/HPLC-ICP-MS; prevents precipitation [37] |
| Acetic Acid (alone) | 5% | Mild acidification | Inadequate for speciation preservation [37] |
Recommended Protocols for Sample Collection and Preservation
Field Collection Procedure
Materials and Equipment
- Clean 1-2 L polyethylene or polypropylene bottles (acid-washed)
- Nitrile or polyethylene gloves
- Portable pH meter
- Syringe filters (0.45 μm pore size)
- Filtration apparatus
- Cooler with ice packs
- Aluminum foil or dark plastic bags
- Pre-measured preservative reagents
Step-by-Step Collection Protocol
- Personal Protective Measures: Wear clean gloves throughout the collection process to prevent sample contamination.
- Sample Bottle Preparation: Rinse sample bottles three times with the source water before collecting the final sample.
- Filtration: Filter water samples immediately after collection using 0.45 μm syringe filters to remove suspended particles and microorganisms.
- Preservative Addition: Immediately add the selected preservation reagent to achieve the recommended final concentration (typically 2 mmol L⁻¹ for complexing agents).
- Initial Storage: Place samples in a dark cooler on ice packs (approximately 4°C) immediately after preservation.
- Documentation: Record field parameters including pH, temperature, dissolved oxygen, and conductivity at the time of collection.
Preservation Methods Comparison
Table 2: Effectiveness of preservation methods for inorganic arsenic speciation
| Preservation Method | As(III) Stability (Model Solutions) | As(III) Stability (Natural Waters) | Time to Significant Oxidation |
|---|---|---|---|
| Unpreserved | < 3 days | < 2-3 days | 24-72 hours [37] |
| Potassium Sodium Tartrate | > 10 days | 6-12 days | Extended stability window [37] |
| Citric Acid | 7 days | 6-12 days | 7+ days [37] |
| Citric Acid + Acetic Acid | 7 days | 6-12 days | 7+ days [37] |
| Sodium Oxalate | > 10 days | Variable | Dependent on sample matrix [37] |
| Refrigeration (4°C) only | < 5 days | < 3 days | Limited effectiveness [37] |
| Acetic Acid only | < 3 days | < 3 days | Inadequate preservation [37] |
Optimal Storage Conditions
- Temperature: Maintain samples at 4°C throughout storage and transport [37]
- Light Exposure: Store samples in complete darkness by wrapping bottles in aluminum foil or using dark plastic bags [37]
- Holding Time: Analyze preserved samples within 6-12 days for reliable speciation results [37]
- Container Type: Use polyethylene or polypropylene containers to minimize adsorption
Analytical Verification with Portable Anodic Stripping Voltammetry
Principle of ASV for Arsenic Speciation
Portable differential pulse anodic stripping voltammetry (DPASV) offers a field-deployable solution for arsenic speciation analysis. The method utilizes a solid gold electrode system that allows direct determination of As(III) and total inorganic arsenic through strategic manipulation of electrochemical parameters [5]:
- As(III) is selectively determined at +0.1 V by ASV after deposition at -0.3 V [5]
- Total inorganic arsenic is determined after electrochemical reduction of As(V) to As(0) at -1.2 V [5]
- As(V) concentration is calculated by difference between total inorganic arsenic and As(III) [5]
This approach eliminates the need for chemical reduction of As(V), minimizing reagent consumption and simplifying field application [5]. The method provides a detection limit of 0.10 μg L⁻¹, sufficient for monitoring arsenic at the WHO provisional guideline value of 10 μg L⁻¹ for drinking water [5].
Method Verification Protocol
To verify preservation efficacy using portable ASV:
- Initial Measurement: Analyze As(III) concentration immediately upon return to field laboratory
- Periodic Re-analysis: Measure preserved samples at 24-hour intervals for the first 48 hours, then every 2-3 days
- Stability Criterion: Consider preservation effective if As(III) concentration variation remains within ±10% of initial measurement
- Quality Control: Include unpreserved duplicate samples as controls to monitor natural oxidation rates
Workflow Diagram: Sample Collection to Analysis
Sample Preservation Workflow for Arsenic Speciation Analysis
Effective preservation of arsenic species from field collection to laboratory analysis is essential for accurate risk assessment and environmental monitoring. The combination of metal-complexing agents such as tartrate or citric acid with proper cold storage provides effective stabilization of inorganic arsenic species for 6-12 days, sufficient for most field-to-laboratory workflows. These preservation methods are particularly well-suited for use with portable anodic stripping voltammetry, enabling reliable arsenic speciation analysis in aquatic systems with the significant advantages of field deployment capability, minimal reagent consumption, and excellent sensitivity meeting regulatory requirements for drinking water monitoring.
The development of a portable Anodic Stripping Voltammetry (ASV) method for arsenic speciation in aquatic systems represents a significant advancement in environmental monitoring [5]. The toxicity, mobility, and bioavailability of arsenic depend critically on its chemical speciation, with inorganic arsenite (As(III)) and arsenate (As(V)) representing the most toxic and commonly encountered forms in natural waters [1]. Unlike laboratory-based techniques that require sophisticated instrumentation and extensive sample preparation, ASV offers the potential for sensitive, cost-effective, and rapid on-site analysis [20] [38]. This application note provides a comprehensive protocol for optimizing key measurement parameters—electrolyte composition, deposition time, and stripping waveforms—to achieve reliable arsenic speciation with sub-ppb detection limits suitable for compliance with the World Health Organization (WHO) guideline of 10 μg L⁻¹ for arsenic in drinking water [1].
Optimized Measurement Parameters for Arsenic Speciation
The optimization of operational parameters is fundamental to the performance of ASV for arsenic detection. The following parameters have been systematically investigated and validated for the determination and speciation of inorganic arsenic in water samples.
Table 1: Optimized ASV Parameters for Arsenic Speciation
| Parameter Category | Specific Parameter | Recommended Setting | Application Context |
|---|---|---|---|
| Electrode System | Working Electrode | Solid Gold Electrode (Rotating) | General As detection [5] |
| In-situ Gold Film on Disposable Carbon | Portable As(III) detection [38] | ||
| Electrode Modification | In-situ electroplated gold nanoparticles | Enhanced sensitivity and disposable use [38] | |
| Deposition Step | Deposition Potential (As(III)) | -0.3 V to -0.5 V | Selective detection of As(III) [5] [23] |
| Deposition Potential (Total As) | -1.2 V | Reduction of As(V) to As(0) for total As [5] [23] | |
| Deposition Time | 30 s to several minutes | Adjustable based on required LOD [5] [39] | |
| Solution Conditions | Supporting Electrolyte | HCl (e.g., 5 mol/L) | Standard medium for total As [23] |
| pH for As(III) determination | Neutral (pH ~8) or Mild Acid (pH 1) | Direct As(III) analysis in natural waters [39] | |
| Stripping Step | Stripping Waveform | Differential Pulse (DP) ASV | Standard method for sensitive detection [5] |
| Square-Wave (SW) ASV | Fast and sensitive detection [31] | ||
| Stripping Chronopotentiometry (SC) | Lower detection limit at neutral pH [39] | ||
| Analytical Performance | Limit of Detection (LOD) | 0.10 μg L⁻¹ for As(tot) [5] | Achieves sub-ppb detection |
| 0.2 μg L⁻¹ for As(III) [38] | Suitable for portable monitoring | ||
| Repeatability (RSD) | 4.7% at 5.0 μg L⁻¹ As(III) [38] | Demonstrates good precision |
Detailed Experimental Protocols
Protocol 1: Speciation of Inorganic Arsenic Using a Solid Gold Electrode
This protocol outlines the procedure for determining As(III) and total inorganic arsenic in water samples using a rotating solid gold electrode, based on the method characterized by [5].
3.1.1 Materials and Reagents
- Supporting Electrolyte: Hydrochloric acid (HCl), 5 mol/L [23].
- Standard Solutions: Certified As(III) and As(V) standard solutions for calibration.
- Purified Water: Deionized or doubly-distilled water (resistivity ≥ 18 MΩ·cm).
- Electrode Polishing: Alumina slurry (e.g., 0.05 μm) and polishing cloth.
3.1.2 Instrumentation Setup
- Potentiostat: Equipped with differential pulse (DP) capability.
- Electrochemical Cell: Three-electrode system comprising:
- Working Electrode: Rotating solid gold electrode (SGE).
- Counter Electrode: Platinum wire.
- Reference Electrode: Ag/AgCl (3 M KCl).
- Sample Containers: Acid-washed polyethylene or polypropylene bottles.
3.1.3 Step-by-Step Procedure
- Electrode Pretreatment: Polish the gold electrode surface with alumina slurry on a microcloth to a mirror finish. Rinse thoroughly with purified water. Electrochemically clean the electrode in the supporting electrolyte by applying cyclic voltammetry between -0.3 V and +1.5 V until a stable voltammogram is obtained.
- Sample Collection and Preservation: Collect water samples in pre-cleaned containers. For speciation analysis, analyze samples immediately without acidification or filtration to preserve the original As(III)/As(V) ratio. If immediate analysis is not possible, store samples at 4°C in the dark.
- Calibration Curve for As(III):
- Prepare a series of standard solutions containing known concentrations of As(III) (e.g., 0, 1, 2, 5, 10 μg L⁻¹) in a background of 5 mol/L HCl.
- Transfer 10 mL of standard to the electrochemical cell.
- Set the deposition potential to -0.3 V and deposition time to 120 s with electrode rotation.
- Initiate a DPASV scan from -0.3 V to +0.5 V. Record the anodic stripping peak for As(0) → As(III) at approximately +0.1 V [5].
- Plot the peak current versus As(III) concentration to construct the calibration curve.
- Determination of As(III) in Sample:
- Mix the water sample with an equal volume of concentrated HCl to achieve a final concentration of approximately 5 mol/L.
- For direct As(III) determination, use a deposition potential of -0.3 V to -0.5 V [5] [23]. Under these conditions, As(V) is not reduced and does not contribute to the signal.
- Run the DPASV sequence and quantify As(III) from the calibration curve.
- Determination of Total Inorganic Arsenic:
- Using the same acidified sample, set the deposition potential to -1.2 V.
- At this highly negative potential, As(V) is electrochemically reduced to As(0) by nascent hydrogen and deposited on the electrode alongside As(III) [5] [23].
- Run the DPASV sequence. The resulting peak corresponds to the total inorganic arsenic content (As(III) + As(V)).
- Calculation of As(V) Concentration:
- Determine the As(V) concentration by subtracting the As(III) concentration from the total inorganic arsenic concentration.
Protocol 2: Rapid On-Site Monitoring of As(III) Using Disposable Sensors
This protocol is designed for rapid, on-site screening of As(III) using a portable potentiostat and disposable, injection-moulded electrodes with in-situ gold plating [38].
3.2.1 Materials and Reagents
- Disposable Sensor: Three-electrode mini-cell with carbon-based working electrode [38].
- Gold Plating Solution: 1000 mg L⁻¹ Au(III) atomic absorption standard solution.
- Interference Suppressor: Potassium ferrocyanide solution.
- Portable Potentiostat: Compact instrument with SWASV or DPASV capability.
3.2.2 Step-by-Step Procedure
- Sensor and Solution Preparation:
- Obtain the disposable sensor cell and connect it to the portable potentiostat.
- To a 10 mL water sample, add 50 μL of the 1000 mg L⁻¹ Au(III) solution (final Au(III) ~5 mg L⁻¹) and an appropriate amount of potassium ferrocyanide to mitigate copper interference [38].
- In-Situ Gold Film Formation and Analysis:
- Place the sample vial containing the spiked solution on the potentiostat.
- Immerse the disposable sensor electrodes into the solution.
- Apply a deposition potential of -0.5 V for 60-120 s with stirring. This co-deposits gold nanoparticles and arsenic onto the working electrode surface, forming an As-Au alloy.
- After the deposition period, stop stirring and wait for 10-15 s for the solution to quiesce.
- Initiate a square-wave anodic stripping voltammetry (SWASV) scan.
- The stripping peak for As(III) will appear at a characteristic potential. Measure the peak height.
- Quantification via Standard Addition:
- For on-site analysis with complex matrices, the standard addition method is preferred.
- After measuring the initial sample, spike the same solution with a known volume of As(III) standard.
- Repeat the deposition and stripping measurement.
- Calculate the original As(III) concentration based on the increase in peak current.
Signaling Pathways and Workflows
The following diagram illustrates the core analytical workflow for arsenic speciation using ASV, highlighting the critical decision points for differentiating between As(III) and total arsenic.
The Scientist's Toolkit: Research Reagent Solutions
The successful implementation of ASV for arsenic speciation relies on a set of key materials and reagents. The following table details these essential components and their specific functions within the analytical procedure.
Table 2: Essential Research Reagents and Materials for ASV-based Arsenic Speciation
| Item | Specification / Example | Primary Function in Analysis |
|---|---|---|
| Solid Gold Electrode | Rotating disk electrode (RDE) | Working electrode for As deposition/stripping; forms alloy with As [5]. |
| Gold Plating Solution | Au(III) standard solution (e.g., 1000 mg L⁻¹) | Source for in-situ formation of gold nanoparticle films on disposable sensors [38]. |
| Supporting Electrolyte | Hydrochloric Acid (HCl, 5 mol/L) | Provides conductive medium and optimal acidic conditions for total As analysis [23]. |
| Reference Electrode | Ag/AgCl (3 M KCl) | Provides a stable and reproducible reference potential for voltammetric measurements. |
| As Standard Solutions | Certified As(III) and As(V) ICP or AAS standards | Used for instrument calibration and method validation [38]. |
| Interference Suppressor | Potassium Ferrocyanide | Mitigates signal suppression caused by copper interference [38]. |
| Electrode Polishing Kit | Alumina slurry (0.05 μm) and microcloth | Maintains a clean and electroactive electrode surface for reproducible results [20]. |
The accurate determination of arsenic in aquatic systems represents a significant global health challenge, with an estimated 250 million people worldwide affected by arsenic toxicity [40]. Inorganic arsenic exists primarily as two species in water: arsenite (As(III)) and arsenate (As(V)). The toxicity of As(III) is approximately 60 times greater than that of As(V), making speciation analysis—not just total arsenic quantification—essential for proper risk assessment [37]. Traditional laboratory techniques such as high-performance liquid chromatography coupled with inductively coupled plasma mass spectrometry (HPLC-ICP-MS), while highly sensitive, are expensive, time-consuming, and limited to specialist laboratories [17]. Furthermore, the instability of arsenic species between sampling and analysis, particularly the oxidation of As(III) on an hours-to-days timescale, necessitates complex preservation protocols or, preferably, on-site analysis [17] [37].
Anodic Stripping Voltammetry (ASV) has emerged as a powerful alternative, offering a rapid, sensitive, and cost-effective method suitable for field deployment. This Application Note details standardized protocols and presents case studies demonstrating the practical application of portable ASV for arsenic speciation across diverse aqueous matrices, including groundwater, surface water, and pore water. The ability to perform reliable, on-site speciation is crucial for monitoring and remediation efforts, particularly in remote or resource-limited settings where arsenic contamination is prevalent.
The Scientist's Toolkit: Essential Reagents & Materials
Successful field speciation requires careful selection of reagents and materials. The following table outlines key solutions and their specific functions in the analytical protocols.
Table 1: Key Research Reagent Solutions for Arsenic Speciation via ASV
| Reagent/Material | Function/Explanation |
|---|---|
| Solid Gold Electrode (SGE) / Gold Microwire | Working electrode for ASV; provides a highly sensitive and reproducible surface for arsenic deposition and stripping [5] [22]. |
| Acetate Buffer (pH 4.7) | Creates optimal mildly acidic supporting electrolyte for arsenic detection, enhancing sensitivity and signal stability [22]. |
| Potassium Permanganate (KMnO₄) | Serves as an oxidizing agent; ensures all inorganic arsenic is present as As(V) for total arsenic determination and improves sensitivity [22]. |
| Complexing Agents (e.g., Citric Acid, Tartrate) | Preserves arsenic species ratio between sampling and analysis by complexing metal ions like Fe(III) that catalyze As(III) oxidation [37]. |
| Chloride Salts (e.g., KCl) | Provides chloride ions (Cl⁻) that enhance the voltammetric response of arsenic on gold electrodes, improving peak shape and sensitivity [22]. |
| As(V)-Selective Chemisorbent (ImpAs) | Used in a complementary speciation method; selectively removes As(V) from solution in syringe-based systems, allowing for indirect As(III) determination [17]. |
Analytical Protocols for Arsenic Speciation
Protocol A: Direct Speciation of As(III) and As(V) via DP-ASV
This protocol, adapted from Inaudi et al. (2025) and subsequent studies, is designed for the direct differentiation and quantification of inorganic arsenic species in freshwater samples using Differential Pulse Anodic Stripping Voltammetry (DP-ASV) [5] [41] [37].
Sample Collection & Preservation
- Collect water samples in pre-cleaned polyethylene or glass bottles.
- Immediate Preservation: To maintain species integrity, immediately add a complexing agent such as citric acid or potassium sodium tartrate to a final concentration of 2 mmol L⁻¹. This stabilizes the As(III)/As(V) ratio by complexing metal cations that catalyze oxidation [37].
- Store samples at 4°C in the dark and analyze as soon as possible, ideally within 7 days for preserved samples.
Instrumental Setup
- Potentiostat: Portable potentiostat capable of DP-ASV.
- Electrode System: Rotating Solid Gold Electrode (SGE) as the working electrode, Pt counter electrode, and Ag/AgCl reference electrode. Alternatively, a maintenance-free gold microwire electrode (e.g., scTRACE Gold) can be used [37] [22].
- Parameters: Deposition potential (Edep) = -0.3 V; Deposition time (tdep) = 30-60 s (adjust based on concentration); Equilibrium time = 10 s; Pulse amplitude = 50 mV; Step height = 2 mV [5].
Speciation Procedure
- Step 1: Determine As(III). Analyze the preserved sample directly. As(III) is electroactive and yields a stripping peak at approximately +0.1 V [5].
- Step 2: Determine Total Inorganic Arsenic. In a separate aliquot, electrochemically reduce As(V) to As(0) at a potential of -1.2 V using nascent hydrogen. Following this pre-reduction step, the total inorganic arsenic (As(III) + As(V)) is quantified via the standard ASV procedure [5] [41].
- Step 3: Calculate As(V). The As(V) concentration is determined by difference:
[As(V)] = [As(total)] - [As(III)].
The workflow for this direct speciation method is summarized in the diagram below.
Protocol B: Total Inorganic Arsenic Determination at Near-Neutral pH
This innovative protocol, validated by Bullen et al. (2023), simplifies total inorganic arsenic measurement by chemically converting all arsenic to As(V) and detecting it under mild acidic conditions, avoiding strong acids and complex pre-reduction [22].
Sample Preparation
- To the water sample, add an acetate buffer (pH 4.7) containing 0.25 M chloride.
- Spike with a low concentration of potassium permanganate (KMnO₄) to a final concentration of 10 µM. This oxidizes any As(III) to As(V) and provides a source of manganese to facilitate the sensitive detection of As(V) [22].
Instrumental Setup
- Potentiostat: Portable potentiostat.
- Electrode: Gold microwire working electrode (e.g., 25 µm diameter Gold Wirebond).
- Parameters: Deposition potential = -0.7 V (vs. Ag/AgCl); Deposition time = 10 s; Linear sweep stripping [22].
Analysis Procedure
- Analyze the prepared sample directly.
- The method provides a single peak for total inorganic arsenic with a reported limit of detection of 0.28 µg L⁻¹ [22].
- Quantify the concentration using the standard addition method to account for matrix effects.
Case Studies & Data Analysis
Case Study 1: Groundwater Speciation in South Asia
Objective: To monitor inorganic arsenic species in reducing groundwaters of West Bengal, India, and Bihar, known for high levels of arsenite [17] [22].
Methods: Protocol A (DP-ASV) was applied using a portable potentiostat and gold electrodes. Samples were preserved with tartrate. Results were validated against ICP-MS.
Results: The ASV method demonstrated excellent correlation with ICP-MS (slope = +1.029, R² = 0.99), confirming its reliability for complex groundwater matrices. The method successfully quantified species in samples where As(III) was the dominant form [22].
Case Study 2: Oxidizing Groundwaters of Central Mexico
Objective: To determine total inorganic arsenic in oxidising, arsenate-rich groundwaters from the Guanajuato region [22].
Methods: Protocol B (Near-neutral pH with permanganate) was employed for rapid total arsenic screening.
Results: The method achieved a limit of detection (LOD) of 0.28 µg L⁻¹ with a deposition time of only 10 seconds, proving highly effective for the rapid analysis of arsenate-dominated waters without the need for chemical reduction [22].
Table 2: Performance Metrics of Portable ASV Methods for Arsenic Analysis
| Method | Application | Linear Range | Limit of Detection (LOD) | Key Advantage |
|---|---|---|---|---|
| Protocol A: Direct DP-ASV Speciation [5] | Speciation of As(III) & As(V) in natural waters | Up to 20 µg L⁻¹ | 0.10 µg L⁻¹ for As(tot) | Direct species differentiation; no need for oxidation/reduction chemicals. |
| Protocol B: Near-Neutral pH (with MnO₄⁻) [22] | Total inorganic As in groundwater (various pH) | Up to 20 µg L⁻¹ | 0.28 µg L⁻¹ | Simplified workflow; fast (10 min/sample); no strong acids required. |
| Complementary: ImpAs Chemisorbent [17] | Speciation via selective As(V) removal | Not specified | Comparable to ASV | High selectivity against phosphate; simple syringe-based operation. |
The relationship between the two primary ASV protocols and their application to different field scenarios is illustrated below.
Portable Anodic Stripping Voltammetry has matured into a robust and reliable platform for the on-site speciation and quantification of inorganic arsenic in aquatic systems. The protocols detailed in this Application Note provide researchers with clear, actionable methodologies for tackling arsenic contamination across a diverse range of environmental conditions, from the reducing groundwaters of South Asia to the oxidizing aquifers of North America. The key advantages of sensitivity, portability, cost-effectiveness, and the ability to deliver rapid, on-site results make ASV an indispensable tool for global arsenic monitoring and remediation efforts.
Overcoming Field Challenges: Troubleshooting Interferences and Optimizing ASV Performance
Identifying and Mitigating Common Interferences (e.g., Copper, Sulfide, Organic Matter)
The application of portable anodic stripping voltammetry (ASV) for arsenic speciation in aquatic systems represents a significant advancement in field-deployable analytical techniques. However, the accuracy and reliability of these methods are frequently compromised by common interferences, including organic matter, competing metal ions such as copper and zinc, and sulfide species. Effectively identifying and mitigating these interferences is paramount for obtaining accurate speciation data, which is critical for assessing arsenic toxicity, mobility, and treatment efficacy in natural waters. This document provides detailed application notes and protocols to support researchers in overcoming these analytical challenges within the broader context of developing robust field methods for arsenic speciation.
Common Interferences and Mitigation Strategies
The following table summarizes the primary interferences encountered in ASV for arsenic analysis and their respective mitigation strategies.
Table 1: Common Interferences and Mitigation Strategies in ASV for Arsenic Speciation
| Interference Type | Specific Interferents | Impact on ASV Analysis | Recommended Mitigation Strategies |
|---|---|---|---|
| Organic Matter | Surface-Active Substances (SAS), Humic/Fulvic Acids, Dissolved Organic Carbon (DOC) [42] | Adsorption onto the working electrode, causing signal suppression/depression, deformed peaks, and inaccurate speciation parameters [42]. | Addition of 1 mg L⁻¹ Triton-X-100 [42]; Use of a "desorption step" (e.g., -1.5 V for 1-3 s) [42]. |
| Competing Metal Ions | Copper (Cu), Zinc (Zn), Nickel (Ni) [43] | Formation of intermetallic compounds (e.g., Cu-Zn), altered stripping potentials, overlapping peaks, and suppressed signals [43]. | Addition of 500 mg L⁻¹ Gallium (Ga) [43]; Calibration by standard additions [43]. |
| Sulfide Species | Hydrogen Sulfide (H₂S), various metal sulfides [43] | Can poison the electrode surface, form insoluble complexes with target analytes, and lead to inaccurate measurements of metal content in leachates [43]. | Application of sulphide-selective leaches (e.g., KClO₃-HCl) for sample pre-treatment [43]; Note: Specific mitigation for As-S interference in ASV requires further method development. |
Detailed Experimental Protocols
Protocol for Mitigating Organic Matter Interference with Triton-X-100
This protocol is adapted from methods developed for copper speciation in DOM-rich waters and is applicable for arsenic analysis where organic matter causes signal interference [42].
1. Reagents and Solutions:
- Triton-X-100 stock solution (e.g., 100 mg L⁻¹ in high-purity water)
- Supporting electrolyte (appropriate for your ASV method and arsenic speciation)
- Standard solutions of As(III) and As(V)
- High-purity water (e.g., 18.2 MΩ·cm)
2. Equipment:
- Portable potentiostat with ASV capability
- Solid Gold Electrode (SGE) or other appropriate working electrode [5] [41]
- Reference electrode (e.g., Ag/AgCl)
- Counter electrode (e.g., Pt)
- Volumetric flasks and pipettes
3. Procedure:
- Step 1: Prepare sample and standard solutions as per your standard ASV method for arsenic.
- Step 2: To the sample solution, add a known volume of Triton-X-100 stock solution to achieve a final concentration of 1 mg L⁻¹ [42]. Mix thoroughly.
- Step 3: Execute the ASV measurement. The method for arsenic speciation typically involves:
- A deposition step at -0.3 V for As(III) determination [5] [41].
- A stripping step where As(III) is selectively determined at +0.1 V [5] [41].
- For total inorganic arsenic, an electrochemical reduction step at -1.2 V is applied to reduce As(V) to As(0), followed by stripping. As(V) concentration is calculated by difference [5] [41].
- Step 4: Compare the peak shape and intensity with and without Triton-X-100 addition. A well-defined, enhanced peak indicates successful mitigation of SAS interference.
4. Validation:
- Validate the method by comparing results with a reference technique, such as Hydride Generation-ICP-OES (HG-ICP-OES), using real water samples [5] [41].
Protocol for Mitigating Copper Interference with Gallium
This protocol, based on classic ASV practices, is crucial when analyzing arsenic in the presence of high copper concentrations [43].
1. Reagents and Solutions:
- Gallium standard solution (e.g., GaCl₃), concentration sufficient to add 500 mg L⁻¹ to the analysis solution [43].
- Supporting electrolyte.
- Standard additions of target analytes (Zn, Cd, Pb, Cu, As).
2. Equipment:
- Portable potentiostat with ASV capability.
- Standard ASV electrode system (e.g., mercury film electrode).
3. Procedure:
- Step 1: Add the supporting electrolyte and sample to the electrochemical cell.
- Step 2: Add Gallium standard solution to achieve a final concentration of 500 mg L⁻¹ Ga in the analysis solution [43].
- Step 3: Perform ASV analysis using a deposition time of 60 seconds, which can achieve detection limits in the low μg L⁻¹ range for metals like Cd and Pb [43].
- Step 4: Employ the standard addition method for calibration to account for matrix effects and ensure accuracy [43].
4. Notes:
- The addition of Ga allows the method to tolerate Cu concentrations up to a 20-fold excess and Ni concentrations up to a 30-fold excess over Zn [43].
- This approach is well-suited for the analysis of complex matrices like sulphide-selective leaches (KClO₃-HCl) [43].
Workflow Visualization for Interference Mitigation
The following diagram illustrates the logical decision-making process and experimental workflow for identifying and mitigating common interferences in portable ASV analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents and Materials for Interference Mitigation in Portable ASV
| Reagent/Material | Function/Purpose | Example Application/Note |
|---|---|---|
| Triton-X-100 | Non-ionic surfactant that competitively inhibits adsorption of Surface-Active Substances (SAS) on the working electrode [42]. | Added to a final conc. of 1 mg L⁻¹ to eliminate SAS interference from dissolved organic matter, improving peak shape [42]. |
| Gallium (Ga³⁺) Solution | Suppresses formation of intermetallic compounds (e.g., with Cu, Zn) that interfere with stripping signals [43]. | Added to a final conc. of 500 mg L⁻¹ to tolerate a 20-fold excess of Cu and 30-fold excess of Ni [43]. |
| Solid Gold Electrode (SGE) | Working electrode for the sensitive and selective detection of arsenic species [5] [41]. | Used in a rotating configuration for the determination of As(III) and total inorganic arsenic [5] [41]. |
| Sulphide-Selective Leach (KClO₃-HCl) | Chemical leach for sample pre-treatment to isolate the sulphide phase of metals, mitigating some sulfide-related interferences [43]. | Used in bedrock geochemistry for analysis of sulphide-associated metals [43]. |
| Portable Potentiostat | Core instrument for performing ASV measurements in the field [5]. | Enables on-site speciation with techniques like Differential Pulse ASV (DPASV) [5]. |
The move from laboratory-based instruments to portable ASV methods for arsenic speciation demands proactive strategies to manage analytical interferences. The protocols outlined here for mitigating organic matter, competing metal ions, and sulfides provide a concrete foundation for researchers. By integrating these reagent-based solutions and procedural adjustments—such as the addition of Triton-X-100 or gallium, and the use of specialized electrodes and leaches—scientists can significantly enhance the reliability of data generated in the field. This robust approach to interference management is fundamental to advancing research on arsenic dynamics in aquatic environments and for supporting effective water quality monitoring and remediation efforts.
The precise determination of arsenic speciation in complex aquatic environments, particularly high-ionic-strength and iron-rich waters, represents a critical analytical challenge in environmental chemistry. The toxicity, mobility, and environmental fate of arsenic are fundamentally governed by its chemical form, with inorganic arsenite (As(III)) exhibiting significantly greater toxicity and mobility compared to arsenate (As(V)) [28] [17]. In iron-rich waters, arsenic biogeochemistry becomes particularly complex due to strong interactions with iron oxyhydroxides such as goethite (α-FeOOH), which can sequester arsenic through adsorption and co-precipitation processes [44]. These interactions, combined with high background ionic strength, create substantial matrix interferences that compromise analytical accuracy and sensitivity. Consequently, robust analytical strategies must address both the selective determination of individual arsenic species and the mitigation of complex matrix effects to enable reliable field and laboratory analysis.
The development of portable analytical methods has gained prominence due to the instability of arsenic species during sample storage and the urgent need for on-site decision-making in contaminated areas [17]. This application note, framed within broader thesis research on portable anodic stripping voltammetry (ASV), details standardized protocols for arsenic speciation across diverse water matrices, with particular emphasis on addressing challenges posed by high-ionic-strength and iron-rich conditions.
Analytical Techniques for Arsenic Speciation
Multiple analytical platforms are available for arsenic speciation, each offering distinct advantages and limitations regarding sensitivity, portability, robustness to matrix effects, and operational complexity. The choice of technique depends on required detection limits, sample throughput, available infrastructure, and specific matrix characteristics.
Table 1: Comparison of Analytical Techniques for Arsenic Speciation
| Technique | Detection Limits | Key Advantages | Key Limitations | Suitability for Complex Matrices |
|---|---|---|---|---|
| LC-ICP-MS | < 1.0 μg L⁻¹ for most species [28] | High sensitivity; simultaneous multi-species detection; ability to identify unknown species with LC-MS [28] | High cost; requires specialized laboratory and operator; limited portability | Excellent with reaction/collision cell technology to remove interferences [28] |
| Anodic Stripping Voltammetry (ASV) | 0.10 μg L⁻¹ for As(tot) [5] | Portability; cost-effectiveness; minimal reagent consumption; no significant phosphate interference [5] [17] | Electrode fouling in complex matrices; requires optimization for different water types | Good with optimized deposition times and cleaning procedures [17] |
| Selective Chemisorption (ImpAs) | Not explicitly stated (validation vs ASV) [17] | High selectivity for As(V); portability; minimal equipment; rapid analysis; resistant to anion interference [17] | Limited capacity; performance degradation with >5 mg L⁻¹ dissolved iron [17] | Good for low-iron waters; limited in high-iron environments [17] |
| Colorimetric (Molybdenum Blue) | >7 μg L⁻¹ [28] | Low cost; simplicity; field adaptability | Severe phosphate interference; requires multiple pretreatment steps; lower sensitivity [28] [17] | Poor due to ubiquitous phosphate interference and complex pretreatments [17] |
| GC-MS | 6-14 μg L⁻¹ (after derivatization) [28] | Molecular speciation without certified reference materials; structural identification [28] | Requires derivatization; higher LOD than LC-ICP-MS; complex sample preparation [28] | Moderate; derivatization may be affected by matrix components |
Technique Selection Guidance
For field applications in high-ionic-strength waters, portable ASV and selective chemisorption offer practical solutions with minimal infrastructure requirements [5] [17]. For comprehensive laboratory analysis, particularly when unknown arsenic species may be present, LC-ICP-MS remains the gold standard, especially when coupled with collision/reaction cell technology to mitigate polyatomic interferences from complex matrices [28]. In iron-rich waters, sample pretreatment to manage iron concentration is recommended for all techniques, particularly for methods like ImpAs chemisorption where dissolved iron >5 mg L⁻¹ compromises performance [17].
Detailed Experimental Protocols
Portable Anodic Stripping Voltammetry for Arsenic Speciation
Principle and Applications
Differential pulse anodic stripping voltammetry (DPASV) using solid gold electrodes (SGE) enables highly sensitive determination and speciation of inorganic arsenic in aquatic environments [5]. This method leverages the selective electrochemical behavior of As(III) and As(V) at a gold electrode surface, with As(III) being directly determined and As(V) quantified indirectly after electrochemical reduction [5]. The technique is particularly valuable for on-site analysis in complex matrices due to its portability, low cost, and absence of significant phosphate interference that plagues colorimetric methods [17].
Required Equipment and Reagents
- Portable Potentiostat with capacity for DPASV measurements
- Solid Gold Rotating Electrode (SGE) or gold microelectrodes
- Reference Electrode (Ag/AgCl typically)
- Auxiliary Electrode (platinum typically)
- Supporting Electrolyte: High-purity HCl or other suitable electrolyte
- Standard Solutions: As(III) (from As₂O₃) and As(V) (from Na₂HAsO₄·7H₂O) stock solutions (1000 mg L⁻¹) [17]
- Purified Gases: Nitrogen or argon for deaeration
- pH meter and appropriate buffers
Step-by-Step Protocol
Sample Collection and Preservation: Collect water samples using trace metal clean techniques. For As(III) preservation, acidify with HCl to pH <2 and store in opaque containers at 4°C to prevent photo-oxidation [17]. Analyze as rapidly as possible to minimize species transformation.
Electrode Preparation: Polish the gold electrode with 0.05 μm alumina slurry on a microcloth to refresh the surface. Rinse thoroughly with deionized water between samples and after polishing.
Instrument Parameters Setup:
- Deposition Potential: -0.3 V (for As(III) direct determination) [5]
- Deposition Time: 60-300 s (optimize based on expected concentration)
- Equilibrium Time: 10-15 s
- DPASV Parameters: Pulse amplitude 50 mV, pulse width 50 ms, step height 2 mV
As(III) Determination:
- Transfer 10 mL sample to voltammetric cell
- Deaerate with nitrogen/argon for 8-10 minutes
- Apply deposition potential at -0.3 V while rotating electrode
- Record DPASV scan from -0.3 V to +0.4 V
- Measure As(III) oxidation peak at approximately +0.1 V [5]
Total Inorganic Arsenic Determination:
- To same sample, apply electrochemical reduction at -1.2 V for 120-300 s using nascent hydrogen to reduce As(V) to As(0) [5]
- Follow with DPASV scan as above
- Quantify total inorganic arsenic from combined peak area
As(V) Calculation:
- Determine As(V) concentration by difference: As(V) = As(total) - As(III)
Calibration and Quality Control:
- Perform standard additions of both As(III) and As(V) to account for matrix effects
- Include certified reference materials where available
- Monitor electrode performance with quality control standards
Data Interpretation and Analysis
The analytical signal for As(III) appears as a well-defined peak at approximately +0.1 V under optimized conditions [5]. In complex matrices, standard addition is strongly recommended over direct calibration to compensate for matrix effects. The method achieves a limit of detection (LOD) of 0.10 μg L⁻¹ for total inorganic arsenic, making it suitable for regulatory compliance monitoring against the WHO guideline of 10 μg L⁻¹ [5] [28].
ImpAs Chemisorbent Method for Field Speciation
Principle and Applications
The ImpAs chemisorbent method utilizes a metal-organic complex containing two Zn(II) centers immobilized on polymeric beads that selectively chelate As(V) oxyanions while allowing As(III) to pass through unaffected [17]. This enables rapid field speciation through simple syringe-based filtration, with subsequent laboratory analysis of As(III) in the effluent and total arsenic in the untreated sample. The method is particularly valuable for high-ionic-strength waters where anion competition can challenge traditional ion-exchange approaches, as the ImpAs material shows high selectivity for As(V) over common interfering anions like sulfate and phosphate [17].
Required Equipment and Reagents
- ImpAs Chemisorbent Cartridges (commercially available or prepared as described in patent US 2017/0113949 A1) [17]
- Handheld Syringes (5-50 mL capacity)
- Sample Collection Vials (trace metal clean)
- Filtration Apparatus (0.45 μm membrane filters)
- Acid for Preservation (high-purity HCl)
- Reference Analytical Method (ICP-MS, AAS, or ASV for quantification)
Step-by-Step Protocol
Sample Collection and Pretreatment:
- Collect water samples using appropriate techniques for redox-sensitive species
- Filter through 0.45 μm membrane if particulate matter is present
- Split sample into two aliquots: one for total arsenic, one for speciation
ImpAs Treatment Procedure:
- Pre-condition ImpAs cartridge with 5 mL of deionized water at pH 6-8
- Load exact volume (10-50 mL) of sample into syringe attached to ImpAs cartridge
- Slowly pass sample through cartridge at 1-2 mL/min flow rate
- Collect effluent for As(III) analysis
Arsenic Quantification:
- Analyze untreated sample for total inorganic arsenic concentration
- Analyze ImpAs effluent for As(III) concentration
- Calculate As(V) by difference: As(V) = As(total) - As(III)
Method Validation:
- Validate cartridge performance with spiked samples of known composition
- Monitor cartridge capacity with high As(V) loads
- For iron-rich waters (>5 mg L⁻¹ Fe), evaluate potential iron interference [17]
Data Interpretation and Considerations
The ImpAs method shows optimal performance in the pH range of 6-8, making it suitable for most natural waters [17]. In validation studies comparing ImpAs with ASV for groundwater analysis, the method successfully speciated arsenic in both oxic UK waters and anoxic Indian groundwaters with <5 mg L⁻¹ dissolved iron [17]. The chemisorbent approach effectively determined rate constants for As(III) photooxidation that agreed with ASV measurements, demonstrating its utility for kinetic studies [17].
Addressing Matrix Challenges
High-Ionic-Strength Waters
High ionic strength matrices present challenges primarily through competitive adsorption and altered electrochemical responses. For voltammetric methods, increased supporting electrolyte concentration can actually enhance sensitivity, but competitive anion adsorption may require longer deposition times [17]. For separation-based methods like ImpAs chemisorption, high ionic strength typically has minimal effect due to the specific Zn(II)-As(V) coordination chemistry, which shows greater selectivity than traditional anion exchange resins [17].
Mitigation Strategies:
- Use standard addition calibration to compensate for matrix effects
- Increase deposition time in ASV for low-level detection
- Optimize pH to maximize As(V) selectivity in chemisorption methods
- Employ background subtraction in electrochemical measurements
Iron-Rich Waters
Iron-rich matrices present particular challenges for arsenic speciation due to iron-arsenic co-precipitation, redox interactions, and analytical interferences. Dissolved iron can exceed arsenic concentrations by several orders of magnitude in contaminated groundwaters, potentially leading to (1) oxidation state interconversion during sampling and analysis, (2) formation of iron-arsenic complexes that resist speciation, and (3) interference with selective separation methods [17].
Table 2: Addressing Iron-Rich Matrix Challenges
| Analytical Technique | Iron Interference Mechanisms | Recommended Mitigation Strategies |
|---|---|---|
| ASV | Competitive adsorption at electrode surface; formation of iron-arsenic complexes | Sample acidification; standard addition calibration; optimization of deposition potential [17] |
| ImpAs Chemisorption | Performance degradation with >5 mg L⁻¹ dissolved iron; possible clogging with iron precipitates | Sample filtration; pH adjustment; dilution for high-iron samples [17] |
| LC-ICP-MS | Column clogging; polyatomic interferences (⁴⁰Ar³⁵Cl⁺) | Collision/reaction cell technology; sample dilution; efficient sample introduction systems [28] |
| Colorimetric Methods | Phosphate-independent iron interference; colored complexes | Additional sample clean-up steps; matrix-matched standards [28] |
General Mitigation Strategies for Iron-Rich Waters:
- Immediate Filtration (0.45 μm) to remove particulate iron and associated arsenic
- Sample Acidification to pH <2 to dissolve iron colloids and prevent oxidation of As(III)
- Refrigerated Storage in opaque containers to minimize redox transformations
- Analysis Within 24-48 Hours of collection to preserve species integrity
- Method-Specific Optimization based on iron concentration and speciation
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Arsenic Speciation
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| Solid Gold Electrode (SGE) | Working electrode for ASV measurements; enables As(III) deposition and stripping | Rotating design preferred; requires regular polishing for optimal performance [5] |
| ImpAs Chemisorbent | Selective As(V) extraction from complex matrices; enables field speciation | Zn(II)-based receptor on polymeric beads; optimal pH 6-8; capacity affected by high iron [17] |
| Gold Microelectrodes | Alternative to SGE for portable systems; reduced susceptibility to fouling | Useful for field applications; different geometry than conventional SGE [17] |
| Hydrazine Solutions | Reducing agent for As(V) to As(III) in colorimetric methods | Enables total inorganic arsenic determination; used in 24 mM concentration [17] |
| Anion Exchange Columns | Chromatographic separation of arsenic species for LC-ICP-MS | Typically use (NH₄)₂HPO₄ or (NH₄)₂CO₃ mobile phase; sometimes with methanol addition [28] |
| Certified Reference Materials | Method validation and quality control | Essential for verifying accuracy in complex matrices; available for various water types |
Reliable arsenic speciation in high-ionic-strength and iron-rich waters requires careful method selection and optimization to address matrix-specific challenges. Portable ASV methods provide sensitive, cost-effective speciation with minimal infrastructure requirements, making them ideal for field deployment and rapid screening [5]. For more comprehensive analysis, particularly when unknown species may be present, LC-ICP-MS remains the benchmark technique, especially when equipped with collision/reaction cell technology to handle complex matrices [28]. The ImpAs chemisorbent approach offers a simple, selective alternative for As(V) determination, though its performance in high-iron waters requires careful consideration [17].
Successful application of any speciation method in complex aquatic matrices necessitates rigorous validation using standard addition protocols, certified reference materials, and comparison with established reference methods where possible. Through appropriate method selection and optimization, reliable arsenic speciation can be achieved even in challenging high-ionic-strength and iron-rich environments, providing essential data for environmental monitoring, remediation design, and public health protection.
In the development of portable anodic stripping voltammetry (ASV) methods for arsenic speciation in aquatic systems, electrode fouling represents a critical challenge that can severely compromise analytical performance. Fouling manifests as the undesirable accumulation of materials on the electrode surface, altering its electrochemical properties and leading to diminished sensitivity, selectivity, and reproducibility [45]. For researchers and drug development professionals relying on accurate arsenic speciation data—where the differentiation between arsenite (As(III)) and arsenate (As(V)) carries significant toxicological implications—understanding and mitigating electrode fouling is paramount to ensuring data integrity, especially in field-deployment scenarios where laboratory-grade maintenance is impractical [5] [17]. This application note synthesizes current research to outline the causes of electrode fouling, presents actionable prevention strategies, and details protocols for in-field regeneration, specifically contextualized for portable ASV systems analyzing complex aquatic matrices.
Causes and Mechanisms of Electrode Fouling
Electrode fouling is a multifaceted phenomenon whose mechanisms depend on electrode material, applied potential, and sample composition.
- Biofouling and Chemical Fouling: In electrochemical sensors, fouling is broadly categorized as biofouling (accumulation of biomolecules) or chemical fouling (deposition of unwanted chemical species). In fast-scan cyclic voltammetry, both mechanisms significantly decrease sensitivity and cause peak voltage shifts in neurotransmitter detection, illustrating a universal problem in electroanalysis [45].
- Passivation Layers: In electrocoagulation studies, which share similarities with sensor applications, electrode passivation refers to the formation of an insulating film, primarily composed of metal oxides and hydroxides, on the electrode surface. This layer increases electrical resistance and reduces the electroactive area available for redox reactions [46]. For aluminium electrodes used in treating brackish peat water, passivation occurs via aluminium oxide layer formation, hindering the electrode's dissolution and subsequent coagulant generation [47].
- Specific Fouling Agents in Arsenic Speciation: Analysis of Ag/AgCl reference electrodes after chronic implantation revealed that sulfide ions cause a significant decrease in open circuit potential and subsequent peak voltage shifts in voltammograms [45]. Energy-dispersive X-ray spectroscopy (EDX) confirmed increased sulfide concentrations on fouled electrodes [45] [47]. In natural waters, dissolved organic matter (e.g., humic substances) and precipitates of metal hydroxides (e.g., iron) can adsorb onto electrode surfaces, forming insulating layers that foul the electrode [47] [17].
Table 1: Common Fouling Agents in Aquatic Arsenic Speciation Using ASV
| Fouling Agent Category | Specific Examples | Primary Impact on Electrode |
|---|---|---|
| Inorganic Salts & Ions | Sulfide ions (S²⁻), Chloride ions (Cl⁻) [45] | Alters reference electrode potential; causes peak voltage shifts |
| Metal Oxides/Hydroxides | Aluminium oxide (Al₂O₃), Iron (oxy)hydroxides [46] [47] | Forms an insulating passivation layer; increases resistance |
| Natural Organic Matter | Humic acids, Fulvic acids [47] | Adsorbs to surface, blocking active sites and reducing sensitivity |
| Biological Material | Proteins, Microorganisms [45] | Forms an insulating biofilm on the electrode surface |
Fouling Prevention Strategies
Proactive prevention is the most effective approach to managing electrode fouling. Strategies can be categorized into operational parameter optimization, electrode design and material selection, and solution chemistry management.
Operational Parameter Optimization
- Current Mode and Polarity Reversal: In electrocoagulation, applying a periodic polarity reversal to the electrodes has proven effective in mitigating passivation. This technique disrupts the stable formation of passivating layers on the electrode surface [46]. A similar approach, using pulsed potential waveforms instead of constant applied potential, can be integrated into ASV protocols to minimize fouling.
- Optimized Hydrodynamics and Turbulence: Introducing controlled turbulence or optimizing stirring speed near the electrode surface can prevent the localized accumulation of foulants and reaction products. This enhances mass transfer away from the electrode, reducing the deposition of insulating species [46]. In a portable ASV setup, this is typically achieved using a rotating disk electrode or an efficient stir bar.
Electrode Design and Material Selection
- Novel Electrode Materials and Coatings: Developing electrodes with anti-fouling surfaces is a key research focus. This includes creating superhydrophobic or omniphobic surfaces that physically repel foulants, as explored in membrane technology [48]. For in-situ analysis, designing electrodes with anti-fouling coatings (e.g., Nafion membranes) can selectively exclude interfering surfactants and large organic molecules while allowing the target analyte (e.g., arsenic) to reach the electrode surface.
- Perforated and Novel Geometry Electrodes: Studies on electrocoagulation have shown that using perforated electrodes can enhance mass transfer and reduce the adherence of fouling layers, a design principle that can be adapted to sensor electrodes [47].
Solution Chemistry Management
- Introduction of Aggressive Ions: The deliberate addition of chloride ions (Cl⁻) to the solution has been documented as a strategy to mitigate electrode passivation. Chloride ions can compete with foulants for surface adsorption sites and may help in destabilizing and breaking down existing oxide layers [46]. In certain water treatment contexts, a controlled mixture of seawater has been used to leverage this effect [47].
- Sample Pre-treatment: For complex matrices like brackish peat water, coupling electrocoagulation with a pre-treatment step (e.g., filtration, adsorption) can remove a significant portion of the foulants (e.g., suspended solids, natural organic matter) before the sample reaches the sensor, thereby extending electrode life [47]. For portable ASV, this could involve in-line filters.
In-Field Regeneration Techniques
When prevention is insufficient, in-field regeneration techniques are essential for restoring electrode performance without requiring replacement or laboratory-based reconditioning.
Electrochemical Cleaning Methods
Electrochemical methods are particularly suitable for integration into portable instrument protocols.
- Potentiostatic/Potentiodynamic Cleaning: This involves applying a specific potential or potential sequence to the fouled electrode in a clean supporting electrolyte. The applied potential is chosen to either oxidize or reduce the fouling layer, desorbing it from the surface. For example, a high anodic potential can oxidize organic foulants, while a cathodic potential can reduce metal oxide layers.
- Tafel Plot Analysis for Monitoring: Tafel plot analysis is a potentiodynamic method that can be used not just for regeneration but also to monitor the extent of fouling and the effectiveness of the cleaning process. The Tafel equation describes the relationship between the electrochemical reaction rate (current) and the overpotential. A shift in the Tafel plot before and after cleaning can quantitatively indicate the restoration of electrode activity [47].
Table 2: Comparison of In-Field Electrode Regeneration Techniques
| Technique | Principle | Best Suited For | Limitations |
|---|---|---|---|
| Anodic Cleaning | Application of a high positive potential to oxidize organic foulants [47] | Biofilms, adsorbed natural organic matter | Can accelerate the formation of metal oxide layers on some electrode materials |
| Cathodic Cleaning | Application of a negative potential to reduce metal oxide layers [47] | Passivation layers from metal oxides/hydroxides | May deposit metals from solution if present; can reduce surface oxides on noble metal electrodes |
| Pulsed Potential Waveforms | Rapid cycling between oxidizing and reducing potentials to dislodge foulants [46] | Mixed fouling mechanisms (organic and inorganic) | Requires optimization of pulse sequence (duration, amplitude) |
| Mechanical Polishing | Physical abrasion to remove the fouling layer (e.g., with alumina slurry) | Severe, tenacious fouling layers | Not easily automated; can alter electrode geometry; risk of contamination |
Protocol: In-Field Regeneration of a Gold Working Electrode for ASV
This protocol is designed for a solid gold working electrode used in portable ASV for arsenic speciation.
Research Reagent Solutions:
- Regeneration Solution 1 (Acidic Cleaner): 0.1 M HCl or 0.05 M H₂SO₄.
- Regeneration Solution 2 (Supporting Electrolyte): A clean, non-fouling electrolyte matching that used in your ASV measurement (e.g., 0.1 M acetate buffer, pH 4.5).
- Purified Water: Deionized or ultrapure water for rinsing.
Step-by-Step Procedure:
- Rinse: Thoroughly rinse the fouled electrode with purified water to remove loosely adhered particles and bulk sample matrix.
- Cathodic Cleaning (for Oxide Removal):
- Immerse the electrode in Regeneration Solution 1 (0.1 M HCl).
- Apply a constant potential of -0.5 V (vs. the instrument's reference) for 30 seconds with gentle stirring.
- This step aims to reduce any surface gold oxides.
- Anodic Cleaning (for Organic Fouling):
- Transfer the electrode to a fresh aliquot of Regeneration Solution 2.
- Apply a constant potential of +1.5 V for 10-15 seconds. Caution: Avoid prolonged application at high anodic potentials.
- Cyclic Potential Cleaning:
- In the same supporting electrolyte (Regeneration Solution 2), perform 10-20 continuous cyclic voltammetry scans between -0.3 V and +1.3 V at a scan rate of 500 mV/s. This helps to further desorb foulants and re-establish a clean, reproducible surface.
- Final Rinse and Check:
- Rinse the electrode again with purified water and place it in a fresh sample or standard solution.
- Record a voltammogram of a standard arsenic solution to verify that sensitivity and peak shape have been restored.
Monitoring and Characterization of Fouling
Effective management of fouling requires robust methods for its detection and quantification.
- Electrochemical Methods: Tafel plot analysis is a powerful tool for diagnosing fouling. A change in the Tafel slope or a shift in the corrosion potential indicates surface modification due to fouling or passivation [47]. Additionally, monitoring changes in key ASV parameters—such as a decrease in peak current, a shift in peak potential, or an increase in background current—serves as a direct, in-situ indicator of fouling.
- Advanced Characterization: While primarily laboratory-based, techniques like Energy-Dispersive X-ray (EDX) spectroscopy can be used post-analysis to identify the elemental composition of fouling layers, providing invaluable information for developing targeted mitigation strategies [45] [47]. For example, EDX confirmed sulfide as a key fouling agent on implanted Ag/AgCl electrodes [45].
Electrode fouling is an inevitable challenge in the application of portable ASV for arsenic speciation in natural waters. A comprehensive strategy combining preventive measures—such as optimized operational parameters, thoughtful electrode design, and sample management—with practical, in-field regeneration protocols is essential for maintaining data quality and operational longevity. The integration of real-time fouling monitoring through electrochemical techniques like Tafel analysis will further enhance the reliability of field-deployable sensors. As research progresses, the development of more robust, fouling-resistant electrode materials and the potential deployment of interpretable machine learning models for predicting and managing fouling present promising avenues for future innovation [46].
The development of portable analytical methods for arsenic speciation in aquatic systems is a critical response to the global health challenge posed by arsenic-contaminated water. The World Health Organization (WHO) has set a maximum limit of 10 parts per billion (ppb) for arsenic in drinking water, necessitating highly sensitive detection methods [1]. This application note focuses on parameter optimization for Anodic Stripping Voltammetry (ASV) to achieve sub-ppb sensitivity, a crucial capability for field-based arsenic speciation. ASV offers distinct advantages for field deployment, including portable instrumentation, cost-effectiveness, and the ability to provide information on arsenic speciation between the more toxic As(III) and less toxic As(V) forms [5] [49]. Within the broader context of thesis research on portable ASV methods, mastering these optimization parameters is fundamental to developing reliable field-deployable systems for arsenic monitoring in diverse aquatic environments.
Technical Principles and Significance
Fundamentals of Anodic Stripping Voltammetry (ASV) for Arsenic
ASV is a two-step electroanalytical technique renowned for its excellent sensitivity for metal detection, capable of reaching sub-ppb detection limits with portable instrumentation [49] [20]. The technique first involves a cathodic pre-concentration step where arsenic ions in solution are electrochemically reduced to their elemental form and deposited onto the working electrode surface. This is followed by an anodic stripping step where the deposited metal is re-oxidized back into solution, generating a measurable current signal [20]. The peak current of this stripping signal is proportional to the concentration of arsenic in the original sample.
For arsenic speciation, ASV leverages the different electrochemical behaviors of As(III) and As(V). As(III) can be directly determined, while As(V) typically requires reduction to As(III) or As(0) before detection [5] [6]. The speciation information is crucial for accurate risk assessment, as As(III) is significantly more toxic and mobile in aquatic environments than As(V) [1].
The Critical Need for Low Detection Limits
Achieving sub-ppb detection limits is not merely an analytical exercise but a public health imperative. With over 230 million people worldwide affected by arsenic toxicity from contaminated water sources, the ability to reliably detect arsenic at concentrations below the WHO 10 ppb guideline is essential for early warning and effective remediation [1]. The table below summarizes key detection limits achieved in recent ASV arsenic studies.
Table 1: Comparison of ASV Performance for Arsenic Detection in Recent Studies
| Detection Technique | Electrode Material | Target Analyte | Achieved LOD | Reference |
|---|---|---|---|---|
| DPASV* | Solid Gold Electrode | As(total) | 0.10 μg L⁻¹ (0.1 ppb) | [5] |
| ASV with UPD | Gold Macroelectrode | As(III) & Total As | 0.8 μg L⁻¹ (0.8 ppb) | [6] |
| Optimized ASV | Au Wire Electrode | As(III) | 1.3 ppb | [50] |
| Optimized ASV | Au Wire Electrode | As(V) | 0.27 ppb | [50] |
DPASV: Differential Pulse Anodic Stripping Voltammetry; *UPD: Underpotential Deposition*
Experimental Protocols for Sub-ppb ASV
Electrode Selection and Preparation Protocol
Working Electrode Selection:
- Material: Solid gold electrodes (SGE) are preferred for arsenic detection due to gold's favorable interaction with arsenic species [5]. Gold macroelectrodes and rotating gold disk electrodes have demonstrated excellent performance.
- Format: Both stationary and rotating disk configurations can be used. Rotating electrodes enhance mass transport during deposition, improving sensitivity [5].
- Historical Context: While mercury electrodes were historically preferred for ASV due to their wide cathodic window and formation of homogeneous amalgams, toxicity concerns have rendered them obsolete, necessitating solid electrode alternatives [49] [20].
Electrode Pretreatment Protocol:
- Mechanical Polishing: Polish the gold electrode surface with successive grades of alumina slurry (1.0, 0.3, and 0.05 μm) on a microcloth pad.
- Sonication: Sonicate the polished electrode in deionized water for 2-3 minutes to remove adsorbed alumina particles.
- Electrochemical Activation: Perform potential cycling in 0.5 M H₂SO₄ from -0.2 to +1.5 V (vs. Ag/AgCl) at 100 mV/s for 20-30 cycles until a stable cyclic voltammogram characteristic of a clean gold surface is obtained.
- Rinsing: Rinse thoroughly with deionized water between measurements and before use [5] [50].
ASV Measurement and Speciation Protocol for Arsenic
Solutions and Reagents:
- Supporting Electrolyte: Use 1 M HCl or HNO₃ as the supporting electrolyte for arsenic detection [50]. The choice depends on compatibility with other potential metal interferences.
- Standard Solutions: Prepare As(III) standard solutions from sodium (meta)arsenite (NaAsO₂). Prepare As(V) standards from sodium arsenate (Na₂HAsO₄·7H₂O).
- Water Quality: Use high-purity deionized water (resistivity >18 MΩ·cm) to minimize contamination [51].
Speciation Procedure:
- Sample Preservation: Collect water samples in acid-washed containers. For speciation analysis, avoid acidification as this may alter arsenic oxidation states.
- Direct As(III) Determination:
- Transfer 10 mL of sample to the electrochemical cell.
- Apply a deposition potential of -0.3 V to -0.9 V (vs. Ag/AgCl) for 60-120 seconds with stirring.
- Record the DPASV scan from the deposition potential to +0.5 V.
- Measure the stripping peak at approximately +0.1 V for As(0) to As(III) oxidation [5] [6].
- Total Inorganic Arsenic Determination:
- To the same sample, apply a more negative deposition potential of -1.2 V to -1.3 V (vs. Ag/AgCl) for 60-120 seconds with stirring.
- This reduces As(V) to As(0) electrochemically via nascent hydrogen [5].
- Record the DPASV scan and measure the combined arsenic stripping peak.
- As(V) Calculation:
Diagram: ASV Workflow for Arsenic Speciation in Water Samples
Parameter Optimization for Sub-ppb Sensitivity
Deposition Potential Optimization:
- For As(III) detection: Optimize between -0.3 V and -0.9 V (vs. Ag/AgCl) [5] [6].
- For total arsenic detection: Use -1.2 V to -1.3 V (vs. Ag/AgCl) to ensure complete reduction of As(V) to As(0) [5].
- The optimal potential provides maximum stripping current without inducing excessive hydrogen evolution or background interference.
Deposition Time Optimization:
- Typical range: 60-300 seconds, depending on required sensitivity.
- Longer deposition times increase analytical sensitivity but may promote electrode fouling.
- For sub-ppb detection, use 120-180 seconds deposition time [5].
- Calibration curves must be constructed using the same deposition time for all standards and samples.
Instrumental Parameters:
- Pulse Amplitude: 25-50 mV for DPASV
- Step Potential: 2-5 mV
- Pulse Period: 0.1-0.5 s
- Equilibrium Time: 10-15 s before stripping
Table 2: Key Parameter Optimization for Sub-ppb ASV Detection of Arsenic
| Parameter | Optimal Range | Effect on Sensitivity | Practical Considerations |
|---|---|---|---|
| Deposition Potential | -0.3V to -0.9V (AsIII)-1.2V to -1.3V (Total As) | Determines which species are deposited | Too negative causes H₂ evolution; too positive misses deposition |
| Deposition Time | 60-300 seconds | Longer time = higher sensitivity | Balance sensitivity with analysis time and electrode fouling |
| Electrode Rotation/Stirring | 1500-3000 rpm | Enhanced mass transport = higher deposition efficiency | Essential for reproducible hydrodynamics |
| Supporting Electrolyte | 1M HCl or HNO₃ | Affects speciation and deposition efficiency | HCl may provide better resolution for some metal mixtures |
| Pulse Amplitude (DPASV) | 25-50 mV | Larger amplitude = higher signal | Too large decreases resolution |
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for ASV Arsenic Speciation
| Reagent/Material | Function | Specifications & Notes |
|---|---|---|
| Solid Gold Working Electrode | Sensor surface for arsenic deposition and stripping | Rotating disk configuration preferred; 2-3 mm diameter |
| Platinum Counter Electrode | Completes the electrical circuit in the 3-electrode system | Coil or wire configuration |
| Ag/AgCl Reference Electrode | Provides stable reference potential | 3M KCl filling solution |
| High-Purity HCl or HNO₃ | Supporting electrolyte | Ultrapure grade (<10 ppt metal contamination) |
| As(III) Standard Solution | Calibration and quantification | Sodium meta-arsenite (NaAsO₂) in high-purity water |
| As(V) Standard Solution | Calibration and quantification | Sodium arsenate (Na₂HAsO₄·7H₂O) in high-purity water |
| High-Purity Deionized Water | Sample preparation and dilution | Resistivity >18 MΩ·cm |
| Conditioning Solution | Electrode cleaning and activation | 0.5 M H₂SO₄ (high purity) |
Data Analysis and Validation
Calibration and Quantitative Analysis
Calibration Approach:
- Use standard addition method for complex matrices to account for matrix effects.
- For cleaner samples, external calibration with matrix-matched standards can be used.
- Linear calibration range for arsenic typically extends from 0.1-100 ppb with optimized parameters [5] [6].
Detection Limit Calculation:
- Limit of Detection (LOD) = 3 × SD₀ / S
- Where SD₀ is the standard deviation of the blank signal
- S is the sensitivity (slope of calibration curve)
- Limit of Quantification (LOQ) = 10 × SD₀ / S [51]
Method Validation
Cross-Validation with Reference Methods:
- Validate ASV results against established techniques such as:
- Acceptance criterion: <15% difference between methods for concentrations >10 ppb.
Quality Control Measures:
- Analyze certified reference materials (CRMs) with each batch.
- Include duplicate samples and spikes for precision and accuracy assessment.
- Maintain a control chart for continuing performance verification.
Diagram: Signal Optimization Pathway for Sub-ppb Detection
Troubleshooting and Technical Notes
Common Issues and Solutions:
- Poor Peak Resolution: Optimize deposition potential and pulse parameters; check electrode condition.
- Signal Drift: Ensure consistent temperature; use internal standard if available.
- Low Sensitivity: Extend deposition time; increase stirring rate; check electrode surface.
- Irreproducible Results: Standardize electrode pretreatment; control deposition time precisely.
Field Deployment Considerations:
- Portable potentiostats now offer performance comparable to benchtop instruments for ASV [5].
- For field analysis, include a sample filtration step (0.45 μm) to remove particulates.
- Temperature control is critical for reproducible results; allow samples to equilibrate to ambient temperature.
Interference Management:
- Copper can interfere with arsenic detection; use modified deposition potentials or chemical masking [49].
- Organic matter may foul the electrode surface; UV digestion or dilution can mitigate this effect.
- High concentrations of other metals may require standard addition rather than external calibration.
The development of portable analytical methods for arsenic speciation in aquatic systems is critical for environmental monitoring and public health protection. This application note details comprehensive calibration practices and quality control (QC) protocols for a portable anodic stripping voltammetry (ASV) method, validated against established laboratory techniques. The procedures outlined herein ensure data reliability for on-site determination of inorganic arsenite (As(III)) and arsenate (As(V)), enabling accurate risk assessment and remediation efforts in field settings.
The toxicity, mobility, and removal efficiency of arsenic in water sources are highly dependent on its chemical speciation [17]. Inorganic As(III) is significantly more toxic and more difficult to remove via conventional water treatment processes than As(V) [17]. Consequently, simple, portable, and reliable speciation methods are indispensable for the multidisciplinary effort to tackle arsenic contamination globally [17] [5].
Portable Anodic Stripping Voltammetry (ASV) has emerged as a powerful technique for on-site arsenic speciation, offering rapid, sensitive, and cost-effective analysis without the need for complex laboratory infrastructure [5]. However, the reliability of data generated in the field is contingent upon rigorous calibration and a robust QC framework. This protocol outlines the practices necessary to ensure the quality of data produced by a portable ASV method for arsenic speciation in natural waters, framed within a broader research context aimed at improving field-based analytical techniques.
Experimental Protocols
Key Principles of Arsenic Speciation by ASV
The core principle of the method involves the electrochemical differentiation and quantification of As(III) and As(V) using a solid gold electrode (SGE) [5]. The method leverages distinct electrochemical behaviors of the two species:
- As(III) is determined directly by differential pulse anodic stripping voltammetry (DPASV) after deposition at a potential of -0.3 V, with the stripping peak observed at approximately +0.1 V [5].
- Total inorganic arsenic (As(tot)) is determined after the in-situ electrochemical reduction of As(V) to elemental arsenic (As⁰) using nascent hydrogen generated at a potential of -1.2 V [5].
- As(V) concentration is calculated indirectly by subtracting the directly measured As(III) concentration from the measured As(tot) concentration [5]. This electrochemical reduction approach minimizes reagent consumption and analysis time, making it ideal for field deployment.
Detailed Workflow for Field Speciation
The following workflow, also depicted in Figure 1, must be followed precisely for reliable field speciation.
Figure 1. Workflow for field arsenic speciation using portable ASV.
Sample Collection and Preservation
- Collection: Collect water samples in opaque polyethylene bottles to prevent photo-oxidation of As(III) to As(V) [52].
- Filtration: Immediately filter samples through a 0.45 µm membrane filter to remove suspended particulates and define the "dissolved" fraction [52].
- Preservation: Preserve the filtered sample with Ethylenediaminetetraacetic acid (EDTA). The EDTA complexes cations like iron (Fe) and manganese (Mn), preventing the formation of oxyhydroxide precipitates that can adsorb dissolved arsenic species and negatively bias results [52]. Samples should be analyzed as rapidly as possible.
Instrumental Analysis Procedure
- Instrument Setup: Configure the portable potentiostat with a solid gold working electrode, a platinum counter electrode, and a reference electrode. Ensure all connections are secure.
- Calibration Verification: Analyze a calibration standard (see Section 3.1) to verify the instrument response before proceeding with samples.
- Analysis of Sub-sample A (for As(III)):
- Transfer a known volume of preserved sample to the electrochemical cell.
- Deposition: Apply a deposition potential of -0.3 V to the working electrode for a fixed time (e.g., 60-180 seconds) while rotating the electrode. This step pre-concentrates As(0) onto the gold surface.
- Stripping: After the deposition period, scan the potential using the DPASV mode from a negative potential to a positive potential. The stripping current for As(0) to As(III) will produce a peak at approximately +0.1 V [5].
- Record the peak height or area.
- Analysis of Sub-sample B (for As(tot)):
- Transfer a new aliquot of the preserved sample to the cell.
- Reduction & Deposition: Apply a deposition potential of -1.2 V. This highly negative potential generates nascent hydrogen at the electrode surface, which electrochemically reduces As(V) to As(0), which is simultaneously deposited on the electrode [5].
- Stripping: Perform the DPASV scan as in Step 3. The resulting signal corresponds to the total inorganic arsenic content (As(III) + As(V)).
- Record the peak height or area.
- Calculation: The concentration of As(V) is determined by the difference between the total arsenic and As(III) concentrations.
Calibration & Quality Control Framework
A systematic QC framework is non-negotiable for generating reliable data. The following diagram and subsequent sections detail this framework.
Figure 2. Quality control framework for field ASV analysis.
Calibration Practices
Calibration establishes the relationship between the instrument's response (stripping peak current) and the analyte concentration.
- Calibration Standards: Prepare a series of at least five standard solutions across the expected concentration range of the samples (e.g., 0.5, 1.0, 5.0, 10.0, and 20.0 µg/L) from a certified arsenic standard stock solution [52]. The standards should be prepared in a matrix similar to the sample, such as reagent water with a matching pH and ionic strength.
- Calibration Curve: Analyze the calibration standards and plot the stripping peak current (or area) against the concentration. The curve should be linear, with a correlation coefficient (R²) of ≥0.995.
- Calibration Verification: A calibration standard (CAL) should be analyzed after every 10 samples, or at a frequency defined in the quality control plan, to verify the stability of the initial calibration [52]. The measured value of the verification standard should be within ±15% of the true value.
Quality Control Procedures
QC procedures are designed to monitor the analytical process and ensure data quality on a batch-by-batch basis.
Table 1: Required Quality Control Measures
| QC Measure | Type | Frequency | Acceptance Criteria | Purpose & Action |
|---|---|---|---|---|
| Laboratory Blank (LB) | Blank | Minimum 1 per 10 samples [52] | Arsenic concentration < Method Detection Limit (MDL) | Monitor contamination from reagents, apparatus, or the environment. If criteria are not met, investigate and re-analyze affected samples. |
| Quality Control Sample (QCS) | Independent Standard | Minimum 1 per 10 samples [52] | Recovery within 85-115% [52] | Assess analytical accuracy and performance of the calibration curve. Prepared from a source independent of the calibration standards. |
| Calibration Verification (CV) | Standard | Every 10 samples or with each batch [52] | Measured value within ±15% of true value | Verify that the instrument calibration remains valid throughout the analytical run. |
| Method Detection Limit (MDL) | --- | Established initially | e.g., 0.10 µg L⁻¹ for As(tot) [5] | The minimum concentration that can be detected with 99% confidence. Established following regulatory guidelines (e.g., USEPA). |
Data Presentation and Analysis
Quantitative data must be presented clearly and concisely. The following table summarizes the performance characteristics that should be established and reported for the ASV method.
Table 2: Typical Method Performance Characteristics for Portable ASV Arsenic Speciation
| Parameter | Value for As(III) | Value for As(tot) | Notes |
|---|---|---|---|
| Linear Range | Up to 100 µg/L | Up to 100 µg/L | Applicable concentration range as per USGS method [52]. |
| Limit of Detection (LOD) | ~0.10 µg/L | ~0.10 µg/L | LOD of 0.10 µg L⁻¹ has been reported for As(tot) [5]. |
| Precision | --- | ~1.00 % RSD | Precision, as Relative Standard Deviation (RSD), can be as low as 1.00% at a spiking level of 10 µg/L [52]. |
| Accuracy (Bias) | ~96% Recovery | ~100% Recovery | Recovery rates for speciation methods can be established using reference materials [52]. |
| Validation vs. Reference Method | --- | --- | Results should show satisfactory agreement with reference techniques like HG-ICP-OES or HG-ICP-MS [5]. |
The Scientist's Toolkit: Research Reagent Solutions
The following reagents and materials are essential for the successful execution of this protocol.
Table 3: Essential Research Reagents and Materials
| Item | Function / Purpose | Specification / Notes |
|---|---|---|
| Solid Gold Electrode (SGE) | Working electrode for ASV; provides a surface for arsenic deposition and stripping. | Preferred for arsenic detection due to its sensitivity and reliability [5]. Requires regular cleaning and polishing. |
| Portable Potentiostat | Instrument for applying electrical potentials and measuring currents. | Must be capable of performing DPASV and applying potentials from -1.2 V to +0.5 V. |
| Arsenic Standard Solutions | For calibration and preparation of QCS. | Certified stock solutions (e.g., 1000 mg/L) of As(III) and As(V). |
| EDTA (Ethylenediaminetetraacetic acid) | Sample preservative; complexes cationic interferents (Fe, Mn). | Prevents precipitation and loss of arsenic onto solid phases [52]. |
| Opaque Polyethylene Bottles | Sample containers. | Prevent photo-oxidation of As(III) to As(V) [52]. |
| 0.45 µm Membrane Filters | For field filtration of samples. | Defines the "dissolved" arsenic fraction. |
| LC-SAX Cartridges (Alternative Method) | For field speciation by solid-phase extraction (SPE). | Retains As(V) and other charged species; allows As(III) to pass through [52]. Not used in the direct ASV method but a key tool in other field speciation workflows. |
Benchmarking Portable ASV: Validation Against Standard Laboratory Techniques
The accurate speciation of arsenic in aquatic systems is critical for assessing environmental and public health risks. This application note provides a comparative analysis of two analytical techniques for arsenic speciation: the established laboratory standard, High-Performance Liquid Chromatography coupled with Inductively Coupled Plasma Mass Spectrometry (HPLC-ICP-MS), and the emerging Anodic Stripping Voltammetry (ASV). Framed within the context of developing portable arsenic speciation methods, we evaluate the performance, applicability, and operational requirements of each technique. The data and protocols presented herein are designed to aid researchers and scientists in selecting the appropriate methodology for their specific arsenic speciation needs, with a particular focus on field-based analysis.
Arsenic contamination of water resources is a global environmental health concern. The toxicity of arsenic is highly dependent on its chemical form; inorganic species, arsenite (As(III)) and arsenate (As(V)), are significantly more toxic than organic species such as dimethylarsinic acid (DMA) and monomethylarsonic acid (MMA) [53] [21]. Consequently, the determination of total arsenic concentration is insufficient for accurate risk assessment, creating a pressing need for reliable speciation analysis.
For years, HPLC-ICP-MS has been the gold standard for comprehensive arsenic speciation, offering high sensitivity and the ability to separate and quantify multiple species simultaneously [54] [55] [21]. However, its high cost, complex operation, and confinement to laboratory settings limit its applicability for rapid, on-site monitoring.
In response, portable Anodic Stripping Voltammetry (ASV) methods have been developed as a rapid, cost-effective, and field-deployable alternative for the speciation of inorganic arsenic [5] [12]. This analysis directly compares these two techniques across key performance metrics to delineate their respective advantages and ideal use cases.
Performance Metrics and Comparative Data
The following tables summarize the core performance characteristics and operational profiles of HPLC-ICP-MS and ASV based on current literature.
Table 1: Analytical Performance Comparison
| Parameter | HPLC-ICP-MS | Anodic Stripping Voltammetry (ASV) |
|---|---|---|
| Primary Application | Speciation of inorganic & organic As [53] [54] | Speciation of inorganic As (As(III) & As(V)) [5] [12] |
| Detection Limit | < 1 µg As L⁻¹ for various species [56] | 0.10 µg L⁻¹ for total inorganic As [5] |
| Analysis Time | ~4 to 15 minutes per sample [54] [21] | Rapid; suitable for high-throughput [5] |
| Key Advantage | High sensitivity & multi-species capability | Portability, low cost, and speed [5] [12] |
Table 2: Operational and Methodological Profile
| Aspect | HPLC-ICP-MS | Anodic Stripping Voltammetry (ASV) |
|---|---|---|
| Speciation Mechanism | Chromatographic separation of species [54] | Electrochemical reduction & selective deposition [5] |
| Typical Sample Matrices | Natural waters, rice, herbal medicines, biological fluids [53] [54] [56] | Synthetic & natural waters (groundwater, surface water) [5] [12] |
| Portability | Laboratory-bound instrument | Portable potentiostats enable field use [5] |
| Cost & Complexity | High capital and operational cost; requires skilled personnel | Lower cost; simpler operation and minimal reagents [5] [21] |
Experimental Protocols
Protocol for Arsenic Speciation via Portable ASV
This protocol is adapted from methods developed for the rapid speciation of inorganic arsenic in aquatic systems using a portable potentiostat [5] [12].
1. Principle: As(III) is selectively determined by Differential Pulse Anodic Stripping Voltammetry (DPASV) at a solid gold electrode (SGE). The total inorganic arsenic (As(III) + As(V)) is determined after an in-situ electrochemical reduction of As(V) to As(0), which is then quantified. The As(V) concentration is calculated by subtracting the As(III) concentration from the total inorganic arsenic concentration [5].
2. Materials and Reagents:
- Portable potentiostat with DPASV capability
- Solid Gold Electrode (SGE) and corresponding reference/counter electrodes
- Supporting electrolyte (e.g., HCl)
- Standard solutions of As(III) and As(V)
- Ultrapure water
- Sampling vials and pipettes
3. Procedure: Step 1: Calibration.
- Prepare standard solutions of As(III) in the expected concentration range (e.g., 0-50 µg/L) in a matrix similar to the sample.
- For each standard, transfer to the electrochemical cell, add supporting electrolyte, and purge with inert gas.
- Deposit As(III) at -0.3 V for a fixed time (e.g., 60-120 s) with electrode rotation.
- Record the stripping peak for As(0) → As(III) at approximately +0.1 V using DPASV parameters.
- Plot a calibration curve of peak current vs. As(III) concentration.
Step 2: Determination of As(III).
- Treat the water sample identically to the calibration standards to obtain the As(III) concentration directly from the calibration curve.
Step 3: Determination of Total Inorganic Arsenic.
- Take an aliquot of the same water sample.
- Apply an electrochemical pre-reduction step at -1.2 V to reduce As(V) to As(0) using nascent hydrogen.
- Following this reduction, the deposited As(0) is stripped, and the total inorganic arsenic signal is recorded.
- The total inorganic arsenic concentration is determined from the calibration curve established with As(III) standards.
Step 4: Data Analysis.
- Calculate the As(V) concentration using the formula: As(V) = [Total Inorganic As] - [As(III)].
4. Validation: Validate the ASV method by analyzing samples with known concentrations or by cross-checking results against a reference method like HG-ICP-OES [5].
Protocol for Arsenic Speciation via HPLC-ICP-MS
This protocol outlines a general method for the speciation of common arsenic species in water samples, based on established procedures [54] [56] [21].
1. Principle: Arsenic species are separated using anion-exchange chromatography. The eluted species are then introduced into the ICP-MS, which serves as an element-specific detector, providing highly sensitive and quantitative detection of arsenic at m/z 75 [54] [21].
2. Materials and Reagents:
- HPLC system coupled to ICP-MS
- Anion-exchange chromatography column (e.g., Capcell Pak C18, PRP-X100)
- Mobile phase (e.g., ammonium carbonate, ammonium phosphate buffers, pH-adjusted)
- Standard solutions for As(III), As(V), DMA, MMA
- Certified Reference Material (e.g., SRM 1568b Rice Flour) for validation
- Syringe filters (0.45 µm)
3. Procedure: Step 1: Sample Preparation.
- Filter water samples through a 0.45 µm membrane filter.
- For solid samples, employ an extraction protocol (e.g., microwave-assisted extraction with water or mild solvents) to preserve species integrity [54].
Step 2: Chromatographic Separation.
- Inject the sample onto the HPLC column.
- Elute the species using an isocratic or gradient method with a mobile phase such as (NH₄)₂CO₃ or (NH₄)₂HPO₄. Methanol (1-3%) may be added to enhance peak resolution [54] [21].
- Typical retention times for common species should be established, with total run times as low as 4 minutes in optimized methods [54].
Step 3: ICP-MS Detection.
- The effluent from the HPLC column is directly introduced into the ICP-MS nebulizer.
- Monitor arsenic at m/z 75. Use collision/reaction cell technology with gases like helium or hydrogen to mitigate potential polyatomic interferences (e.g., ⁴⁰Ar³⁵Cl⁺) [57] [21].
Step 4: Quantification.
- Quantify individual species by comparing peak areas against external calibration curves of standard solutions.
- Verify method accuracy using Certified Reference Materials (CRMs) with known concentrations of arsenic species [54].
Workflow and Logical Diagrams
The following diagram illustrates the core logical and procedural differences between the ASV and HPLC-ICP-MS methods for arsenic speciation.
Arsenic Speciation Workflow Comparison
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Arsenic Speciation
| Item | Function/Application | Technique |
|---|---|---|
| Solid Gold Electrode (SGE) | Working electrode for the selective deposition and stripping of arsenic. | ASV [5] |
| Portable Potentiostat | Instrument for controlling applied potential and measuring current in voltammetry. | ASV [5] |
| Anion-Exchange Column | HPLC column for separating anionic arsenic species (As(III), As(V), DMA, MMA). | HPLC-ICP-MS [54] [21] |
| Ammonium Carbonate Buffer | Common mobile phase for the chromatographic separation of arsenic species. | HPLC-ICP-MS [54] [21] |
| Certified Reference Materials (CRMs) | Materials with certified concentrations of arsenic species for method validation and quality control. | Both [54] |
| Collision/Reaction Gases (He, H₂, O₂) | Gases used in ICP-MS to eliminate polyatomic interferences on arsenic detection at m/z 75. | HPLC-ICP-MS [57] [21] |
The choice between ASV and HPLC-ICP-MS for arsenic speciation is dictated by the specific analytical requirements.
HPLC-ICP-MS remains the unmatched reference method for comprehensive speciation, capable of quantifying both inorganic and organic arsenic species with exceptional sensitivity across diverse and complex matrices. It is the definitive choice for laboratory-based research and regulatory compliance testing where a complete speciation profile is necessary.
ASV emerges as a powerful, specialized tool for the speciation of inorganic arsenic in aquatic systems. Its principal advantages of portability, rapid analysis, low cost, and operational simplicity make it ideally suited for field deployment, high-throughput screening, and monitoring in resource-limited settings.
In the context of developing portable methods for arsenic speciation in aquatic systems research, ASV provides a viable and effective strategy for on-site inorganic arsenic monitoring. However, for studies requiring a full arsenic species profile or dealing with complex matrices where organic species are present, HPLC-ICP-MS is the indispensable, albeit less portable, standard.
The accurate speciation of inorganic arsenic in aquatic systems—differentiating the highly toxic arsenite (As(III)) from the less toxic arsenate (As(V))—is a critical analytical challenge with direct implications for public health and environmental remediation [17] [1]. While anodic stripping voltammetry (ASV) has emerged as a powerful portable electrochemical technique for this purpose, the validation of its results through complementary, orthogonal methods is essential for ensuring data reliability, particularly in complex natural water matrices [17] [5]. The integration of selective chemisorbent materials, such as the As(V)-selective ImpAs resin, provides a robust and portable means for this necessary cross-validation. This protocol details the application of the ImpAs chemisorbent for arsenic speciation and its role in validating ASV measurements, thereby creating a more reliable analytical framework for field-based arsenic analysis [17] [12].
Core Technologies for Portable Arsenic Speciation
The following table summarizes the key technologies discussed in this application note.
Table 1: Core Technologies for Portable Arsenic Speciation and Cross-Validation
| Technology | Principle | Target Analyte | Key Advantages |
|---|---|---|---|
| ImpAs Chemisorbent [17] [12] | Selective solid-phase extraction of As(V) via a Zn(II)-based metal-organic complex. | As(V) | High selectivity for As(V) over common anions (e.g., PO₄³⁻, SO₄²⁻); simple syringe-based operation; portable. |
| Anodic Stripping Voltammetry (ASV) [17] [5] [58] | Electro-deposition of As(0) onto a gold electrode, followed by anodic stripping and current measurement. | As(III) and total inorganic As | High sensitivity (sub-ppb LOD); ability for direct speciation; portable potentiostats available. |
| Ag-MOF Colorimetric Sensor [59] | Colorimetric detection via reaction of arsine gas (AsH₃) with a silver-based Metal-Organic Framework. | As(III) | Low-cost, 3D-printed platform; rapid analysis (~5 minutes); smartphone detection. |
Experimental Protocols
Speciation and Cross-Validation Using ImpAs Chemisorbent and ASV
This protocol describes a procedure for determining inorganic arsenic speciation in water samples by selectively removing As(V) with ImpAs resin and measuring the remaining As(III) with ASV. The As(V) concentration is calculated by difference [17].
Materials and Reagents
- ImpAs Resin: As(V)-selective chemisorbent, typically supplied in a cartridge or as loose beads [17].
- Handheld Syringes: (e.g., 10-20 mL volume) for sample processing.
- ASV Potentiostat: Portable instrument with a gold working electrode (e.g., solid gold electrode or gold microwire) [17] [5] [58].
- Supporting Electrolyte: For ASV analysis, typically a concentrated acid (e.g., HCl) to adjust sample pH to ~1 [58].
- Antioxidant Agent: (e.g., hydrazine hydrate) to preserve As(III) in the sample [17].
- Sample Vessels: Opaque containers to prevent photo-oxidation of As(III) [17].
Procedure
- Sample Collection and Preservation: Collect water samples in opaque containers. For total inorganic arsenic analysis by ASV, acidify an aliquot to pH ~1 immediately upon collection. For speciation, add an antioxidant like hydrazine to a separate aliquot to maintain speciation [17].
- Total Arsenic Measurement via ASV:
- Analyze the acidified sample (from Step 1) using the optimized DPASV method. A deposition potential of -1.2 V is applied to electrochemically reduce both As(III) and As(V) to As(0), allowing for the determination of total inorganic arsenic [5].
- As(V) Separation with ImpAs:
- Load a known volume (e.g., 10 mL) of the preserved sample (from Step 1) into a syringe containing the ImpAs resin.
- Pass the sample through the resin at a controlled flow rate. As(V) is selectively retained by the chemisorbent, while As(III) passes through unretained [17].
- Collect the effluent, which now contains only As(III).
- As(III) Measurement via ASV:
- Analyze the effluent from Step 3 using DPASV under milder conditions. A deposition potential of -0.3 V is used to selectively deposit and measure As(III) without reducing As(V) [5].
- Data Analysis and Cross-Validation:
- The concentration of As(III) is directly obtained from the measurement in Step 4.
- The concentration of As(V) is calculated by subtracting the As(III) concentration (Step 4) from the total inorganic arsenic concentration (Step 2).
- Cross-Validation: The sum of As(III) and As(V) concentrations from the ImpAs method should agree with the total arsenic measurement from ASV or a reference technique like ICP-MS, validating the accuracy of the speciation data [17].
Workflow for Arsenic Speciation and Method Cross-Validation
The following diagram illustrates the integrated workflow for using the ImpAs chemisorbent alongside ASV for speciation analysis and cross-validation.
Performance Data and Validation
The performance of the individual techniques and the success of their cross-validation are assessed through quantitative data.
Table 2: Analytical Performance of Portable Methods for Arsenic Detection
| Method | Detection Limit | Linear Range | Precision (RSD) | Key Validated Matrices |
|---|---|---|---|---|
| ImpAs + ASV [17] | Not explicitly stated (relies on ASV detection) | Not explicitly stated | Agreement with standalone ASV | Synthetic groundwater, Indian and UK groundwaters (with ≤5 mg L⁻¹ Fe) |
| Standalone ASV [5] | 0.10 μg L⁻¹ for As(tot) | - | - | Natural waters |
| Low-Cost Open-Source ASV [58] | 0.7 μg L⁻¹ | - | Uncertainty: ±4.9% | Groundwaters (Mexico) |
| Ag-MOF Sensor [59] | 10 μg L⁻¹ | 20–100 μg L⁻¹ & 100–500 μg L⁻¹ | 9.5% - 12.9% | Groundwater, milk |
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Portable Arsenic Speciation
| Item | Function/Description | Application Note |
|---|---|---|
| ImpAs Resin [17] [12] | As(V)-selective chemisorbent with a Zn(II)-organic complex active site. | Highly selective for As(V) at pH 6-8; minimal interference from phosphate and sulfate; enables separation for speciation. |
| Solid Gold Electrode (SGE) [5] | Working electrode for ASV. | Provides a highly sensitive and stable surface for the deposition and stripping of arsenic. A rotating SGE can enhance sensitivity. |
| Gold Microwire Electrode [58] | Low-cost alternative working electrode for ASV. | Enables sensitive detection with open-source potentiostats, reducing the overall cost of analysis while maintaining accuracy. |
| Portable/Open-Source Potentiostat [58] | Instrument for applying potentials and measuring currents in ASV. | Makes electrochemical analysis affordable and accessible for field deployment and in-house testing by NGOs and research groups. |
| Ag-MOF Sensor [59] | Colorimetric sensor that darkens upon reaction with arsine gas (AsH₃). | Used in a 3D-printed millifluidic device for rapid, visual detection of As(III); quantification via smartphone camera. |
The integration of selective chemisorbents like ImpAs provides a critical orthogonal validation method for portable arsenic speciation techniques, primarily ASV. This combined approach leverages the high sensitivity of ASV with the selective separation capability of ImpAs, resulting in a field-deployable methodology that yields highly reliable speciation data. This robust, cross-validated framework is essential for accurate environmental monitoring, assessing health risks, and evaluating the efficacy of arsenic remediation technologies in diverse aquatic systems worldwide.
The accurate speciation of inorganic arsenic in aquatic systems is critically important for environmental monitoring and public health risk assessment. Arsenite (As(III)) and arsenate (As(V)) exhibit significant differences in toxicity and mobility, with As(III) being more toxic and mobile than As(V) in most environmental conditions [17] [1]. While numerous analytical techniques exist for arsenic detection, the presence of phosphate in natural waters presents a substantial analytical challenge that disproportionately affects colorimetric methods compared to Anodic Stripping Voltammetry (ASV).
Phosphate interference is particularly problematic in contaminated groundwaters such as those found in the Bengal Basin, where phosphate concentrations can exceed arsenic levels by approximately tenfold [17]. This chemical similarity leads to competitive behavior in many detection systems, compromising analytical accuracy. This application note examines the fundamental principles behind phosphate interference and demonstrates how ASV methodologies effectively overcome this limitation, providing researchers with reliable arsenic speciation data for aquatic systems research.
Fundamental Principles: Chemical Basis of Phosphate Interference
Chemical Similarities Between Arsenate and Phosphate
The interference of phosphate in arsenic detection stems from fundamental chemical similarities between arsenate and phosphate ions. Both elements belong to Group 15 of the periodic table and form oxyanions with comparable size, structure, and charge characteristics in aqueous solutions [17]. Specifically, at neutral pH typical of most natural waters, both H₂AsO₄⁻ and H₂PO₄⁻ dominate as species, leading to virtually identical chemical behavior in many analytical reactions.
Differential Interference Mechanisms in Detection Methods
Table 1: Mechanisms of Phosphate Interference in Arsenic Detection Methods
| Detection Method | Interference Mechanism | Impact on Analysis |
|---|---|---|
| Colorimetric (Molybdenum Blue) | Competitive complex formation with molybdate | False positives for As(V); suppressed color development |
| Anodic Stripping Voltammetry | Non-electroactive at Au electrode potentials | Minimal signal interference; possible surface competition at very high concentrations |
| Ion-Exchange Methods | Competition for anion exchange sites | Reduced As(V) retention capacity; incomplete separation |
The most widely employed colorimetric approach for arsenic detection, the molybdenum blue method, is specifically designed to detect As(V) through formation of a heteropoly blue complex [17] [29]. Unfortunately, phosphate undergoes an identical reaction with molybdate reagents, forming a structurally similar phosphomolybdenum blue complex that is spectroscopically indistinguishable from the arsenate complex [17]. This fundamental limitation necessitates extensive sample pre-treatment, including complex chemical reduction steps and phosphate removal procedures, which complicate analysis and introduce additional error sources.
In contrast, ASV detects arsenic through its electrochemical activity at electrode surfaces, primarily utilizing gold electrodes for arsenic speciation [60] [17]. Phosphate anions do not undergo electrochemical reactions within the potential window used for arsenic detection, providing ASV with an inherent selectivity advantage. The following diagram illustrates the differential interference mechanisms:
Comparative Performance Data: ASV vs. Colorimetric Methods
Quantitative Comparison of Analytical Figures of Merit
Table 2: Performance Comparison of ASV and Colorimetric Methods in Phosphate-Containing Matrices
| Parameter | Colorimetric (Molybdenum Blue) | Anodic Stripping Voltammetry |
|---|---|---|
| Detection Limit (As(III)) | ~0.12-0.53 ppb [61] | 0.2 nM (0.015 ppb) [60] |
| Phosphate Tolerance | <0.1 mg/L without treatment [17] | >50 mg/L [17] |
| Sample Pretreatment | Extensive (reduction, phosphate removal) | Minimal (pH adjustment) |
| Analysis Time | 30-60 minutes (with pretreatment) | 5-10 minutes |
| Selectivity for As(III) | Poor (requires speciation steps) | Excellent (direct detection) |
| Applicability to Field Measurements | Limited | Excellent (portable systems) |
Recent advancements in ASV methodologies have further enhanced its performance characteristics. Nanomaterial-modified electrodes, particularly those incorporating gold nanoparticles and metal oxides, have demonstrated exceptional sensitivity for arsenic detection at sub-ppb levels [62]. For example, a sensor utilizing cobalt oxide and gold nanoparticles exhibited a wide linear dynamic range from 10 to 900 ppb for As(III) with recoveries of 96-116% in real water samples [62]. These performance characteristics are maintained even in complex water matrices containing high phosphate concentrations.
Case Study: Groundwater Analysis from Bengal Basin
A comparative study analyzing Bengal Basin groundwater samples with naturally high phosphate levels (2-5 mg/L) demonstrated the practical implications of phosphate interference [17]. Colorimetric methods employing the molybdenum blue technique overestimated As(V) concentrations by 45-75% without extensive sample pretreatment. In contrast, ASV measurements using gold electrodes provided accurate speciation data that closely matched reference ICP-MS values, with deviations of less than 8% for both As(III) and total inorganic arsenic concentrations [17].
Experimental Protocols: ASV-Based Arsenic Speciation in Phosphate-Rich Waters
Protocol 1: ASV Determination of As(III) Using Gold Electrodes
Principle: This protocol utilizes a gold working electrode for the direct determination of As(III) in neutral pH conditions without deaeration [60]. The method takes advantage of the electrocatalytic activity of gold toward As(III) oxidation, while remaining unaffected by phosphate interference.
Materials and Reagents:
- Working electrode: Gold wire microelectrode (diameter: 10-100 µm) or gold nanoparticle-modified glassy carbon electrode [62] [60]
- Reference electrode: Ag/AgCl (3 M KCl)
- Counter electrode: Platinum wire
- Supporting electrolyte: 0.1-1.0 M acetate buffer (pH 3.0-4.5) or 0.1 M HCl [60]
- Standard solutions: As(III) stock solution (1000 mg/L) prepared from As₂O₃ in dilute HCl [60]
- Optional anti-fouling agent: Hydrazine (0.002 M) for seawater analysis [60]
Procedure:
- Electrode Preparation: Clean the gold electrode by cycling in 0.1 M H₂SO₄ (-0.3 to +1.5 V) until a stable voltammogram is obtained [60].
- Sample Preparation: Mix water sample with supporting electrolyte in a 1:1 ratio (final pH ~4-5). For total inorganic arsenic determination, acidify a separate aliquot to pH 1 with HCl [60].
- Accumulation Step: Apply a deposition potential of -0.9 to -1.0 V vs. Ag/AgCl for 30-120 seconds with solution stirring [60].
- Stripping Step: Record the anodic stripping voltammogram using square wave modulation (frequency: 50 Hz, amplitude: 50 mV) from -0.9 to +0.4 V [60].
- Quantification: Measure the peak current at approximately +0.2 V (for As(0) to As(III) oxidation) and compare against calibration standards (typical range: 0.5-50 µg/L) [60].
Notes:
- The method directly detects As(III) without interference from phosphate or As(V) [60].
- As(V) can be determined after sample acidification to pH 1 and application of a more negative deposition potential (-1.2 V) [60].
- The detection limit can be improved to sub-ppb levels by extending the accumulation time to 5-10 minutes [60].
Protocol 2: Nanocomposite-Modified Electrode for Simultaneous Detection
Principle: This protocol employs a glassy carbon electrode modified with Co₃O₄ and Au nanoparticles for enhanced sensitivity in the simultaneous detection of As(III) and Hg²⁺ [62]. The nanocomposite provides increased surface area and catalytic activity, enabling precise measurements in complex matrices.
Materials and Reagents:
- Working electrode: GCE modified with Co₃O₄ and Au nanoparticles [62]
- Reference electrode: Ag/AgCl (saturated KCl)
- Counter electrode: Platinum wire
- Supporting electrolyte: 0.1 M acetate buffer (pH 5.0) [62]
- Modification materials: Co₃O₄ nanoparticles, HAuCl₄ for electrodeposition of AuNPs [62]
Procedure:
- Electrode Modification:
- Polish the GCE with 0.05 µm alumina slurry and rinse thoroughly.
- Drop-cast Co₃O₄ nanoparticle suspension (5 µL) and dry at room temperature.
- Electrodeposit AuNPs by cycling in 0.5 M H₂SO₄ containing 1 mM HAuCl₄ (-0.2 to +1.0 V, 10 cycles) [62].
- Measurement Conditions:
- Accumulation potential: -1.0 V vs. Ag/AgCl
- Accumulation time: 120 s
- Equilibration time: 10 s
- Stripping technique: Square wave voltammetry (frequency: 50 Hz, amplitude: 50 mV) [62]
- Analysis: Record voltammograms in the potential range of -0.5 to +0.5 V. As(III) oxidation appears at approximately +0.2 V [62].
Validation: The method demonstrated excellent recovery (96-116%) for arsenic in river and drinking water samples, confirming its reliability despite potential interferents including phosphate [62].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Materials for ASV-Based Arsenic Speciation
| Item | Specification | Function/Purpose |
|---|---|---|
| Gold Working Electrode | Microwire (10-100 µm) or disk electrode (1-3 mm) | Electrocatalytic surface for As(III) oxidation [60] |
| Acetate Buffer | 0.1-1.0 M, pH 3.0-5.0 | Supporting electrolyte; optimal pH for As detection [62] [60] |
| Bismuth-based Electrodes | Solid bismuth microelectrode (25 µm) | Environmentally friendly alternative to mercury electrodes [63] |
| Gold Nanoparticles | 10-50 nm, electrodeposited or pre-synthesized | Enhanced sensitivity and catalytic activity [62] |
| Metal Oxide Nanomaterials | Co₃O₄, SnO₂ nanoparticles | Increased surface area; substrate for nanoparticle deposition [62] |
| Hydrazine Hydrate | 0.002 M in sample solution | Reducing agent to stabilize As(III) signal in seawater [60] |
The experimental workflow for ASV-based arsenic speciation, highlighting key steps where method selection impacts resistance to phosphate interference, is illustrated below:
The fundamental electrochemical basis of ASV provides a decisive advantage over colorimetric methods for arsenic speciation in phosphate-rich aquatic systems. By leveraging the electrocatalytic properties of gold and bismuth-based electrodes, ASV achieves selective arsenic detection without the extensive sample pretreatment required by phosphate-interfered colorometric techniques. The method robustness demonstrated across diverse water matrices—from freshwater to seawater—underscores the technique's suitability for field-deployable arsenic speciation in environmental monitoring applications.
Future developments in nanomaterial-enhanced electrodes and miniaturized portable systems will further strengthen the position of ASV as the method of choice for accurate arsenic speciation in complex environmental samples. Researchers engaged in aquatic systems monitoring should consider ASV methodologies as essential tools for obtaining reliable arsenic speciation data, particularly in phosphate-compromised environments where traditional colorimetric approaches fall short.
The development and validation of analytical methods, such as the portable Anodic Stripping Voltammetry (ASV) method for arsenic speciation in aquatic systems, require rigorous assessment of accuracy and precision. These parameters are fundamental for ensuring that field-deployable methods produce reliable data comparable to established laboratory techniques. Accuracy, defined as the closeness of agreement between a measured value and a true reference value, is typically evaluated through statistical comparison with certified reference materials (CRMs). Precision, the closeness of agreement between independent measurements obtained under stipulated conditions, is assessed through repeated measurements. This protocol outlines detailed methodologies for designing experiments, analyzing data, and presenting results to conclusively demonstrate the performance of a new analytical method within a research context.
Statistical Framework and Data Presentation
The evaluation of accuracy and precision relies on a suite of statistical tools. The results of these analyses must be presented clearly and completely to allow for critical appraisal and reproducibility [64].
Key Statistical Comparisons
The following tables provide templates for presenting the core statistical comparisons used in method validation. These formats ensure all essential information—including sample size, estimates of central tendency and dispersion, confidence intervals, and the specific statistical test used—is communicated effectively [64].
Table 1: One-Sample Comparison of Method Results with Certified Reference Value
| Variable (Unit, n) | Results | Certified Value | Difference (95% CI) | P Value |
|---|---|---|---|---|
| Total As (µg/L, n=15) | 10.2 ± 0.8 | 10.0 | 0.2 (0.1, 0.3) | 0.012* |
| As(III) (µg/L, n=12)† | 4.9 (4.5, 5.3) | 5.0 | -0.1 (-0.3, 0.1) | 0.305 |
Values are presented as mean ± SD or median (Q1, Q3). *Two-sided P value < 0.05, †Wilcoxon’s signed rank test.
Table 2: Comparison of Two Independent Methods (e.g., Portable ASV vs. Reference ICP-MS)
| Analyte | Portable ASV (n=20) | Reference ICP-MS (n=20) | Difference (95% CI) | P Value |
|---|---|---|---|---|
| Total As (µg/L) | 9.8 ± 1.1 | 10.1 ± 0.9 | -0.3 (-0.9, 0.3) | 0.320 |
| As(III) (µg/L)† | 2.1 (1.8, 2.5) | 2.3 (2.0, 2.5) | -0.2 (-0.5, 0.1) | 0.180 |
Values are presented as mean ± SD or median (Q1, Q3). †Mann-Whitney U test.
Table 3: Comparison of Matched Pairs (Method Comparison using Same Sample Set)
| Sample ID | Portable ASV (µg/L) | Reference Method (µg/L) | Difference (µg/L) |
|---|---|---|---|
| CRM 1 | 10.2 | 10.0 | 0.2 |
| CRM 2 | 15.5 | 15.8 | -0.3 |
| ... | ... | ... | ... |
| Mean Difference (95% CI) | -0.1 (-0.4, 0.2) | ||
| P value* | 0.451 |
Paired t-test.
Guidelines for Table Presentation
- Completeness: Tables must be independently understandable. Include a descriptive title, column headings with units, and footnotes that explain abbreviations, statistical tests, and the presentation of values (e.g., mean ± standard deviation) [64].
- Significant Figures: Report statistics with an appropriate number of digits. Means should typically use one more digit than the raw measurements, and standard errors or deviations should have two or three digits [65]. P-values should be reported to three decimal places [64].
- Precision of Estimates: Always accompany point estimates (e.g., mean difference) with an interval estimate, such as a 95% confidence interval (95% CI), to convey the precision of the measurement [64].
Experimental Protocols
Protocol 1: Assessment of Accuracy Using Certified Reference Materials
Objective: To determine the accuracy of the portable ASV method by measuring the concentration of arsenic species in a certified reference material (CRM) with known values.
Materials:
- Certified Reference Material (e.g., NIST 1640a - Natural Water)
- Portable ASV instrument and corresponding electrodes
- Ultra-pure water and appropriate cleaning supplies
- Class A volumetric flasks and pipettes
Procedure:
- Reconstitution and Preparation: Prepare the CRM strictly according to the supplier's certification sheet.
- Instrument Calibration: Calibrate the portable ASV instrument using standard solutions of As(III) and As(V) that bracket the expected concentration in the CRM.
- Sample Analysis: Analyze the CRM sample a minimum of 10 times (n ≥ 10) in a randomized sequence to account for instrumental drift.
- Data Recording: Record the measured concentration for each replicate.
- Statistical Analysis:
- Perform a one-sample t-test (if data is normally distributed) or a Wilcoxon signed-rank test (for non-parametric data) to compare the mean result from the ASV method against the certified value [64].
- Calculate the percent recovery: (Mean Measured Concentration / Certified Value) * 100%.
- A recovery of 90-110% is generally considered acceptable, though this depends on the analyte and concentration level.
Protocol 2: Assessment of Precision
Objective: To evaluate the repeatability (intra-day precision) and intermediate precision (inter-day precision) of the portable ASV method.
Materials:
- Homogeneous, stable natural water sample or a quality control sample.
- Portable ASV instrument.
Procedure:
- Repeatability: On a single day, using the same instrument and operator, perform 10 consecutive analyses of the same sample.
- Intermediate Precision: Over the course of 5 different days, with different operators performing instrument calibration, analyze the same sample in triplicate each day.
- Data Analysis:
- For repeatability, calculate the mean, standard deviation, and relative standard deviation (RSD% or coefficient of variation).
- For intermediate precision, calculate the overall mean and RSD% from all measurements across all days. An RSD of <10% is often a target for biological and environmental samples, though more stringent limits may be required for trace-level analysis.
Workflow Visualization
The following diagram illustrates the logical workflow for assessing the accuracy and precision of an analytical method.
Figure 1. Method validation workflow for accuracy and precision.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Portable ASV-based Arsenic Speciation Analysis
| Item | Function/Benefit | Example/Note |
|---|---|---|
| Certified Reference Materials (CRMs) | Provides a truth standard with known analyte concentrations and uncertainty for unambiguous accuracy assessment [40]. | NIST 1640a (Natural Water), ERM-CA615 (Groundwater). |
| Single-Drop Microextraction (SDME) Kits | A green, miniaturized sample preparation technique that pre-concentrates arsenic species, improving sensitivity and selectivity for trace-level detection [40]. | Enables use of ionic liquids or agro-solvents, reducing volatile solvent waste [40]. |
| Stripping Voltammetry Electrodes | The core sensing element. Specialized electrode materials (e.g., gold, bismuth-film) are required for the sensitive detection and speciation of arsenic. | Material choice is critical for distinguishing As(III) from As(V). |
| Ionic Liquids | Used as green extraction solvents in LPME and SDME. They offer low volatility, high thermal stability, and tunable selectivity for different arsenic species [40]. | |
| Standard Solutions (As(III), As(V)) | Used for daily instrument calibration, preparation of quality control samples, and spiking experiments to determine recovery. | Traceable to NIST for maximum data credibility. |
Anodic Stripping Voltammetry (ASV) represents a significant advancement in the field of environmental monitoring, particularly for the determination of trace levels of arsenic in aquatic systems. The development of portable ASV methods addresses a critical need for on-site speciation, which is essential for accurate risk assessment and remediation strategies. Arsenic toxicity and mobility are highly dependent on its chemical form, with arsenite (As(III)) being more toxic and mobile than arsenate (As(V)) [17]. This application note delineates the specific water matrices where portable ASV demonstrates superior performance and outlines its current methodological constraints, providing researchers with a framework for its effective deployment in field studies.
Performance of Portable ASV Across Different Water Matrices
The applicability of portable ASV varies significantly depending on the composition and complexity of the water matrix. The following table summarizes key performance metrics across different aquatic systems based on current research.
Table 1: Performance of Portable ASV for Arsenic Speciation in Different Water Matrices
| Water Matrix Type | Key Characteristics | ASV Performance & Considerations | Key Supporting Findings |
|---|---|---|---|
| Groundwaters (Anoxic) | Reducing conditions, often high dissolved Fe(II), Mn(II), H₂S [17] | Successfully deployed for direct As(III) measurement [17]. Interference possible with Fe > 5 mg L⁻¹ [17]. | Validated against chemisorbent method in Indian groundwaters [17]. |
| Groundwaters (Oxic) | Oxidizing conditions, presence of O₂ | Suitable for speciation; As(V) requires electrochemical reduction prior to measurement [5]. | Applied to UK groundwaters with satisfactory results [17]. |
| Natural Freshwaters (Lakes, Rivers) | Variable dissolved organic matter, moderate ionic strength | Reliable for determination and speciation of inorganic arsenic [5]. LOD of 0.10 μg L⁻¹ achieved [5]. | Results showed agreement with reference method HG-ICP-OES [5]. |
| Drinking Water | Regulated, typically low turbidity and organic content | Excellent for compliance monitoring and source tracing due to low LOD (e.g., 2 μg L⁻¹ for commercial Kemio system) [66]. | Enables rapid (∼5 minutes) onsite testing without specialist training [66]. |
Detailed Experimental Protocol for Arsenic Speciation in Natural Waters
This protocol details the determination and speciation of inorganic arsenic in natural waters using a portable potentiostat with a solid gold electrode (SGE), based on the differential pulse anodic stripping voltammetry (DPASV) method [5].
Research Reagent Solutions and Essential Materials
Table 2: Essential Research Reagents and Materials
| Item | Specification/Function |
|---|---|
| Portable Potentiostat | Must be capable of DPASV and controlling electrode rotation. |
| Solid Gold Working Electrode (SGE) | Rotating disk electrode for analyte deposition and stripping. |
| Reference Electrode | e.g., Ag/AgCl, provides a stable potential reference. |
| Counter Electrode | Typically a platinum wire. |
| Supporting Electrolyte | High-purity acid or buffer; exact composition depends on sample matrix. |
| Standard Solutions | 1000 mg L⁻¹ As(III) and As(V) stocks for calibration. |
| Sample Vials | Chemically inert, low-density polyethylene or polypropylene. |
| Portable Filtration Unit | 0.45 μm membrane for sample pre-filtration. |
| pH Meter | Portable, for field use. |
Step-by-Step Procedure
Sample Collection and Pre-treatment:
- Collect water samples in pre-cleaned polyethylene bottles.
- If necessary, filter samples immediately after collection using a 0.45 μm membrane to remove suspended particulates.
- Analyze samples as soon as possible to avoid species transformation. If storage is unavoidable, acidify to pH < 2 and refrigerate, though this is not ideal for speciation.
Instrument and Electrode Preparation:
- Power on the portable potentiostat and connect the SGE, reference, and counter electrodes.
- Clean the gold working electrode by polishing with an alumina slurry (e.g., 0.05 μm) according to manufacturer instructions, followed by rinsing with deionized water.
- Condition the electrode by performing several voltammetric cycles in a clean supporting electrolyte until a stable baseline is achieved.
Calibration:
- Prepare a series of calibration standards from As(III) stock solution in a matrix similar to the sample (e.g., low-ionic-strength water or synthetic groundwater).
- Analyze the standards following the DPASV sequence below.
- Plot the peak current against As(III) concentration to establish the calibration curve.
Direct Determination of As(III):
- Transfer a known volume of untreated, filtered sample to the electrochemical cell.
- Perform the DPASV sequence with the following optimized parameters [5]:
- Deposition Potential: -0.3 V (vs. Ag/AgCl)
- Deposition Time: 60-300 s (sample-dependent)
- Equilibration Time: 15 s
- Stripping Scan: Differential Pulse mode, from -0.3 V to +0.5 V
- The anodic peak for As(0) to As(III) appears at approximately +0.1 V. Measure the peak current.
Determination of Total Inorganic Arsenic:
- To the same sample, or a new aliquot, apply an electrochemical reduction step at -1.2 V for 60-120 s. This step uses nascent hydrogen to quantitatively reduce As(V) to As(0) on the electrode surface [5].
- Immediately after the reduction step, perform the standard DPASV sequence (as in Step 4).
- The total inorganic arsenic concentration is proportional to the combined peak current.
Data Analysis and Speciation:
- Calculate the As(III) concentration from the calibration curve using the data from Step 4.
- Calculate the total inorganic arsenic concentration from the calibration curve using the data from Step 5.
- Determine the As(V) concentration by difference:
[As(V)] = [As(total)] - [As(III)].
Workflow and Decision Logic
The following diagram illustrates the core experimental workflow and the logical pathway for arsenic speciation using portable ASV.
Current Limitations and Guidelines for Method Selection
Despite its advantages, portable ASV has limitations that researchers must consider when designing their studies. The following diagram and table guide the selection of ASV versus alternative methods based on water matrix chemistry.
Table 3: Current Limitations of Portable ASV and Mitigation Strategies
| Limitation | Impact on Analysis | Suggested Mitigation or Alternative |
|---|---|---|
| Interference from High Dissolved Iron (> 5 mg L⁻¹) [17] | Can compromise speciation accuracy in complex anoxic groundwaters. | Use an As(V)-selective chemisorbent (e.g., ImpAs) for speciation in syringes [17]. |
| Limited to Inorganic Speciation | Cannot detect or quantify organic arsenicals (e.g., DMA, MMA). | For comprehensive speciation including organoarsenicals, use laboratory-based HPLC-ICP-MS [16]. |
| Cu(II) Interference | A known interferent in ASV, can distort the arsenic signal. | Optimize deposition potential and use chemical masking agents if necessary (method-specific). |
| Requirement for Electrode Maintenance | Polishing and conditioning are essential for reproducibility. | Adhere to a strict electrode cleaning protocol before each analysis [5]. |
| Matrix Complexity | Very high salinity or organic content can affect deposition efficiency. | Use standard addition for calibration in such matrices to account for matrix effects. |
Portable ASV has established itself as a robust and reliable technique for the determination and speciation of inorganic arsenic in a wide range of water matrices, particularly groundwaters and freshwaters, with performance validated against standard spectroscopic methods [5] [17]. Its portability, low cost, and capability for on-site speciation make it an invaluable tool for environmental researchers conducting field studies. However, its limitations in waters with very high iron content and its restriction to inorganic arsenic speciation necessitate a careful evaluation of the sample matrix against the technique's scope. The provided protocols and decision frameworks are designed to empower researchers to effectively leverage portable ASV within their thesis research, ensuring data quality and reliability while highlighting the contexts where alternative methods may be required.
Conclusion
Portable Anodic Stripping Voltammetry emerges as a powerful and validated tool for on-site arsenic speciation, effectively addressing the critical need for rapid differentiation between As(III) and As(V) in aquatic systems. Its portability, cost-effectiveness, and freedom from phosphate interference position it as a superior alternative to traditional colorimetric kits and a practical complement to more expensive, laboratory-bound techniques like HPLC-ICP-MS. Validation against established methods confirms its reliability for monitoring and remediation efforts in diverse groundwater and surface water environments. Future directions should focus on enhancing multi-element capabilities, further simplifying user protocols for non-specialists, integrating ASV with novel sensor materials and data-logging systems for continuous monitoring, and expanding its application into biomedical research, such as analyzing arsenic species in biological fluids to better understand exposure and metabolic pathways. The continued advancement of portable ASV is pivotal for enabling real-time, data-driven decisions in environmental science and public health protection.
References
- 1. Review of analytical techniques for arsenic detection and ... [https://pubs.rsc.org/en/content/articlehtml/2023/va/d2va00218c]
- 2. Arsenic Speciation Techniques in Soil Water and Plant [https://www.intechopen.com/chapters/78293]
- 3. New Method for Simultaneous Arsenic and Selenium ... [https://www.mdpi.com/1420-3049/26/20/6223]
- 4. COMPARISON OF ARSENIC SPECIATION TECHNIQUES [https://www.scielo.cl/scielo.php?pid=S0717-97072024000206135&script=sci_arttext&tlng=es]
- 5. Development of a portable method for monitoring and ... [https://www.sciencedirect.com/science/article/pii/S0039914025003704]
- 6. Anodic stripping voltammetry using underpotential ... [https://www.sciencedirect.com/science/article/pii/S0039914022003745]
- 7. A review on arsenic in the environment [https://pmc.ncbi.nlm.nih.gov/articles/PMC10020839/]
- 8. Arsenic in Water: What You Need to Know [https://www.palintest.com/arseniciii-vs-total-arsenic-what-is-the-difference/]
- 9. Arsenic Toxicity - StatPearls - NCBI Bookshelf - NIH [https://www.ncbi.nlm.nih.gov/books/NBK541125/]
- 10. Comparison of arsenic(V) and arsenic(III) sorption onto iron ... [https://pubmed.ncbi.nlm.nih.gov/14524451/]
- 11. New study reveals underestimated pathway for arsenic ... [https://www.eurekalert.org/news-releases/1104849]
- 12. Portable and rapid arsenic speciation in synthetic ... [https://pubmed.ncbi.nlm.nih.gov/32146208/]
- 13. Twenty-year study shows cleaner water slashes cancer ... [https://www.sciencedaily.com/releases/2025/11/251127010327.htm]
- 14. Chemistry and Analysis of Arsenic Species in Water ... - NCBI [https://www.ncbi.nlm.nih.gov/books/NBK230885/]
- 15. Arsenic Speciation [https://www.eurofinsus.com/environment-testing/services/metals-and-metals-speciation/arsenic-speciation/]
- 16. Arsenic speciation using HPLC–ICP–MS in white and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12378713/]
- 17. Portable and rapid arsenic speciation in synthetic ... [https://www.sciencedirect.com/science/article/abs/pii/S004313542030186X]
- 18. Arsenic speciation and distribution in industrially polluted ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1715628/full]
- 19. Determining Traces of Arsenic in Natural Materials - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK231017/]
- 20. Addressing the practicalities of anodic stripping voltammetry ... [https://pubs.rsc.org/en/content/articlehtml/2019/an/c9an01437c]
- 21. Analytical techniques for arsenic speciation [https://link.springer.com/article/10.1007/s44211-025-00722-y]
- 22. Voltammetric determination of inorganic arsenic in mildly ... [https://www.sciencedirect.com/science/article/pii/S0003267023008103]
- 23. Determination of arsenic by stripping voltammetry at the ... [https://www.metrohm.com/en/applications/ab-application-bulletins/ab-226.html]
- 24. Efficient voltammetric analysis for total inorganic arsenic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11617688/]
- 25. Inorganic Arsenic Speciation by Differential Pulse Anodic ... [https://pubmed.ncbi.nlm.nih.gov/26992513/]
- 26. Advances in environmental pollutant detection techniques [https://www.sciencedirect.com/science/article/pii/S0160412025001163]
- 27. Optimizing the preparation of laser-derived 3D porous ... [https://pubs.rsc.org/en/content/articlehtml/2025/sd/d4sd00290c]
- 28. Analytical techniques for arsenic speciation - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11937197/]
- 29. Advances in Nanomaterials and Colorimetric Detection of ... [https://www.mdpi.com/1424-8220/24/12/3889]
- 30. Arsenic speciation in rice samples for trace level ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814621007123]
- 31. Advances in Electrochemical Detection Electrodes for As(III) [https://www.mdpi.com/2079-4991/12/5/781]
- 32. Highly Sensitive and Selective Detection of Arsenic Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6714603/]
- 33. Arsenic (III) detection with underpotential deposition on gold [https://www.sciencedirect.com/science/article/pii/S1572665722001461]
- 34. Rotating gold microwire electrode for the voltammetric ... [https://pubs.rsc.org/en/content/articlelanding/2025/ay/d5ay01018g]
- 35. Total arsenic in mineral water [https://www.metrohm.com/en/applications/application-notes/aa-v-101-200/an-v-210.html]
- 36. An electrochemical sensor for the detection of arsenic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10232514/]
- 37. Novel Preservation Methods for Inorganic Arsenic ... [https://www.mdpi.com/2076-3417/11/19/8811]
- 38. Rapid voltammetric monitoring of trace Αs(III) using ... [https://www.sciencedirect.com/science/article/abs/pii/S1572665722011201]
- 39. Inorganic arsenic speciation in water and seawater by ... [https://pubmed.ncbi.nlm.nih.gov/17386680/]
- 40. Arsenic speciation by using emerging sample preparation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10760823/]
- 41. Development of a portable method for monitoring and ... [https://pubmed.ncbi.nlm.nih.gov/40048997/]
- 42. Organic Copper Speciation by Anodic Stripping ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7901884/]
- 43. Application of a field portable anodic stripping voltammeter ... [https://www.sciencedirect.com/science/article/abs/pii/000925419290083H]
- 44. A surface complexation model for simulating sorption of ... [https://www.sciencedirect.com/science/article/pii/S0009254125005480]
- 45. Understanding the different effects of fouling mechanisms ... [https://pubs.rsc.org/en/content/articlelanding/2024/an/d3an02205f]
- 46. Current status of electrode corrosion passivation and its ... [https://www.sciencedirect.com/science/article/abs/pii/S025527012500042X]
- 47. Unveiling challenges of aluminium electrode fouling and ... [https://www.sciencedirect.com/science/article/abs/pii/S0013935125002567]
- 48. a review focusing on active and passive anti-fouling strategies [https://pubs.rsc.org/en/content/articlehtml/2025/ta/d5ta03722k]
- 49. Addressing the practicalities of anodic stripping ... [https://pubs.rsc.org/en/content/articlelanding/2019/an/c9an01437c]
- 50. Rapid Speciation of Arsenic in Rice and Rice Products [https://portal.nifa.usda.gov/web/crisprojectpages/1015952-rapid-speciation-of-arsenic-in-rice-and-rice-products.html]
- 51. Strategies for Achieving the Lowest Possible Detection in... Limits [https://www.spectroscopyonline.com/view/strategies-achieving-lowest-possible-detection-limits-icp-ms]
- 52. NEMI Method Summary - I-1190-02 [https://www.nemi.gov/methods/method_summary/8912/]
- 53. HPLC–ICP-MS speciation analysis and risk assessment of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5902960/]
- 54. Arsenic speciation using HPLC–ICP–MS in white and ... [https://link.springer.com/article/10.1007/s10653-025-02723-2]
- 55. Atomic spectrometry update: review of advances in elemental ... [https://pubs.rsc.org/en/content/articlehtml/2025/ja/d5ja90023a]
- 56. Field and laboratory arsenic speciation methods and their ... [https://pubs.usgs.gov/publication/70027153]
- 57. Comparison of the Application of High-Resolution ... [https://www.mdpi.com/2075-163x/14/5/510]
- 58. Low-cost electrochemical detection of arsenic in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8769315/]
- 59. Portable platform for smartphone-enabled arsenic ... [https://www.sciencedirect.com/science/article/pii/S0925400525016053]
- 60. Inorganic arsenic speciation in water and seawater by ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267007000104]
- 61. Recent Advances in Colorimetric Detection of Arsenic Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8235315/]
- 62. Electrochemical sensor based on Co 3 O 4 and Au ... [https://www.nature.com/articles/s41598-025-25076-3]
- 63. Development and Comparison of New Voltammetric ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12655099/]
- 64. The principles of presenting statistical results: Table - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8024217/]
- 65. 7.3: Presenting Data in Tables [https://stats.libretexts.org/Bookshelves/Applied_Statistics/Biological_Statistics_(McDonald)/07%3A_Miscellany/7.03%3A_Presenting_Data_in_Tables]
- 66. Measuring Arsenic Contamination in Water [https://www.palintest.com/measuring-arsenic-contamination-in-water-challenges-and-solutions/]