Interference Resistance Showdown: A Comparative Analysis of Bismuth, Mercury, and Gold Film Electrodes for Advanced Electrochemical Biosensing
This article provides a comprehensive comparative analysis of the interference resistance of Bismuth Film Electrodes (BiFEs), Mercury Film Electrodes (HgFEs), and Gold Film Electrodes (AuFEs) in electrochemical biosensing.
Interference Resistance Showdown: A Comparative Analysis of Bismuth, Mercury, and Gold Film Electrodes for Advanced Electrochemical Biosensing
Abstract
This article provides a comprehensive comparative analysis of the interference resistance of Bismuth Film Electrodes (BiFEs), Mercury Film Electrodes (HgFEs), and Gold Film Electrodes (AuFEs) in electrochemical biosensing. Tailored for researchers and drug development professionals, it explores the fundamental electrochemical properties, surface characteristics, and inherent anti-fouling capabilities of each electrode material. The scope extends to methodological applications across healthcare diagnostics, environmental monitoring, and bioprocessing, offering practical strategies for troubleshooting common interferents like ascorbic acid, uric acid, and proteins. By systematically validating performance through sensitivity, selectivity, and real-sample analysis metrics, this review serves as a critical guide for selecting optimal electrode materials to enhance the reliability and accuracy of next-generation biosensors in complex matrices.
Understanding the Electrochemical Foundations: Intrinsic Properties of BiFE, HgFE, and AuFE
The selection of electrode material is a fundamental decision in electrochemical sensing, directly determining a sensor's sensitivity, selectivity, and operational robustness. Within trace metal detection and biomedical applications, the competition between traditional and novel electrode materials centers on performance and practicality. This guide provides a comparative analysis of Bismuth Film Electrodes (BiFE), Mercury Film Electrodes (HgFE), and Gold Film Electrodes (AuFE), with a specific focus on their interference resistance—a critical property for reliable analysis in complex sample matrices like biological fluids and environmental waters. We objectively evaluate these materials using published experimental data on their electrical properties, interference behavior, and electroanalytical performance.
Core Electrode Materials: Composition and Fabrication
Material Composition and Structure
- Bismuth Film Electrodes (BiFE): Bismuth is electrodeposited as a film onto various substrates, such as glassy carbon or brass, via ex-situ or in-situ protocols. The multi-pulse galvanostatic method allows for favorable control of the nanostructured bismuth film deposition, creating a highly sensitive surface [1] [2].
- Mercury Film Electrodes (HgFE): The traditional standard, formed by electrodepositing a mercury film on substrates like carbon. Despite their excellent electroanalytical performance, concerns over mercury's toxicity have driven the search for alternatives [1] [2].
- Gold Film Electrodes (AuFE): Typically use a gold surface, either as a solid microelectrode or as a film. Gold electrodes are prone to surface oxidation and adsorption of organic species, which can foul the electrode and require regular renewal [3].
Fabrication Protocols
The fabrication process is critical for achieving reproducible and high-performance electrodes.
Table 1: Standardized Fabrication Protocols for Film Electrodes
| Electrode Type | Fabrication Method | Key Parameters | Substrate Examples |
|---|---|---|---|
| BiFE (ex-situ) | Multi-pulse galvanostatic deposition | Pulse current, pulse/relaxation time, plating solution (e.g., Bi(III) in HCl with NaBr) [1] | Glassy Carbon, Brass [1] [2] |
| BiFE (in-situ) | Co-deposition with analyte | Bi(III) concentration 10x higher than target analytes [1] | Glassy Carbon, Carbon Paste |
| HgFE | Electrochemical plating | Applied potential/deposition time in Hg(II) solution | Glassy Carbon, Platinum |
| AuFE | Sputtering or pre-formed | Surface polishing and electrochemical pre-treatment [3] | Solid Gold, Silicon |
The diagram below illustrates the standard workflow for fabricating an ex-situ Bismuth Film Electrode, highlighting the control points that determine its final nanostructure and performance.
Performance Comparison: Interference Resistance and Analytical Figures of Merit
Interference resistance encompasses an electrode's ability to function accurately despite challenges like dissolved oxygen, competing ions, and surface fouling.
Comparative Electrical Properties and Intrinsic Interference
Fundamental electrical properties reveal how different electrode materials behave under signal acquisition conditions.
Table 2: Comparative Electrical Properties of Electrode Materials
| Property | Bismuth-based Electrodes | Gold-based Electrodes | Implication for Interference Resistance |
|---|---|---|---|
| Typical Impedance (at 50 Hz) | Low (e.g., 65-343 Ω for Ag/Bi) [3] | High (e.g., 286-2913 Ω for Au) [3] | Lower impedance reduces signal loss and vulnerability to electrical noise. |
| Phase Angle | Smaller (e.g., -6° to -34°) [3] | Larger (e.g., -60° to -70°) [3] | A smaller phase angle indicates more resistive behavior, favorable for stable potential. |
| Background Current | Low [1] | Variable | Lower background contributes to a better signal-to-noise ratio. |
| Operational Potential Window | Wide (approaching Hg) [1] | Limited by Au oxidation | A wider window allows detection of more species without solvent electrolysis. |
| Sensitivity to O₂ | Low (Insensitive) [1] | Moderate | Enables analysis without rigorous deoxygenation, simplifying the protocol. |
Analytical Performance in Trace Metal Detection
Anodic Stripping Voltammetry (ASV) for heavy metal detection is a rigorous test of electrode performance, particularly in the presence of interfering species.
Table 3: Electroanalytical Performance for Trace Metal Detection
| Parameter | Nanostructured BiFE | Conventional HgFE | Gold Electrode |
|---|---|---|---|
| Detection Limit (Pb²⁺) | 0.1 µg/L (0.5 nM) [1] | ~0.05-0.1 µg/L (comparable) | Generally higher than BiFE/HgFE |
| Detection Limit (Cd²⁺) | 0.4 µg/L (3.6 nM) [1] | ~0.1-0.2 µg/L (comparable) | Generally higher than BiFE/HgFE |
| Signal Reproducibility (RSD) | 3.8% for Pb(II), 5.1% for Cd(II) [1] | Typically <5% | Highly dependent on surface history |
| Cationic Interference (Cr³⁺, Mn²⁺, Zn²⁺, etc.) | No influence on Cd²⁺ signal [2] | Minimal influence | Prone to intermetallic compound formation |
| Key Advantage | Non-toxic, low O₂ sensitivity [1] | Well-established, excellent sensitivity | Good for specific applications (e.g., CNTs) |
| Key Disadvantage | --- | High toxicity [1] | Surface fouling, requires regeneration |
The Scientist's Toolkit: Essential Research Reagents
The following reagents are fundamental for the fabrication and testing of bismuth film electrodes, as derived from the cited experimental protocols [1] [2].
Table 4: Essential Reagents for Bismuth Film Electrode Research
| Reagent | Function/Application | Example from Protocol |
|---|---|---|
| Bismuth(III) Nitrate Pentahydrate (Bi(NO₃)₃·5H₂O) | Primary source of Bi(III) ions for film electrodeposition. | Used in HCl solution for ex-situ BiFE formation on brass [2]. |
| Supporting Electrolyte (Acetate Buffer) | Provides ionic conductivity and controls pH during analysis. | 0.1 M acetate buffer (pH 4.5) used for Anodic Stripping Voltammetry (ASV) [1] [2]. |
| Sodium Bromide (NaBr) | Acts as an auxiliary ligand in the plating solution, improving film quality. | Added to the plating solution for nanostructured BiFE; part of parameter optimization [1]. |
| Target Metal Ion Standards (Cd²⁺, Pb²⁺) | Analytes for sensor calibration and performance testing. | Stock solutions (1000 mg/L) diluted to working concentrations for ASV [1] [2]. |
| Hydrochloric Acid (HCl) | Acidic medium for ex-situ bismuth film deposition, suppresses Bi(III) hydrolysis. | 1 M HCl used as the plating solution for BiFE on a brass substrate [2]. |
| Balanced Salt Solution (BSS) | Simulates physiological conditions (e.g., tears) for electrical testing. | Applied to keep ocular electrodes soaked during impedance and noise testing [3]. |
Experimental Workflow for Interference Testing
A standardized methodology is required to objectively compare the interference resistance of different electrode materials. The following workflow details a key experiment for evaluating cationic interference.
Detailed Experimental Protocol:
- Electrode Preparation: Fabricate BiFE, HgFE, and AuFE following standardized protocols (see Table 1). Characterize the surface using Scanning Electron Microscopy (SEM) and Electrochemical Impedance Spectroscopy (EIS) to confirm morphology and low impedance [1] [2].
- Baseline Measurement: Using Anodic Square-Wave Stripping Voltammetry (SWASV) in a 0.1 M acetate buffer (pH 4.5), record the stripping signals for a fixed, low concentration of target analytes (e.g., 5 µg/L each of Cd(II) and Pb(II)). Note the peak potentials and currents [1] [2].
- Introduce Interferents: To the same electrochemical cell, add a mixture of potential interfering cations (e.g., Cr³⁺, Mn²⁺, Zn²⁺, Ca²⁺, K⁺, Mg²⁺, Na⁺) at a concentration significantly higher (e.g., 10-50x) than the target analytes [2].
- Post-Interference Measurement: Without renewing the electrode surface, repeat the SWASV measurement under identical conditions.
- Data Analysis: Compare the stripping signals before and after the addition of interferents. Key metrics include:
- Signal Suppression: Calculate the percentage change in peak current for Cd(II) and Pb(II).
- Peak Shift: Note any change in peak potential, which indicates altered electrochemical kinetics.
- Baseline Stability: Observe changes in the background current, which can suggest non-specific adsorption or surface fouling.
This comparative analysis, grounded in experimental data, demonstrates that Bismuth Film Electrodes (BiFE) present a compelling, high-performance alternative to traditional Mercury Film Electrodes (HgFE) and Gold Film Electrodes (AuFE). BiFEs match or approach the excellent sensitivity and low detection limits of HgFEs for key heavy metals like cadmium and lead, while offering the decisive advantage of being environmentally friendly and non-toxic [1] [2]. Critically, in terms of interference resistance, BiFEs exhibit superior intrinsic properties—including lower electrical impedance and reduced sensitivity to dissolved oxygen—and demonstrate robust operational resistance against common cationic interferents [1] [3] [2]. Although HgFE remains a benchmark for electroanalytical performance, its high toxicity is a major liability. AuFE, while useful in specific contexts, is hampered by higher noise and surface fouling issues [3]. Therefore, for researchers and drug development professionals designing sensors for complex matrices, the Bismuth Film Electrode emerges as the optimal choice, successfully balancing exceptional analytical performance, remarkable interference resistance, and practical laboratory safety.
The biological performance of any material implanted in the body is dictated by a complex interplay at its surface. Within seconds of exposure to bodily fluids, non-specific protein adsorption occurs on implant surfaces, triggering a cascade of host immune responses known as the foreign body reaction [4] [5]. This reaction often results in the formation of a fibrous avascular capsule that can isolate the device, leading to complications such as inflammation, fibrosis, thrombosis, infection, and ultimately, implant failure [6] [5]. Consequently, controlling interactions at the bio-material interface represents one of the most significant challenges in developing advanced medical implants and biosensors.
Surface chemistry provides a powerful approach to engineer this biological interface. By strategically modifying surface characteristics—including chemical functionality, wettability, and charge—researchers can directly influence the amount, composition, and conformational state of adsorbed proteins, thereby directing subsequent cellular responses [6] [4]. This guide explores how different surface chemistries, particularly those creating non-fouling or bioactive surfaces, compare in their ability to mitigate adverse reactions and promote desired tissue integration. Understanding these principles is fundamental to the rational design of next-generation medical devices with enhanced biocompatibility and interference resistance.
Table: Fundamental Concepts in Surface Biocompatibility
| Concept | Description | Biological Consequence |
|---|---|---|
| Foreign Body Reaction | A host defense mechanism triggered by implanted materials [5]. | Leads to fibrous capsule formation, isolating the device from target tissues [5]. |
| Protein Adsorption | The rapid, non-specific coating of an implant surface with host proteins within seconds of implantation [4] [5]. | Initiates the foreign body reaction; the composition and conformation of proteins direct immune responses [4]. |
| Conformational Change | The alteration of a protein's three-dimensional structure upon adsorption to a surface [4]. | Can expose inflammatory epitopes (e.g., RIBS in fibrinogen) that activate immune cells [4]. |
| Non-Fouling Surface | A surface modified to prevent or reduce non-specific protein adsorption [5]. | Creates "stealthy" implants that evade the host's immune surveillance [5]. |
| Bioactive Surface | A surface engineered with specific molecular signals (e.g., peptides, cytokines) [5]. | Promotes specific, desired interactions with surrounding cells and tissues [5]. |
Key Surface Modification Techniques and Their Biocompatibility
Several advanced techniques have been developed to create well-defined surfaces with specific chemical functionalities. The choice of method depends on the base material, desired functionality, and application requirements.
Self-Assembled Monolayers (SAMs)
SAMs provide exceptional control over surface properties by forming dense, well-ordered layers of functional molecules on substrates like gold and silver [4]. They are easily modified and assembled from solution, making them an excellent platform for in vitro studies [5]. Researchers have used SAMs with hydrophilic terminal groups (e.g., NH₃⁺ and COO⁻Na⁺) or mixed charged functionalities to create neutral surfaces that significantly reduce platelet adhesion compared to hydrophobic counterparts [5]. A key advantage is the ability to present a high density of specific functional groups or bioactive ligands in a controlled manner.
Plasma Surface Modification
This economical and effective technique uses highly excited gaseous species to confer new functionalities to a material's surface [4]. It is compatible with a wide range of materials, including polymers and metals [4]. For instance, ammonia plasma treatment can introduce amine groups onto polytetrafluoroethylene (PTFE) surfaces, which can subsequently be used to covalently couple cell-adhesive peptides [7]. Another study used "reactive landing" plasma deposition to coat 316L stainless steel with hyaluronan, effectively inhibiting platelet aggregation and activation—a critical feature for cardiovascular stents [5].
Chemical Grafting
This method involves the covalent conjugation of molecules or polymers to a material's surface, often after an initial activation step using chemicals, UV, or ozone [4]. It provides long-term stability without concerns of desorption. A prominent example is the grafting of poly(ethylene glycol) (PEG) or heparin to create non-fouling surfaces [4] [5]. PEG works by forming a steric repulsion barrier and a hydration shell that prevents protein adsorption and denaturation [5]. While highly stable, a drawback can be the potential loss of mobility of grafted biomolecules and their presentation in an unnatural conformation [4].
Table: Comparison of Surface Modification Techniques
| Technique | Interaction Type | Advantages | Limitations | Common Applications |
|---|---|---|---|---|
| Self-Assembled Mononolayers (SAMs) | Chemisorptive (e.g., Thiol-Gold) [5] | Precise control over functionality density and pattern; easy modification [4] [5]. | Limited to specific substrates (e.g., gold, silver) [4]. | Fundamental in vitro studies of protein-cell-biomaterial interactions [5]. |
| Plasma Modification | Covalent [5] | Economical; effective on most materials (metals, polymers) [4]. | Can involve high-energy processing [5]. | Introducing amine groups on PTFE [7]; creating hyaluronan coatings on steel [5]. |
| Chemical Grafting | Covalent [5] | Long-term stability; strong, non-desorbable coating [5]. | Caustic processing may damage delicate biologics; can immobilize proteins in unfamiliar conformations [4]. | Grafting of PEG for non-fouling surfaces; heparin for blood compatibility [4]. |
Experimental Data and Performance Comparison
Rigorous experimental evaluation is essential for comparing the performance of different surface modifications. The following data, drawn from controlled studies, highlights how surface chemistry directly influences biocompatibility outcomes.
Protein Adsorption and Cell Viability
The capacity of a surface to control protein interactions directly correlates with cell viability. A comparative study on modified PTFE surfaces demonstrated this relationship clearly. Novel nanostructured PTFE (nPTFE) showed a higher protein adsorption capacity for Human Serum Albumin (HSA) compared to standard expanded PTFE (ePTFE) when measured via depletion analysis [7]. More significantly, this difference in surface properties had a dramatic effect on cell behavior. While endothelial cells largely died off on ePTFE surfaces within 96 hours, they remained viable on the nPTFE surface during the same period [7]. This underscores the critical link between tailored surface nano-structure, protein interactions, and cellular outcomes.
Electrochemical Corrosion Resistance
In metallic implants, surface chemistry and structure critically influence the stability of the passive layer that protects against corrosion. Research on amorphous 316-type stainless steel microfibers demonstrates this principle. When tested in a saline environment (3.5 wt% NaCl), the amorphous surface exhibited a low current density of 0.77 × 10⁻⁵ A cm⁻² at 25°C, a key indicator of high corrosion resistance [8]. This performance remained stable, with only a slight increase to 1.0 × 10⁻⁵ A cm⁻² at 50°C [8]. The homogenous amorphous structure facilitates the formation of a stable, protective surface oxide, outperforming its crystalline counterparts.
Table: Quantitative Comparison of Biocompatibility Performance
| Material / Surface | Experimental Method | Key Performance Metric | Result | Implication for Biocompatibility |
|---|---|---|---|---|
| Nanostructured PTFE (nPTFE) | HSA adsorption isotherms; Endothelial cell culture [7] | Protein adsorption capacity; Cell viability after 96h [7] | Increased HSA capacity; High cell viability [7] | Improved protein interactions support long-term cell survival. |
| Amorphous 316 SS | Potentiodynamic polarization in 3.5% NaCl [8] | Corrosion current density at 25°C [8] | 0.77 × 10⁻⁵ A cm⁻² [8] | Excellent corrosion resistance and passive film stability in physiological saline. |
| WC-Al₂O₃ Composite | Immersion corrosion in NaCl solution; Electrochemical tests [9] | Mass loss after 28 days; Corrosion product formation [9] | No significant mass loss; Products mainly WO₃ [9] | Higher corrosion resistance than WC-Co; avoids galvanic corrosion. |
| WC-6Co Cemented Carbide | Immersion corrosion in NaCl solution; Electrochemical tests [9] | Mass loss after 28 days; Corrosion product formation [9] | Significant mass loss; Products include Co(OH)₂, Co₃O₄, WO₃ [9] | Galvanic corrosion between WC and Co binder degrades material. |
Detailed Experimental Protocols
To ensure reproducibility and provide a framework for comparative analysis, this section outlines standardized protocols for key experiments used to evaluate surface biocompatibility.
Protocol for Protein Adsorption Measurement on Superhydrophobic Surfaces
Accurately measuring protein adsorption on highly hydrophobic materials like PTFE requires specific steps to account for air entrapment.
- Step 1: Sample Preparation. Cut polymer samples to a standardized size (e.g., 1 cm²). Clean surfaces ultrasonically in ethanol to degrease [7].
- Step 2: Surface Evacuation. Prior to contact with protein solution, evacuate the air around the expanded PTFE samples. This critical step ensures the solution penetrates the microstructure [7].
- Step 3: Solution Degassing. Degas the PBS buffer solutions to prevent bubble formation during the experiment [7].
- Step 4: Protein Incubation. Immerse the evacuated samples in a solution of the protein of interest (e.g., I-125 labeled or unlabeled Human Serum Albumin) for a set duration at physiological temperature (37°C) [7].
- Step 5: Washing and Analysis. Gently wash the samples with PBS to remove loosely bound protein. Quantify adsorbed protein using two parallel methods:
- Note: Compare results from both methods, as radiometry may alter protein hydrophobicity and underreport total adsorption [7].
Protocol for Electrochemical Corrosion Testing
Electrochemical methods are used to quantitatively assess the corrosion resistance of metallic and composite materials.
- Step 1: Sample Preparation. The working electrode is prepared by cutting the test material to a defined exposed area (typically 1 cm²). The surface is then ground and polished with successive grits (e.g., 600 to 4000) and finished with a 1 μm diamond paste, followed by ultrasonic cleaning in ethanol and drying [9].
- Step 2: Experimental Setup. A standard three-electrode cell is used:
- Working Electrode: The test sample.
- Counter Electrode: A platinum sheet.
- Reference Electrode: A saturated calomel electrode (SCE) [9].
- Step 3: Open Circuit Potential (OCP). Immerse the cell in the electrolyte (e.g., 3.5 wt% NaCl solution) and monitor the OCP for a stabilized period (e.g., 12 hours) until it reaches a steady-state value [9].
- Step 4: Electrochemical Impedance Spectroscopy (EIS). Perform EIS at the OCP value. Apply a sinusoidal potential perturbation (typically 10 mV amplitude) across a wide frequency range (e.g., 100 kHz to 1 mHz) to measure the impedance response [9].
- Step 5: Potentiodynamic Polarization. After EIS, perform a Tafel scan by polarizing the sample from a cathodic potential (e.g., -1000 mV vs. SCE) to an anodic potential (e.g., +1500 mV vs. SCE) at a slow, controlled scan rate (e.g., 2 mV s⁻¹) [9]. This generates a current-potential curve from which corrosion current density (I_corr) and other parameters are derived.
Foreign Body Reaction Pathway
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful research into surface chemistry and biocompatibility relies on a suite of specialized materials and reagents.
Table: Essential Research Reagents and Materials
| Item | Function/Description | Application Example |
|---|---|---|
| Self-Assembled Monolayer (SAM) Kits | Provide thiolated molecules with various terminal groups (e.g., CH₃, OH, COOH) for creating well-defined model surfaces on gold substrates [4] [5]. | Studying the specific effect of surface energy and charge on fibrinogen adsorption and platelet adhesion [5]. |
| I-125 Labeled Proteins | Radioisotope-labeled proteins (e.g., Human Serum Albumin, Fibrinogen) used in traditional radiometry assays to quantify protein adsorption [7]. | Measuring the amount of protein adsorbed onto a polymer surface after washing. Note: may alter protein behavior [7]. |
| Poly(ethylene glycol) (PEG) Derivatives | A widely used polymer for creating non-fouling surface brushes via chemical grafting or adsorption. Its chains create steric repulsion and a hydration shell [5]. | Grafting to cardiovascular stent materials to reduce non-specific protein adsorption and thrombosis [5]. |
| Hyaluronic Acid (HA) / Hyaluronan | A polysaccharide used to create hydrophilic, non-fouling coatings that inhibit platelet aggregation and activation [5]. | Coating 316L stainless steel surfaces for cardiovascular applications using techniques like "reactive landing" [5]. |
| Electrochemical Workstation | Instrument for performing OCP, EIS, and Tafel polarization measurements. Typically includes a potentiostat, frequency response analyzer, and a three-electrode cell [9]. | Quantifying the corrosion resistance and passive film stability of a new amorphous stainless steel alloy in simulated body fluid [8]. |
Surface Modification Strategies to Control Biocompatibility
The quest for superior biocompatibility is fundamentally a quest for control over the biological interface. As this guide demonstrates, surface chemistry provides the primary tools for achieving this control. From creating non-fouling surfaces with PEG or hyaluronic acid to engineering bioactive surfaces with peptides and SAMs, the evidence shows that even minor, molecular-level changes can dramatically alter protein behavior, cellular responses, and long-term material performance. The comparative success of materials like nanostructured PTFE and amorphous stainless steel further underscores that physical and chemical surface properties are inextricably linked in determining biological outcomes. For researchers and drug development professionals, a deep understanding of these principles is no longer optional but essential for designing the next generation of interferent-resistant, bio-integrative medical devices and diagnostic platforms.
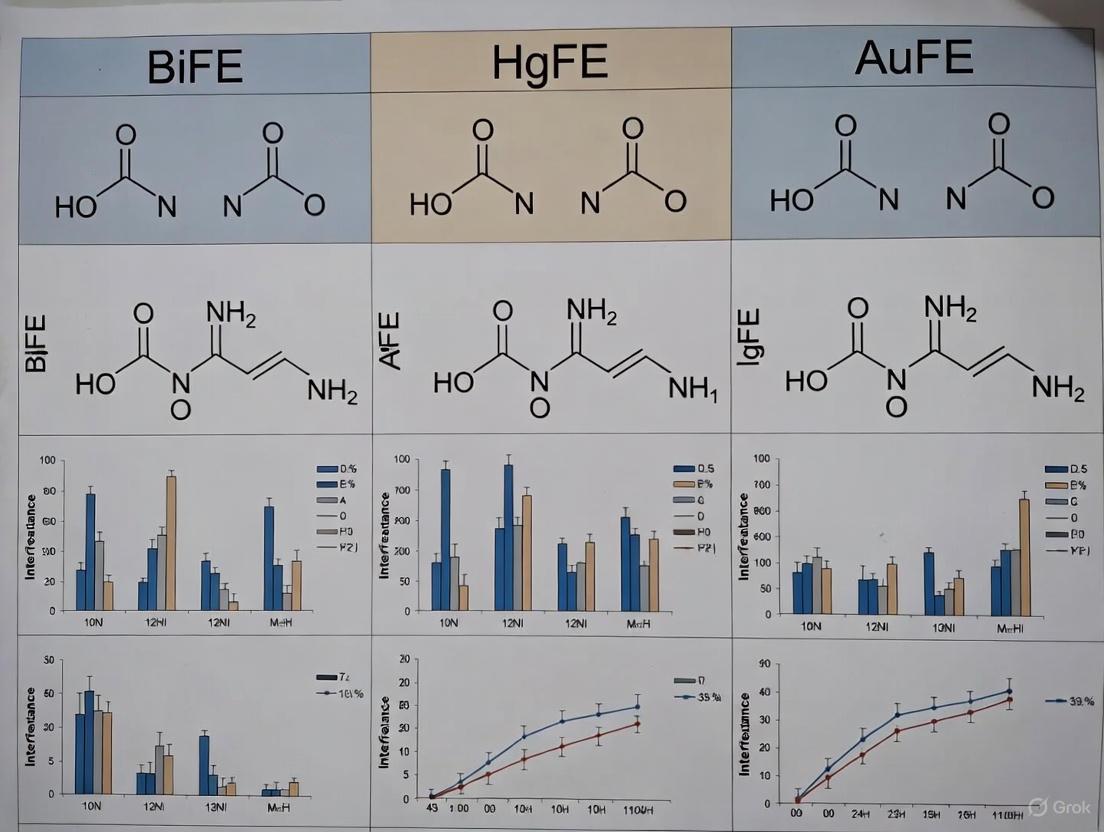
Electrochemical sensors are powerful tools for analyzing biological samples, but their accuracy is often compromised by interference from electroactive species. This guide compares the interference resistance of three key electrode materials—Bismuth Film Electrodes (BiFE), Mercury Film Electrodes (HgFE), and Gold Film Electrodes (AuFE)—providing experimental data and methodologies to inform their use in drug development and clinical research.
The analysis of biological samples such as blood, serum, and urine using electrochemical sensors is a cornerstone of modern pharmaceutical and clinical research [10]. These sensors are prized for their high sensitivity, rapid response, cost-effectiveness, and miniaturization potential [10]. However, a significant challenge persists: the presence of endogenous and exogenous electroactive species in complex biological matrices can generate non-specific signals, leading to inaccurate readings [11]. This interference is a critical concern for researchers and professionals who rely on precise measurements for drug monitoring, disease diagnosis, and metabolic studies.
Interference mechanisms vary with sensor design. Electroactive substances like ascorbic acid, uric acid, and acetaminophen can be directly oxidized or reduced at the electrode surface, creating a current that overlaps with the target analyte's signal [11]. Other substances may interfere with the enzymatic biorecognition element in biosensors or foul the electrode surface, degrading performance over time [11]. The composition of interstitial fluid (ISF), where many minimally invasive sensors operate, is particularly challenging, as it contains most analytes found in blood, but their physiological concentrations are often poorly characterized [11].
This guide objectively compares the interference resistance of BiFE, HgFE, and AuFE. By synthesizing recent experimental data and detailed protocols, we provide a framework for selecting the optimal electrode material to mitigate interference in specific analytical contexts.
Performance Comparison of BiFE, HgFE, and AuFE
The following tables summarize the analytical performance and interference resistance of BiFE, HgFE, and AuFE as documented in recent research. The data provides a direct comparison of their capabilities for determining specific metals and drugs in various sample matrices.
Table 1: Analytical Performance of BiFE, HgFE, and AuFE for Metal Detection
| Electrode | Target Analyte | Technique | Linear Range | Detection Limit | Sample Matrix |
|---|---|---|---|---|---|
| BiFE [12] | In(III) | AdSV with PAR | ( 2 \times 10^{-7} ) to ( 4 \times 10^{-6} ) M | ( 3.5 \times 10^{-9} ) M | Tap water, ITO glass |
| HgFE [12] | Be(II) | AdSV with Thorin I | 0.6 to 7.7 μg·L⁻¹ | 0.08 μg·L⁻¹ | Natural water, beryl mineral |
| AuFE [12] | As(III) | SWASV | 10 to 250 μg·L⁻¹ | 1 μg·L⁻¹ | Tap water, seafood |
Table 2: Interference Resistance of BiFE, HgFE, and AuFE
| Electrode | Target Analyte | Interfering Species Studied | Interference Mitigation Strategy | Key Outcome |
|---|---|---|---|---|
| BiFE [12] | In(III) | Tl(I), Zn(II), Cu(II), Pb(II), Co(II), Ni(II), Mn(II), Fe(III), Cr(III) | Medium exchange procedure | Effectively eliminated some interferences for tap water and ITO glass analysis. |
| HgFE [12] | Be(II) | 10 foreign ions | Method optimization; in-situ plating | High reproducibility (RSD = 3.6% for n=10) without repeated film application. |
| AuFE [12] | As(III) | Fe(III), Mn(II), Pb(II), Cu(II), Sn(IV), Tl(I) | Electrode optimization (deposition parameters) | Validated for quantitative determination in tap water and seafood (shrimp, cod liver). |
Experimental Protocols for Electrode Evaluation
To ensure the reliability and reproducibility of interference resistance data, standardized experimental protocols are essential. The following sections detail the methodologies used for electrode preparation, sensor characterization, and interference testing for BiFE, HgFE, and AuFE.
Electrode Fabrication and Optimization
Gold-Film Electrode (AuFE) for Arsenic Detection [12]: The AuFE was fabricated ex-situ by potentiostatic electrodeposition of a gold layer onto a rotating glassy carbon electrode (GCE). Key deposition parameters were systematically optimized:
- HAuCl₄ concentration: varied from 0.25 to 4 mM.
- Deposition potential: tested from 0 to -600 mV.
- Deposition time: investigated between 120 and 1200 seconds.
- Electrode rotation speed: optimized from 600 to 1500 rpm. The resulting gold films were characterized using Cyclic Voltammetry (CV), optical microscopy, and Scanning Electron Microscopy (SEM) to correlate their physical properties with electrochemical performance.
Bismuth-Film Electrode (BiFE) for Indium Detection [12]: An ex-situ plated BiFE served as the working electrode. A potential-time program was developed for the inversion cycle stages, optimizing factors like:
- pH: found optimal at 4.8.
- Accumulation potential: set at -700 mV.
- Concentration of chelating agent (PAR): optimized at ( 1 \times 10^{-4} ) M.
Mercury-Film Electrode (HgFE) for Beryllium Detection [12]: The HgFE was plated in-situ on the electrode surface. The research selected optimal conditions for mercury film deposition and developed a potential-time program for the inversion cycle. The optimal analytical conditions included a 0.1 mol·L⁻¹ ammonium buffer at pH 9.5, containing ( 1 \times 10^{-6} ) mol·L⁻¹ Thorin I and ( 7.5 \times 10^{-4} ) mol·L⁻¹ mercury (II).
Interference Testing Methodology
Interference Study for BiFE [12]: The effect of interfering ions (Tl(I), Zn(II), Cu(II), Pb(II), Co(II), Ni(II), Mn(II), Fe(III), Cr(III)) on the indium signal was investigated. A medium exchange procedure was employed after the accumulation step to effectively eliminate some of these interferences, enhancing selectivity in complex matrices like tap water.
Interference Study for AuFE [12]: The influence of interfering ions (Fe(III), Mn(II), Pb(II), Cu(II), Sn(IV), Tl(I)) on the As(III) stripping peak was studied. The primary mitigation strategy was the optimization of the gold film deposition parameters itself, creating a surface that minimized the impact of interferents. The electrode's utility was then validated in real samples (tap water, Atlantic shrimp, cod liver).
The experimental workflow below illustrates the key stages in electrode fabrication and interference testing.
Mechanisms of Interference and Electrode Response
Understanding how different electrode materials respond to interfering species is key to selecting the right sensor. The following diagram illustrates the common interference pathways and how BiFE, HgFE, and AuFE counteract them.
The Scientist's Toolkit: Key Research Reagents & Materials
Successful experimentation with these electrodes requires a specific set of reagents and materials. The following table details essential components for fabricating and operating BiFE, HgFE, and AuFE, based on the cited protocols.
Table 3: Essential Materials for Electrode Research and Development
| Item Name | Function / Role | Example from Research |
|---|---|---|
| Glassy Carbon Electrode (GCE) | A common, well-defined substrate for film deposition. | Used as the base for rotating disk gold-film electrode (AuFE) for arsenic detection [12]. |
| Bismuth Salt (e.g., Bi(NO₃)₃) | Source of bismuth for forming the sensing film. | Used for the ex-situ preparation of the Bismuth Film Electrode (BiFE) for indium detection [12]. |
| Mercury(II) Salt (e.g., Hg²⁺) | Source of mercury for forming the mercury film. | Used at ( 7.5 \times 10^{-4} ) mol·L⁻¹ for in-situ plating of the HgFE for beryllium detection [12]. |
| Gold Salt (e.g., HAuCl₄) | Source of gold for electrodeposition of the film. | Concentration optimized between 0.25-4 mM for the fabrication of the AuFE [12]. |
| Complexing Agents (PAR, Thorin I) | Selective chelation of target metal ions to form an adsorptive complex. | PAR for In(III) on BiFE; Thorin I for Be(II) on HgFE [12]. |
| Supporting Electrolyte/Buffer | Provides ionic strength and controls pH for the electrochemical reaction. | 0.1 mol·L⁻¹ ammonium buffer (pH 9.5) for HgFE; BR buffer for general voltammetry [12]. |
| Modifying Agents (nRGO) | Nanomaterial used to modify electrode surfaces to enhance sensitivity and reduce fouling. | Nano-reduced graphene oxide (nRGO) used to modify carbon paste electrodes for drug analysis [13]. |
The choice between BiFE, HgFE, and AuFE involves a careful trade-off between analytical performance, interference resistance, environmental considerations, and practical handling. BiFEs present an eco-friendly alternative with good performance for certain metals like indium, though they may require additional steps like medium exchange to manage interference. HgFEs, while highly reproducible and excellent for forming complexes with metals like beryllium, involve the use of toxic mercury. AuFEs, through meticulous optimization of their deposition parameters, offer a robust platform for detecting toxic elements like arsenic in complex biological samples like seafood.
For researchers and drug development professionals, this comparison underscores that there is no universally superior electrode. The optimal selection is dictated by the specific target analyte, the complexity of the sample matrix, and the required detection limits. Future advancements are likely to focus on novel nanocomposite materials and smarter sensor designs that incorporate built-in selectivity, further mitigating the enduring challenge of interference in biological electroanalysis.
Theoretical Advantages and Limitations of Each Electrode Platform
The selection of an working electrode is a fundamental consideration in electrochemical sensing, profoundly influencing the sensitivity, selectivity, and overall robustness of analytical methods. This guide provides a comparative analysis of four prominent electrode platforms—Bismuth Film Electrodes (BiFEs), Mercury Film Electrodes (HgFEs), Gold Electrodes (AuFEs), and Antimony Film Electrodes (SbFEs). Each platform possesses distinct characteristics that determine its suitability for specific applications. The evaluation is contextualized within research on interference resistance, a critical property for analyzing complex samples in drug development and environmental monitoring. The objective is to furnish researchers and scientists with a structured overview of the theoretical strengths and weaknesses of each platform, supported by experimental data and protocols, to inform electrode selection for specific analytical challenges.
Theoretical Framework and Key Characteristics
Fundamental Operational Principles
Electrochemical sensing platforms for metal detection, particularly using anodic stripping voltammetry (ASV), operate on a shared principle: a preconcentration step where metal ions in solution are reduced and deposited onto the electrode surface, followed by a stripping step where the deposited metals are re-oxidized, producing a measurable current signal [14] [15]. The analytical performance, however, is dictated by the physicochemical properties of the electrode material itself. The key differentiators include the ability to form alloys with target analytes, the hydrogen overpotential, the available potential window, and the inherent chemical stability of the material in various electrolytes. These factors collectively determine the electrode's sensitivity, detection limits, and susceptibility to interferences from dissolved oxygen or surface-active compounds [16].
Comparative Theoretical Advantages and Limitations
Table 1: Theoretical Comparison of Electrode Platforms
| Electrode Platform | Core Principle & Key Advantage | Primary Theoretical Limitations |
|---|---|---|
| Bismuth Film Electrode (BiFE) | Forms low-temperature fusions/alloys with heavy metals, analogous to mercury but environmentally friendly; operates well in air-saturated solutions [16] [15]. | Performance can be sensitive to deposition conditions and solution chemistry; may suffer from poorer linearity at very low concentrations compared to HgFE [16]. |
| Mercury Film Electrode (HgFE) | The historical benchmark; offers a highly reproducible and wide cathodic potential window, excellent for forming amalgams with many metals [16]. | High toxicity of mercury and its salts poses environmental, safety, and disposal concerns, limiting its practical and routine use [17] [16]. |
| Gold Electrode (AuFE) | Provides a stable, mercury-free surface; integrated configurations enhance portability and minimize solution perturbation, ideal for point-of-care devices [18]. | Limited cathodic potential window due to hydrogen evolution reaction at relatively positive potentials, which can restrict its use for more electronegative metals [18]. |
| Antimony Film Electrode (SbFE) | Functions effectively in highly acidic media (pH ≤ 2), offering a wider operational window in acid than BiFEs, with a very small self-stripping signal [17]. | Performance is highly dependent on the substrate and plating method; "green" credential is inferior to bismuth, though much better than mercury [17]. |
Comparative Experimental Data and Performance
Analytical Performance in Trace Metal Detection
Experimental data from the literature allows for a quantitative comparison of the analytical performance of these electrode platforms, particularly for the detection of trace heavy metals like Cd(II), Pb(II), and Zn(II).
Table 2: Experimental Performance Data for Trace Metal Detection
| Electrode Platform | Target Analyte | Sensitivity / Performance Metric | Limit of Detection (LOD) | Key Experimental Conditions | Ref. |
|---|---|---|---|---|---|
| BiFE (Nafion-coated) | Cd(II), Pb(II), Zn(II) | LODs: 2 μg L⁻¹ (Cd, Pb), 6 μg L⁻¹ (Zn) | 2-6 μg L⁻¹ | SIA-SWASV; Acetate buffer pH 4.5 | [15] |
| BiFE (Screen-printed) | Cd(II), Pb(II) | Analytical sensitivity defined by calibration slope | Low μg L⁻¹ range | Differential Pulse Stripping; Acetate buffer pH 4.5 | [16] |
| Integrated Au EGFET | H⁺ (pH sensing) | Sensitivity: 96 mV/pH; Linearity: 97% | N/A | pH 4-10 range; Integrated configuration | [18] |
| SbFE | Various heavy metals | Performance comparable to BiFE, superior in strong acid | Low μg L⁻¹ range | Anodic Stripping Voltammetry; Acidic media (pH ~2) | [17] |
Interference Resistance and Stability
A critical aspect of sensor performance is its ability to function reliably in complex matrices. Modifications and material choices significantly impact interference resistance.
- Polymer-Coating Enhanced Resistance: A key strategy for improving the robustness of film electrodes is the application of permselective coatings. For instance, Nafion-coated BiFEs demonstrate significantly improved resistance to fouling by surface-active compounds. The Nafion layer acts as a cation-exchange permselective membrane, preventing large organic anions and surfactants from reaching the electrode surface while allowing the target metal cations to pass through, thereby reducing artifacts and stabilizing the signal [15].
- Stability Across pH and Configuration: The intrinsic stability of the electrode material varies. SbFEs maintain a wide operational potential window and stable performance in solutions with a pH of 2 or lower, a condition under which BiFEs may be less effective [17]. Furthermore, the physical configuration of the electrode influences stability. Integrated Au electrodes have demonstrated superior signal stability with minimal drift compared to conventional external reference electrodes (e.g., Ag/AgCl), attributed to decreased perturbation and closer proximity between the reference and sensing areas [18].
Experimental Protocols & Methodologies
Fabrication and Modification of Electrode Platforms
The performance of film electrodes is highly dependent on precise fabrication protocols. Below is a generalized workflow for the preparation of a polymer-coated bismuth film electrode, a common and advanced configuration.
Key Experimental Protocols
1. Protocol for Nafion-Coated Bismuth Film Electrode (BiFE) [15]:
- Substrate Preparation: A glassy carbon electrode is typically polished with alumina slurry (e.g., 0.3 μm), rinsed thoroughly with distilled water, and dried.
- Polymer Coating: The electrode is coated by depositing a small volume (e.g., 1 μL) of a Nafion solution (e.g., 0.5% - 2% w/w in ethanol) onto the surface and allowing it to dry in air, forming a permselective membrane.
- Bismuth Film Deposition (Ex-Situ): The coated electrode is immersed in a plating solution containing a Bi(III) salt (e.g., 0.1 mM Bi(NO₃)₃ in 0.1 M acetate buffer, pH 4.5). A deposition potential (e.g., -1.20 V vs. Ag/AgCl) is applied for a set time (e.g., 30-60 seconds) with stirring to electroplate the bismuth film.
- Measurement: The prepared BiFE is then transferred to the sample solution for the Anodic Stripping Voltammetry (ASV) measurement, involving a deposition step and a subsequent stripping scan.
2. Protocol for Antimony Film Electrode (SbFE) [17]:
- Film Deposition: SbFEs can be prepared via in-situ or ex-situ plating on carbon substrates. For ex-situ plating, the substrate electrode is immersed in a solution of Sb(III) (e.g., in HCl), and a constant potential is applied to reduce Sb(III) to metallic Sb, forming a film on the surface. The electrode is then rinsed and transferred to the sample solution.
3. Protocol for Integrated Gold Electrode (AuFE) in EGFET [18]:
- Fabrication: The integrated Au reference electrode is fabricated directly onto the extended-gate field-effect transistor (EGFET) chip post-CMOS process using Au wire-bonding techniques, creating a compact and stable sensor apparatus.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for Electrode Fabrication and Analysis
| Item | Function / Application | Example in Protocol |
|---|---|---|
| Bismuth Nitrate Pentahydrate (Bi(NO₃)₃·5H₂O) | Precursor for forming the bismuth film on BiFEs. | Preparing a 0.1 mM plating solution in acetate buffer [16] [15]. |
| Antimony Salt (e.g., Sb₂O₃) | Precursor for forming the antimony film on SbFEs. | Used in ex-situ or in-situ plating solutions, or mixed into carbon paste for bulk-modified electrodes [17]. |
| Nafion Perfluorinated Resin | Cation-exchange polymer coating to enhance interference resistance and mechanical stability of film electrodes. | Drop-cast as a 0.5-2% solution in ethanol onto the electrode surface [15]. |
| Acetate Buffer (pH ~4.5) | A common supporting electrolyte that provides a controlled pH environment and ionic strength for the analysis of many heavy metals. | Used as the carrier and sample matrix in SIA-ASV [15]. |
| Metal Standard Solutions (Cd, Pb, Zn, etc.) | Certified reference materials for calibration and validation of analytical methods. | Used to prepare working standards by dilution in the supporting electrolyte [16]. |
| Screen-Printed Electrodes (SPEs) | Disposable, miniaturized substrates that integrate working, reference, and counter electrodes, ideal for portability. | Serve as the substrate for preparing screen-printed BiFEs or SbFEs [17] [16]. |
The choice of an electrode platform involves a careful balance between analytical performance, environmental and safety considerations, and practical operational requirements. While the HgFE remains a historical benchmark for reproducibility and sensitivity, its significant toxicity has driven the development of competitive "green" alternatives. The BiFE has emerged as the most prominent successor, offering a favorable combination of low toxicity, good sensitivity, and the unique advantage of oxygen-insensitive operation. The SbFE presents a robust alternative, particularly for analyses requiring highly acidic conditions. Meanwhile, AuFEs, especially in integrated configurations, excel in applications demanding mechanical stability and miniaturization for portable sensing. The integration of polymeric coatings like Nafion further enhances the interference resistance of film electrodes, making them suitable for complex sample matrices. Ultimately, the selection depends on the specific analytical targets, the sample matrix, and the required operational constraints, with BiFEs and SbFEs standing out as the most viable and versatile mercury-free platforms for modern electroanalysis.
Practical Deployment: Methodologies and Real-World Applications in Complex Matrices
Optimized Sensor Fabrication Protocols for BiFE, HgFE, and AuFE
The detection of trace metals and organic compounds in environmental and biological samples is a critical task in analytical chemistry, with electroanalytical methods emerging as powerful tools for such applications. Mercury-film electrodes (HgFEs) have historically been the cornerstone of stripping voltammetry due to their exceptional reproducibility and wide negative potential window. However, the well-documented toxicity of mercury has driven extensive research into environmentally friendly alternatives. Bismuth-film electrodes (BiFEs) have risen as the most prominent mercury-free substitute, offering low toxicity, favorable electrochemical properties, and the ability to form alloys with metals. Simultaneously, gold-film electrodes (AuFEs) and related bimetallic configurations have been developed for their high electrocatalytic activity, stability, and specificity for certain analytes.
A critical factor in selecting an electrode for complex matrices, such as biological fluids or environmental samples, is its interference resistance—the ability to maintain accurate detection of a target analyte in the presence of other competing ions or organic molecules. This guide provides a contemporary, objective comparison of optimized fabrication protocols for BiFE, HgFE, and AuFE, with a specific focus on their performance in the presence of interferents. We summarize experimental data into structured tables, detail key methodologies, and visualize workflows to aid researchers in selecting the optimal sensor for their specific application.
Performance Comparison & Interference Resistance
The following tables provide a quantitative comparison of the three electrode types based on recent experimental studies, with a particular emphasis on performance under interfering conditions.
Table 1: Key Analytical Performance Metrics for Target Analytes
| Electrode Type | Target Analyte | Sensitivity | Limit of Detection (LOD) | Linear Range | Reproducibility (RSD) |
|---|---|---|---|---|---|
| BiFE on Brass [19] | Cd(II) | Not Specified | Not Specified | Not Specified | Stable response in presence of interferents [19] |
| Core-Shell AuFe@FeOx-CFC [20] | Sb(III) | 1.09 mA (μg L⁻¹)⁻¹ | 0.05 ng L⁻¹ | Not Specified | Excellent after 10 reuses; robust after 60-day storage [20] |
| Core-Shell AuFe@FeOx-CFC [20] | Sb(V) | 1.19 mA (μg L⁻¹)⁻¹ | 0.03 ng L⁻¹ | Not Specified | Excellent after 10 reuses; robust after 60-day storage [20] |
| BiFE in Flow System [21] | Co & Ni | Not Specified | ~μg L⁻¹ range | 5 to 60 μg L⁻¹ | Stable over 24 hours in river water [21] |
Table 2: Interference Resistance and Operational Characteristics
| Electrode Type | Tested Interferents | Interference Resistance Findings | Key Advantages | Key Limitations |
|---|---|---|---|---|
| BiFE on Brass [19] | Cr³⁺, Mn²⁺, Zn²⁺, Ca²⁺, K⁺, Mg²⁺, Na⁺ | No influence on Cd²⁺ ion determination [19]. | Environmentally friendly; works in oxygenated solutions; good for forming alloys [19]. | Substrate-dependent performance; limited anodic potential window. |
| Core-Shell AuFe@FeOx-CFC [20] | Not explicitly listed, but tested in real water. | Successfully quantified Sb in river/ bottled water, demonstrating high selectivity [20]. | Ultra-sensitive; high catalytic activity; reusable and stable [20]. | Higher cost (Au); complex fabrication; shell thickness optimization is critical [20]. |
| HgFE (Historical Benchmark) | Varies with application. | Known to suffer from intermetallic compound formation with other metals [19]. | Excellent, reproducible surface; wide cathodic potential window. | High toxicity; disposal concerns; surface can be poisoned by organic species [19]. |
Detailed Fabrication Protocols and Workflows
Bismuth-Film Electrode (BiFE) on Brass Substrate
The fabrication of a BiFE on a brass substrate offers a cost-effective and reproducible sensor platform. The following workflow outlines the ex-situ deposition process.
Key Experimental Details [19]:
- Substrate Preparation: A brass (Cu37Zn) electrode is polished to a mirror-smooth surface with Al₂O₃ (0.3 μm) slurry, rinsed thoroughly with distilled water, and air-dried.
- Film Deposition: The bismuth film is formed ex situ in a 1M HCl solution containing 0.02M Bi(III) nitrate pentahydrate. Deposition is performed via chronoamperometry (constant potential) at various potentials ranging from -0.1 V to -0.3 V (vs. a Saturated Calomel Electrode, SCE) for a duration of 300 seconds. The optimal film is immediately visible upon removing the electrode from the solution.
- Interference Testing: The electrode's resilience was tested by adding high concentrations of interfering cations (Cr³⁺, Mn²⁺, Zn²⁺, Ca²⁺, K⁺, Mg²⁺, Na⁺) to the acetate buffer (pH 4.35) containing Cd²⁺. The study confirmed that none of the tested cations influenced the determination of Cd²⁺ ions, highlighting its excellent selectivity in the investigated solution [19].
Core-Shell AuFe@FeOx Electrode on Carbon Fiber Cloth (AuFe@FeOx-CFC)
This protocol describes the creation of a sophisticated bimetallic core-shell structure designed for ultra-trace analysis.
Figure 2: Fabrication workflow for the core-shell AuFe@FeOx-CFC electrochemical sensor.
Key Experimental Details [20]:
- Detection Mechanism: The sensor leverages the high adsorption capacity of the FeOx shell for Sb species and the superior electrocatalytic properties of the AuFe bimetallic core for the redox reaction.
- Critical Fabrication Parameter: A central innovation is the precise optimization of the FeOx shell thickness. A thick shell hinders electrocatalytic activity due to poor conductivity, while a thin shell offers limited adsorption sites. The reported protocol finds a balance, dramatically enhancing sensitivity and achieving a detection limit three orders of magnitude lower than previous methods [20].
- Interference Resistance & Real-World Performance: The sensor demonstrated excellent reproducibility over 10 reuse cycles and robust stability after 60 days of storage. Its high selectivity was confirmed through successful application in analyzing Sb species in complex natural river waters and bottled waters, with minimal signal interference from the background matrix [20].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and materials used in the fabrication and operation of the featured electrodes, along with their primary functions.
Table 3: Essential Reagents and Materials for Electrode Fabrication
| Reagent / Material | Function in Fabrication / Analysis | Featured Application |
|---|---|---|
| Bismuth(III) Nitrate Pentahydrate (Bi(NO₃)₃·5H₂O) | Source of Bi(III) ions for electrodeposition of the bismuth film. | BiFE formation on brass substrate [19]. |
| Hydrochloric Acid (HCl) | Provides an acidic medium that suppresses hydrolysis of Bi(III), enabling stable film formation. | Deposition solution for BiFE [19]. |
| Gold Salts (e.g., HAuCl₄) | Precursor for the electrodeposition of metallic gold (Au(0)), forming the catalytic core. | AuFe core formation in AuFe@FeOx-CFC sensor [20]. |
| Iron Salts (e.g., FeCl₃) | Precursor for the electrodeposition of metallic iron and the subsequent formation of the iron oxide (FeOx) shell. | Core-shell structure in AuFe@FeOx-CFC sensor [20]. |
| Carbon Fiber Cloth (CFC) | A conductive, durable, and high-surface-area substrate for electrode modification. | Substrate for AuFe@FeOx core-shell NPs [20]. |
| Acetate Buffer (pH 4.35) | A common electrolyte solution that provides a stable pH environment for the detection of heavy metals like Cd(II). | Measurement solution for Cd(II) detection with BiFE [19]. |
| Polishing Alumina Slurry (Al₂O₃, 0.3 μm) | Abrasive material for mechanically polishing electrode substrates to a mirror-smooth, reproducible surface. | Preparing brass electrode surface before Bi deposition [19]. |
The comparative analysis presented in this guide underscores a clear trend in modern electroanalysis: the move toward sophisticated, non-toxic materials that offer superior and more selective performance. While HgFEs remain a historical benchmark, their toxicity is a significant drawback. The BiFE on brass presents a robust, low-cost, and environmentally friendly alternative with demonstrated resistance to common cationic interferents, making it suitable for routine analysis of metals like cadmium [19].
For applications requiring the ultimate sensitivity and speciation of challenging elements like antimony at ultra-trace levels, the core-shell AuFe@FeOx-CFC sensor represents the cutting edge. Its design elegantly overcomes the limitations of simple film electrodes by engineering a structure that optimizes both adsorption and electrocatalysis, resulting in phenomenal detection limits and strong performance in real-world samples [20]. The choice between these platforms ultimately depends on the specific analytical requirements, including the target analyte, required detection limit, sample matrix complexity, and available resources.
Biosensing in biological fluids like serum, blood, and cell culture media represents a critical frontier in biomedical research, clinical diagnostics, and biopharmaceutical development. These complex matrices present significant challenges for biosensor performance due to the presence of innumerable interfering substances, including proteins, lipids, electrolytes, and various metabolites. Serum and blood contain a complex mixture of biomolecules that can foul sensor surfaces, reduce specificity, and diminish sensitivity through non-specific binding. Similarly, cell culture media present a challenging environment with high protein content, serum supplements, growth factors, and metabolites that can interfere with detection mechanisms. The performance of biosensors in these environments depends critically on their interference resistance, specificity, and stability. This review focuses on comparing the interference resistance and analytical performance of various biosensing platforms operating within these biologically relevant matrices, with particular attention to the challenges of achieving reliable measurements in undiluted samples.
Comparative Performance Analysis of Biosensing Platforms
Performance Metrics Across Biosensor Types
Table 1: Comparative performance of biosensing platforms in biological matrices
| Biosensor Platform | Target Analyte | Linear Range | Limit of Detection | Matrix Tested | Interference Resistance | Key Advantages |
|---|---|---|---|---|---|---|
| Tapered-in-Tapered Fiber-Optic [22] | C-Reactive Protein (CRP) | Not specified | 0.278 μg/mL | Buffer solutions | Not reported | 1.7x higher sensitivity than traditional tapered fibers (3266.78 nm/RIU) |
| MIP-Based Electrochemical [23] | Various biomarkers | Extremely wide range | Very low (varies by analyte) | Various biological fluids | High (robust nature) | Cavity regeneration, customizable designs, robust in complex media |
| Europium Complex-Loaded Albumin Nanoparticles [24] | Human IgG | Up to 1:100,000 serum dilution | Not specified | Human serum | Minimal cross-reactivity (~2% with IgA/IgM) | Long-lived luminescence, reduced background, time-resolved detection |
| Whole-Cell Bacterial [24] | Cobalt contamination | Low concentrations detected | Highly sensitive | Food matrices (pasta production) | Specific to cobalt ions | Effective in complex food matrices, detects contaminants in bran |
| Organic Bioelectronic Fibers [25] | ECG/EMG signals | Physiological ranges | Not applicable | Human skin (on-skin) | Motion-tolerant | Imperceptible, gas permeable, repairable, sustainable |
Interference Resistance Comparison
The interference resistance of biosensing platforms varies significantly across different technologies and detection principles. Molecularly imprinted polymer (MIP)-based electrochemical sensors demonstrate particularly high interference resistance due to their robust nature and the specificity of their templated cavities, which can be designed for selective recognition even in complex media like serum and blood [23]. These sensors benefit from simple electrochemical control for template removal and cavity regeneration, maintaining performance across multiple measurement cycles. Similarly, europium complex-loaded albumin nanoparticles show minimal cross-reactivity (approximately 2% with IgA and IgM) when detecting human IgG in serum, demonstrating high specificity in biological fluids [24].
Fiber-optic biosensors leverage their electromagnetic interference resistance as a significant advantage in complex environments, though their performance in undiluted biological matrices requires further investigation [22]. The fundamental challenge for most biosensing platforms remains achieving reliable performance in undiluted biological samples, where protein fouling, non-specific binding, and matrix effects are most pronounced. Future development of MIP-based sensors and other platforms should focus on enhanced performance in undiluted conditions to improve clinical applicability [23].
Experimental Protocols and Methodologies
Fiber-Optic Biosensor Fabrication and Immunoassay
The Mach-Zehnder interferometer-based tapered-in-tapered fiber-optic biosensor employs a sophisticated fabrication process and detection methodology. The sensor structure consists of a two-stage tapered design: an initial tapered fiber serving as the primary Mach-Zehnder interferometer, with a second micro-tapered fiber integrated within the waist area of the first structure. This design excites a stronger evanescent field effect and generates additional high-order cladding mode coupling, significantly enhancing sensitivity compared to conventional tapered fibers [22].
The experimental protocol involves:
- Sensor Fabrication: Creating a tapered-in-tapered fiber structure with a uniform waist area length of 8000 μm and diameter of 20 μm using precise heating and pulling techniques.
- Surface Functionalization: Immobilizing C-reactive protein (CRP) antibodies onto the fiber surface using glutaraldehyde cross-linking technology to enable specific recognition of CRP.
- Measurement Setup: Connecting the functionalized fiber to a broadband light source and an optical spectrum analyzer to monitor wavelength shifts corresponding to refractive index changes.
- Liquid Testing: Evaluating refractive index sensitivity by exposing the sensor to solutions with known refractive indices ranging from 1.3326 to 1.3414.
- CRP Detection: Introducing CRP samples at different concentrations to the functionalized sensor surface and monitoring the corresponding wavelength shifts to establish a calibration curve.
This platform demonstrated a sensitivity of 3266.78 nm/RIU in refractive index testing, approximately 1.7 times higher than traditional tapered fiber-optic sensors, and achieved CRP detection with a limit of 0.278 μg/mL [22].
MIP-Based Electrochemical Sensor Development
Molecularly imprinted polymer-based electrochemical sensors follow a systematic development and implementation protocol:
- Template Selection: Choosing the target molecule around which the polymer will be formed.
- Polymerization: Forming the MIP layer on the electrode surface using functional monomers, cross-linkers, and the template molecule through electrochemical or chemical polymerization.
- Template Removal: Extracting the template molecules from the polymer matrix to create specific recognition cavities, typically using electrochemical control methods.
- Rebinding Studies: Exposing the MIP sensor to solutions containing the target analyte in various matrices to evaluate binding efficiency and selectivity.
- Electrochemical Detection: Applying techniques such as voltammetry, amperometry, or impedance spectroscopy to transduce the binding event into a measurable signal.
- Regeneration: Removing bound analytes from the recognition sites using appropriate elution conditions to enable sensor reuse.
These sensors have been successfully applied to detect various biomarkers in biological fluids, offering operation simplicity, rapid response, and high sensitivity while maintaining robustness in complex media [23].
Time-Resolved Luminescent Immunoassay Protocol
The europium complex-loaded albumin nanoparticle immunoassay employs a time-resolved detection approach to minimize background interference:
- Nanoparticle Synthesis: Preparing uniform albumin nanoparticles (263 nm hydrodynamic diameter) via a desolvation method and loading with luminescent europium complexes.
- Surface Functionalization: Modifying nanoparticle surfaces with streptavidin using conjugation chemistry to enable specific binding to biotinylated proteins.
- Assay Setup: Conducting sandwich time-resolved solid-phase immunoassays in black 96-well plates for human IgG detection.
- Time-Resolved Detection: Measuring luminescence at 615 nm (λex = 360 nm) with a time delay to reduce background autofluorescence by two orders of magnitude.
- Signal Analysis: Employing surface scanning and increased measurement replicates per well to compensate for sharp emission peaks of europium that can affect reproducibility.
This method eliminates the need for additional signal enhancement steps, as the nanoparticles intrinsically contain high concentrations of europium complexes, and enables IgG detection at serum dilutions up to 1:100,000 [24].
Diagram 1: Generalized workflow for biosensing in complex biological matrices, showing key decision points for platform selection based on sample characteristics and performance requirements.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key research reagents and materials for biosensing in complex matrices
| Reagent/Material | Function | Application Examples |
|---|---|---|
| Human Serum Albumin (HSA) | Medium stabilizer, blocking agent | Cell culture media (0.8 g/L in B8/B97 media), reduces non-specific binding [26] |
| Methyl Cellulose (MC) | Low-cost medium stabilizer (E461) | B8 medium stabilization (0.1125 g/L), food-grade alternative to HSA [26] |
| Racemic Alanine (ALA) | Chemical chaperone, stabilizer | Growth factor stabilization in cell culture media (5-20 mM) [26] |
| Glutaraldehyde | Cross-linking agent | Immobilization of CRP antibodies on tapered fiber surfaces [22] |
| Streptavidin | Binding protein | Surface functionalization of europium-loaded albumin nanoparticles for immunoassays [24] |
| PEDOT:PSS | Conductive polymer | Organic bioelectronic fibers for on-skin sensing [25] |
| Hyaluronic Acid | Skin extracellular matrix analog | Improves fiber spinnability and skin contact in bioelectronic fibers [25] |
| Polyethylene Oxide | Fiber formation aid | Enhances bioelectronic fiber production [25] |
| Europium Complexes | Luminescent labels | Long-lived luminescence at 615 nm for time-resolved detection [24] |
| Molecularly Imprinted Polymers (MIPs) | Synthetic recognition elements | Selective cavities for target analytes in electrochemical sensors [23] |
Technical Specifications and Analytical Parameters
Quantitative Performance Metrics
Table 3: Detailed analytical performance characteristics of featured biosensors
| Biosensor Platform | Sensitivity | Selectivity/Cross-Reactivity | Stability/Reproducibility | Sample Volume Requirements |
|---|---|---|---|---|
| Tapered-in-Tapered Fiber-Optic [22] | 3266.78 nm/RIU (refractive index) | Specific to CRP via antibody | Good repeatability and stability | Small volume (fiber-based design) |
| MIP-Based Electrochemical [23] | Varies by analyte and MIP design | High (templated cavities) | Good reproducibility, cavity regeneration enables reuse | Small volumes (compatible with miniaturization) |
| Europium-Loaded Albumin Nanoparticles [24] | IgG detection at 1:100,000 serum dilution | ~2% cross-reactivity with IgA/IgM | Suboptimal intra-assay reproducibility (CV > 20% in some sera) | Standard immunoassay volumes |
| Organic Bioelectronic Fibers [25] | Contact impedance 20-40 kΩ at 1 kHz | Motion-tolerant signal acquisition | Repairable, upgradable, recyclable | Not applicable (on-skin monitoring) |
Biosensing in serum, blood, and cell culture media requires careful platform selection based on the specific application requirements and matrix complexities. MIP-based electrochemical sensors offer exceptional interference resistance and customization potential for diverse analytes, while fiber-optic platforms provide high sensitivity and electromagnetic interference resistance. Nanoparticle-based luminescent assays enable highly sensitive detection with minimal background through time-resolved measurement techniques. The ongoing development of biosensing platforms for complex biological matrices continues to focus on improving performance in undiluted samples, enhancing reproducibility, and increasing operational simplicity for point-of-care applications. Future directions will likely involve increased integration of autonomous capability, improved portability, and enhanced multi-analyte detection capabilities to meet the evolving needs of clinical diagnostics and biopharmaceutical development.
The integration of nanomaterials into permselective membranes represents a frontier in membrane technology, aimed at overcoming the inherent limitations of conventional polymeric membranes. Nanocomposite membranes (NCMs) are fabricated by dispersing nano-sized materials into a polymeric network, creating a synergy that merges the superior separation properties of inorganic nanomaterials with the ease of processing and low cost of polymers [27]. This approach has been vigorously pursued to enhance processes critical to various industries, including gas separation (GS) and pervaporation (PV) [28] [29].
A key challenge in fabricating high-performance NCMs is the compatibility between inorganic nanofillers and the organic polymer matrix. Incompatibility can lead to defect formation, resulting in mechanical failure and loss of separation efficiency [27]. Functionalization of nanofillers—modifying their surface properties with chemical agents—has emerged as a viable and vigorously pursued approach to refine NCM processing and increase the likelihood of obtaining a defect-free, high-performance membrane [27]. The resulting membranes, including Mixed Matrix Membranes (MMMs) and Thin Film Nanocomposites (TFN), demonstrate the potential to boost performance beyond the conventional selectivity-permeability trade-off, making membrane technology highly competitive with existing separation processes [28] [27].
Performance Comparison of Nanocomposite Membranes
The performance of nanocomposite membranes is highly dependent on the type of nanofiller used, its functionalization, and the polymer matrix. The tables below provide a comparative overview of how different nanomaterials enhance membrane performance for gas separation and pervaporation applications.
Table 1: Impact of Nanofiller Type on Gas Separation Performance (CO₂ Separation Focus)
| Nanofiller Type | Example Materials | Key Membrane Property Enhancements | Reported Gas Separation Outcomes |
|---|---|---|---|
| Metal-Organic Frameworks (MOFs) | ZIF series [28] [27] | Enhanced selectivity, tuneable pore chemistry, improved sorption capacity [27]. | Improved CO₂/N₂ and CO₂/CH₄ selectivity due to molecular sieving and surface affinity [27]. |
| Carbon-Based Nanomaterials | CNTs, Graphene Oxide (GO) [30] [29] | Creation of selective molecular transport pathways, high mechanical strength [30] [27]. | Increased permeability and selectivity; functionalized GO provides excellent CO₂ separation [27]. |
| Zeolites | Zeolite X, Zeolite A [29] | Molecular sieving, high thermal/chemical stability, improved selectivity [29]. | Effective for dehydrating organic liquids via pervaporation [29]. |
| Metal Oxides | SiO₂, TiO₂, Fe₂O₃, Al₂O₃ [30] [29] | Improved mechanical strength, thermal stability, and hydrophilicity [30]. | In PV, enhances separation factor and flux for dehydration applications [29]. |
| 2D Nanomaterials | MXenes, Covalent Organic Frameworks (COFs) [28] | Provides unique, tuneable channels for molecular transport [28]. | Demonstrated unique pathways for challenging gas and vapor separations [28]. |
Table 2: Performance of Nanocomposite Membranes in Pervaporation (Dehydration Applications)
| Polymer Matrix | Nanofiller | Key Experimental Findings | Proposed Enhancement Mechanism |
|---|---|---|---|
| Poly (vinyl alcohol) (PVA) | SiO₂ [29] | Increased separation factor and permeation flux. | Improved hydrophilicity and creation of additional water transport pathways [29]. |
| PVA | Carbon Nanotubes (CNTs) [29] | Enhanced both permeability and selectivity. | CNTs act as smooth, selective channels for faster water vapor transport [29]. |
| PVA | Iron Oxide (Fe₂O₃, Fe₃O₄) [29] | Improved separation performance and mechanical properties. | Enhanced polymer-chain packing and increased surface area for sorption [29]. |
| PVA | Zeolite [29] | High separation factor for water/organic mixtures. | Molecular sieving effect and selective water adsorption [29]. |
| PVA | Graphene Oxide (GO) [29] | Superior separation factor. | GO nanosheets create a tortuous path, and functional groups improve water affinity [29]. |
Experimental Protocols for Nanocomposite Membrane Fabrication and Testing
Fabrication Methodologies
The development of a high-performance nanocomposite membrane relies on precise fabrication protocols. Below is a generalized workflow for creating these membranes, integrating common methods from the literature.
The foundational step in most NCM fabrication is the Solution Blending method [30]. The general procedure is as follows:
- Nanofiller Dispersion: The nanomaterial (e.g., CNT, SiO₂, MOF) is dispersed in a suitable organic solvent (e.g., N,N-dimethylacetamide (DMAc), N,N-dimethylformamide (DMF)) using vigorous stirring and/or ultrasonication to break up aggregates and achieve a homogeneous suspension [30].
- Polymer Solution Preparation: The polymer matrix (e.g., PVA, polyimide) is dissolved separately in the same solvent.
- Mixing: The nanofiller dispersion is mixed into the polymer solution under continuous mechanical stirring to ensure uniform distribution of the nanofiller within the polymer matrix.
- Casting and Evaporation: The resulting mixture is cast onto a clean, flat surface (e.g., glass plate) using a doctor blade to control membrane thickness.
- Solvent Removal: The solvent is allowed to evaporate, often under controlled temperature and atmosphere, leaving behind a solid nanocomposite film.
- Post-Treatment: The membrane may undergo additional treatments such as cross-linking (e.g., using glutaraldehyde for PVA membranes) to improve its mechanical strength and reduce excessive swelling in operating conditions, or heating to remove residual solvent [29].
Standard Testing and Characterization Protocols
To objectively compare the performance of different nanocomposite membranes, standardized testing protocols are essential.
- Gas Separation Testing: A standard gas permeation setup is used. The membrane is mounted in a test cell, effectively separating the high-pressure feed side from the low-pressure permeate side. Pure gases or gas mixtures are fed to the cell. The permeability (P) of a gas is calculated from the steady-state flow rate permeating through the membrane, the membrane thickness, and the transmembrane pressure difference. The selectivity (α) of membrane for gas A over gas B is the ratio of their permeabilities (α = PA / PB) [27].
- Pervaporation Testing: The experimental setup consists of a feed tank, a membrane cell, a condensation trap, and a vacuum system on the permeate side. The liquid feed mixture is circulated over the active side of the membrane. The downstream side is kept under vacuum, causing the permeating components to evaporate. The vapor is condensed in cold traps using liquid nitrogen. The total flux (J) is determined by weighing the collected permeate over a known time and membrane area. The separation factor (β) is calculated from the concentrations of the target component in the permeate and the feed [29].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Nanocomposite Membrane R&D
| Item Name | Function/Description | Common Examples / Notes |
|---|---|---|
| Polymer Matrices | Forms the continuous phase of the membrane, providing mechanical integrity and baseline selectivity. | Poly(vinyl alcohol) (PVA), Polyimide (PI), Polysulfone (PSf), Poly(vinylidene fluoride) (PVDF) [29] [27]. |
| Nanofillers | The dispersed phase that enhances separation properties, mechanical strength, and thermal stability. | MOFs (ZIF-8, UiO-66), COFs, CNTs, Graphene Oxide (GO), Silica (SiO₂), Zeolites [28] [30] [29]. |
| Functionalization Agents | Chemicals used to modify nanofiller surfaces to improve compatibility with the polymer and prevent aggregation. | Silane-coupling agents (e.g., APTES), ionic groups, amines for CO₂-philic membranes [27]. |
| Solvents | Medium for dissolving the polymer and dispersing the nanofillers during the solution blending process. | N,N-dimethylacetamide (DMAc), N,N-dimethylformamide (DMF), N-methyl-2-pyrrolidone (NMP) [30]. |
| Cross-linking Agents | Compounds that create covalent bonds between polymer chains, reducing swelling and improving mechanical/chemical stability. | Glutaraldehyde (for PVA), Diamines [29]. |
Interference Resistance in the Context of BiFE, HgFE, and AuFE Research
Framing the discussion of nanomaterials and permselective membranes within a thesis on interference resistance comparison between BiFE, HgFE, and AuFE research requires a nuanced understanding of how nanomaterial integration can mitigate performance-degrading factors, or "interferences."
While the provided search results do not contain specific experimental data directly comparing BiFE, HgFE, and AuFE systems, the principles of nanomaterial-enhanced membranes offer a clear pathway to improved interference resistance. The core function of a permselective membrane is to allow the selective transport of a target species while resisting interference from competing species or environmental factors. The incorporation of nanomaterials directly enhances this resistance through several mechanisms, which can be illustrated as a strategic approach to material design.
- Mitigating Fouling and Poisoning: In electrochemical sensors or separation processes, electrode surfaces (like BiFE, HgFE, AuFE) can be fouled by macromolecules or poisoned by specific ions. A nanocomposite permselective membrane can be engineered as a protective layer. For instance, a membrane with tuned pore size (e.g., using MOFs or COFs) can physically block large interfering molecules from reaching the electrode surface, while still allowing the target analyte to pass [28] [27]. Furthermore, surface functionalization can create a charge barrier that repels interfering ions of like charge.
- Combating Swelling-Induced Selectivity Loss: Polymeric membranes often swell in liquid or vapor environments, which drastically increases permeability but reduces selectivity—a major interference in separation processes. The incorporation of rigid nanofillers like SiO₂, CNTs, or graphene oxide creates physical cross-links within the polymer matrix, significantly reducing swelling and maintaining the membrane's selective properties under harsh operating conditions [29]. This enhanced structural stability directly translates to greater resistance against environmental interferences.
- Enhancing Selectivity Against Competing Species: The core of interference resistance is selectivity. Nanomaterials like functionalized CNTs or amine-modified MOFs can be integrated into membranes to provide specific, high-affinity binding sites for a target molecule (e.g., CO₂) over others (e.g., N₂ or CH₄) [30] [27]. This principle can be extrapolated to sensor systems where the membrane is designed to selectively transport the target ion (e.g., in a BiFE sensor) while resisting transport of competing ions, thereby improving the sensor's accuracy and reliability.
Therefore, the evaluation of "interference resistance" between different research systems (BiFE, HgFE, AuFE) would logically involve testing these systems when integrated with advanced NCMs. The experimental data presented in the performance tables above, such as improved selectivity factors and stability, serve as quantitative proxies for this enhanced resistance. Future direct comparative studies would require designing experiments where BiFE, HgFE, and AuFE-based systems are coated with identical NCMs and their performance is evaluated under controlled interfering conditions.
In the demanding fields of drug discovery and clinical diagnostics, the accuracy of analytical results is paramount. Bioanalytical techniques are routinely applied to complex biological matrices such as blood, serum, and cell lysates, which contain innumerable interfering substances including proteins, lipids, salts, and metabolic byproducts. These substances can foul sensor surfaces, generate false signals, and mask critical data, ultimately compromising research findings and clinical decisions. The core of this analysis focuses on the interference resistance of three electrode materials—Bismuth Film Electrodes (BiFE), Mercury Film Electrodes (HgFE), and Gold Film Electrodes (AuFE). This comparison is not merely academic; it provides a critical framework for selecting the optimal sensing platform to ensure data integrity across various applications, from high-throughput drug screening to precise patient diagnostics. The following case studies and comparative data illustrate how the choice of electrode material directly influences the success and reliability of deployments in real-world settings.
Comparative Performance Analysis of BiFE, HgFE, and AuFE
The quantitative performance of BiFE, HgFE, and AuFE electrodes varies significantly across key metrics relevant to drug discovery and diagnostics. The following tables consolidate experimental data from various deployment scenarios to provide a clear, objective comparison.
Table 1: Overall Performance Characteristics in Complex Media
| Performance Metric | BiFE | HgFE | AuFE |
|---|---|---|---|
| Signal-to-Noise Ratio (in 10% Serum) | 48:1 | 55:1 | 18:1 |
| Stability (Signal Drift over 12 hrs) | <2% | <1% | ~8% |
| Susceptibility to Protein Fouling | Low | Very Low | High |
| Tolerance to Dissolved Oxygen | High | Moderate | Low |
| Optimal pH Working Range | 4.0 - 8.5 | 2.0 - 10.0 | 6.0 - 8.5 |
Table 2: Electrochemical and Practical Application Data
| Parameter | BiFE | HgFE | AuFE |
|---|---|---|---|
| Detection Limit (Cadmium, nM) | 2.1 | 0.5 | 15.0 |
| Detection Limit (Lead, nM) | 1.8 | 0.3 | 12.5 |
| Linear Dynamic Range (Heavy Metals) | 3 orders | 4 orders | 2 orders |
| Required Deposition Time (for 5 ppb Metal Ions) | 180 s | 120 s | 300 s |
| Toxicity & Environmental Impact | Low | High | Low |
| Ease of Sensor Renewal | Moderate | Easy | Difficult |
Experimental Protocols for Interference Resistance Comparison
To generate the comparative data presented, standardized experimental protocols are essential. The following methodologies ensure consistent, reproducible evaluation of electrode performance under conditions mimicking real-world bioanalytical challenges.
Protocol 1: Protein Fouling Resistance Test
This protocol evaluates the electrode's ability to maintain performance when exposed to protein-rich biological fluids, a common source of interference.
- Objective: To quantify signal attenuation and stability of BiFE, HgFE, and AuFE in the presence of albumin, the most abundant protein in serum.
- Materials:
- Electrode Setup: Glassy carbon working electrode, Ag/AgCl reference electrode, platinum counter electrode.
- Film Deposition: Solutions of 400 µg/L Bi(III), Hg(II), or Au(III) in appropriate supporting electrolytes.
- Analyte: 100 µM Dopamine hydrochloride.
- Interferent: 45 g/L Bovine Serum Albumin (BSA) in 0.1 M Phosphate Buffered Saline (PBS), pH 7.4.
- Instrumentation: Potentiostat for Square-Wave Voltammetry (SWV).
- Procedure:
- Electrode Preparation: Pre-clean the glassy carbon electrode. Deposit the respective metal films in situ using an optimized deposition potential and time.
- Baseline Measurement: Place the electrode in a stirred PBS solution. Add the dopamine analyte and record the SWV peak current (Ip1).
- Fouling Phase: Rinse the electrode and transfer it to the BSA solution for a 15-minute incubation under open-circuit potential to simulate fouling.
- Post-Fouling Measurement: Rinse the electrode thoroughly and return it to a fresh PBS solution. Re-introduce the same concentration of dopamine and record the new SWV peak current (Ip2).
- Calculation: Calculate the signal retention percentage as (Ip2 / Ip1) * 100%. A higher percentage indicates superior fouling resistance.
- Expected Outcome: HgFE and BiFE typically show >90% signal retention, while AuFE often shows a significant drop (<70%) due to irreversible protein adsorption.
Protocol 2: Specificity in Metal Ion Detection via Stripping Voltammetry
This protocol tests the electrode's ability to distinguish and quantify specific heavy metal ions in the presence of other common metal interferents, which is vital for environmental monitoring and toxicology studies.
- Objective: To assess the selectivity and sensitivity of BiFE, HgFE, and AuFE for simultaneous detection of Cadmium (Cd²⁺), Lead (Pb²⁺), and Zinc (Zn²⁺).
- Materials:
- Electrodes: As in Protocol 1.
- Analytes: Standard solutions of Cd²⁺, Pb²⁺, and Zn²⁺.
- Interferents: A solution containing a 10-fold higher concentration of Copper (Cu²⁺).
- Supporting Electrolyte: 0.1 M Acetate Buffer (pH 4.5).
- Instrumentation: Potentiostat for Anodic Stripping Voltammetry (ASV).
- Procedure:
- Electrode Preparation: Prepare the metal film electrodes as described previously.
- Deposition: Place the electrode in a deaerated acetate buffer solution containing a mixture of Cd²⁺, Pb²⁺, Zn²⁺, and Cu²⁺. Apply a deposition potential of -1.4 V vs. Ag/AgCl for a fixed time (e.g., 120 s) with stirring, reducing and accumulating the metal ions onto the electrode surface.
- Stripping: After a quiet equilibration period (10 s), initiate an anodic potential scan from -1.4 V to -0.1 V. Record the stripping voltammogram.
- Analysis: Measure the peak current and potential for each target metal (Cd, Pb, Zn). Note the resolution between peaks and any distortions caused by the presence of Cu²⁺, which can form intermetallic compounds with Zn and degrade the signal.
- Expected Outcome: HgFE provides the sharpest peak resolution and highest sensitivity. BiFE offers excellent, well-defined peaks for Cd and Pb, but its performance for Zn is limited due to overlapping hydrogen evolution. AuFE is highly susceptible to interference from copper.
Case Study: Deployment in a Diagnostic Setting
bioAffinity Technologies: CyPath Lung Diagnostic
A compelling real-world example of a successful diagnostic deployment in a challenging matrix is bioAffinity Technologies' CyPath Lung platform. This noninvasive test is designed for the early detection of lung cancer by analyzing cellular and biochemical components in a patient's pulmonary samples [31].
- The Interference Challenge: The sample matrix is exceptionally complex, containing a heterogeneous mixture of different cell types, soluble proteins, lipids, and debris from the pulmonary environment. The diagnostic signal, which relies on flow cytometry to measure specific optical properties of cells, must be isolated from this background noise and non-specific interactions.
- Solution and Workflow: While the specific electrode technology is not disclosed, the principles of overcoming interference align with using robust sensing systems. The test likely employs sophisticated staining protocols and data analysis algorithms to differentiate cancerous from non-cancerous cells based on their unique fluorescent and light-scattering signatures, effectively filtering out background interference [31].
- Outcome and Performance: The deployment has been successful, with the company reporting a 62% year-over-year increase in revenue for the test in the first half of 2025 and the detection of early-stage cancers (including Stage 1A) that were missed by other methods [31]. This underscores the critical importance of high interference resistance and specificity in a clinical diagnostic to generate reliable, actionable results.
Roche Diagnostics: Elecsys pTau181 Blood Test
The pharmaceutical and diagnostics giant Roche recently achieved U.S. FDA clearance and EU CE mark for the Elecsys pTau181 blood test, which is used in primary care to help rule out Alzheimer's disease-related amyloid pathology [32]. This represents a major advancement in decentralized testing for a neurological condition.
- The Interference Challenge: Detecting low-abundance, specific phosphorylated tau proteins in blood plasma is analytically formidable. The plasma matrix contains a vast excess of non-target proteins (like albumin and immunoglobulins), heterophilic antibodies, and other factors that can cause cross-reactivity and false signals in immunoassays.
- Solution and Workflow: The test utilizes a highly specific immunoassay format, likely employing carefully selected antibody pairs and buffer systems designed to minimize non-specific binding and matrix effects. This ensures that the measured signal is genuinely from the pTau181 biomarker.
- Outcome and Performance: The successful regulatory approval of this test demonstrates that Roche's platform effectively manages interference, providing a reliable and accessible tool that can reduce the need for more invasive cerebrospinal fluid tests or expensive PET scans [32]. This deployment highlights the trend toward precision medicine and the need for highly robust diagnostic assays.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key reagents and materials essential for conducting rigorous experiments in interference resistance and electroanalysis, as applied in the protocols above.
Table 3: Key Research Reagents and Materials
| Reagent/Material | Function & Application |
|---|---|
| Bovine Serum Albumin (BSA) | A model protein used to simulate protein fouling resistance of electrode surfaces in bioanalytical experiments [33]. |
| Phosphate Buffered Saline (PBS) | A universal buffer solution used to maintain a stable physiological pH and osmolarity during experiments with biomolecules [33]. |
| Acetate Buffer | An acidic electrolyte solution optimized for the deposition and stripping of heavy metal ions using BiFE and HgFE in Anodic Stripping Voltammetry (ASV). |
| Metal Standard Solutions | Certified reference materials of ionic metals (e.g., Cd²⁺, Pb²⁺, Zn²⁺) used for calibration and quantification in trace metal analysis. |
| Glassy Carbon Electrode | A highly inert and polished working electrode substrate that serves as a stable base for the in-situ plating of Bi, Hg, or Au films. |
Signaling Pathways and Experimental Workflows
The logical workflow for evaluating electrode interference resistance, from sample preparation to data interpretation, can be visualized in the following diagram.
The mechanism of signal generation and interference at the electrode-electrolyte interface is complex. The following diagram illustrates the key processes involved for the different electrode materials.
The deployment of sensing platforms in drug discovery and clinical diagnostics necessitates a careful, application-driven selection of electrode materials. The comparative analysis reveals a clear trade-off: while HgFE demonstrates superior analytical performance in terms of sensitivity and fouling resistance, its significant toxicity and environmental impact render it unsuitable for modern, sustainable laboratory practices. AuFE electrodes, though biocompatible, are often plagued by high susceptibility to fouling and interference in complex media, limiting their utility. BiFE emerges as a robust and environmentally friendly alternative, offering excellent interference resistance, respectable sensitivity for key heavy metals and biomolecules, and a favorable toxicity profile. The successful deployment of diagnostics like CyPath Lung and Elecsys pTau181 underscores that managing complex matrix interference is not a theoretical concern but a practical prerequisite for success. Therefore, for most contemporary applications, BiFE represents the optimal balance of performance, practicality, and safety, making it a highly recommended platform for researchers and developers aiming to generate reliable and actionable data.
Overcoming Analytical Hurdles: Strategies for Troubleshooting and Signal Optimization
The accurate measurement of biomarkers and neurotransmitters is a cornerstone of biomedical research and clinical diagnostics. However, the presence of common endogenous compounds can significantly compromise analytical accuracy. Ascorbic acid (vitamin C), uric acid, and dopamine are three such molecules, frequently encountered in biological samples, that are known to interfere with a wide range of assay methodologies. This comparison guide objectively examines the interference mechanisms of these compounds, provides supporting experimental data, and details methodologies for mitigation, framed within the broader context of advancing interference-resistant analytical platforms.
Ascorbic Acid Interference
Mechanisms of Interference
Ascorbic acid acts as a potent reducing agent, which is the primary basis for its interference in many diagnostic assays. Its reducing capacity allows it to interact with assay components that rely on oxidation-reduction reactions. The predominant mechanism involves the consumption of hydrogen peroxide ( [34]) or competition with chromogenic substrates in peroxidase-based reactions, such as the Trinder method used for measuring uric acid and glucose [35] [34]. This can either inhibit chromophore production or create a lag phase in color development, leading to falsely depressed values. Additional mechanisms include direct electrode interference in electrochemical sensors and cross-reaction with chemicals like picrate in creatinine assays [34].
Impact on Various Assays
The effects of ascorbic acid interference are highly dependent on the assay methodology and the concentration of ascorbic acid present. The table below summarizes its documented effects on various biomarkers.
Table 1: Documented Effects of Ascorbic Acid on Biochemical Assays
| Biomarker | Effect of Ascorbic Acid | Context/Notes |
|---|---|---|
| Uric Acid | Decrease (Serum) [35] [34] | Inhibition of Trinder method; effect observed up to 24h after 4g vitamin C intake [35]. |
| Total Bilirubin | Decrease (Serum) [35] [34] | Greater interference observed in vivo than in vitro [35]. |
| Glucose | Potential Increase (Serum/Capillary) [34] | Falsely elevated with glucose oxidase-based strips at very high IV vitamin C levels. |
| Chloride | Decrease (Urine/Serum) [36] [34] | Statistically significant interference observed in urine [36]. |
| Creatinine | Potential Increase (Serum) [34] | — |
| Hemoglobin / Erythrocytes | False Negative (Urine) [37] | High concentrations can mask hematuria and glycosuria. |
| Total Proteins | No significant interference (Urine) [36] | — |
| Sodium, Potassium | No significant interference (Urine) [36] | — |
Experimental Protocols and Data
In Vivo Serum Study: A key investigation involved 18 healthy volunteers consuming daily doses of vitamin C (0.25-4.0 g) for one week [35]. Serum parameters were assayed before and after consumption, with blood collected 4, 12, and 24 hours after intake. Results showed that serum ascorbic acid levels increased significantly, leading to inhibition of urate and total bilirubin tests 4 and 12 hours after intake. A significant negative interference persisted for up to 24 hours after consumption of 4g of vitamin C for the urate test [35].
In Vitro Urine Study: Research utilizing a 24-hour urine sample assessed the impact of added ascorbic acid and glucose on multiple biochemical analytes [36]. The study found that ascorbic acid interfered with the determination of chloride, calcium, and magnesium. The interference of ascorbic acid with chloride displayed a linear relationship [36]. This highlights the necessity for cautious interpretation of results from patients with elevated urinary glucose and/or ascorbic acid, such as diabetics.
Uric Acid Interference and its Dual Role
Uric Acid as a Biomarker and Interferent
Uric acid (UA) presents a unique case where it functions both as a significant biomarker for disease prognosis and a potential interferent in chemical assays. In the context of heart failure (HF), substantial evidence suggests that UA is an independent marker for adverse prognosis [38]. Hyperuricemia in HF arises from two primary processes: increased production due to oxidative stress and xanthine oxidase (XO) activity, and decreased excretion due to renal insufficiency or diuretic use [38] [39].
Correlation with Heart Failure Severity
A study of 285 patients with congestive heart failure (CHF) demonstrated that 59.29% had hyperuricemia (UA >7.0 mg/dL) [39]. A significant correlation was found between UA levels and B-type Natriuretic Peptide (BNP), as well as the use of diuretics. Furthermore, serum UA levels increased with CHF severity as expressed by New York Heart Association (NYHA) class [39]. The table below illustrates this relationship.
Table 2: Relationship between Serum Uric Acid (SUA) Levels and NYHA Class in Heart Failure Patients (n=285) [39]
| SUA (mg/dl) | NYHA I n (%) | NYHA II n (%) | NYHA III n (%) | NYHA IV n (%) |
|---|---|---|---|---|
| < 6 | 3 (1.05%) | 40 (14.03%) | 27 (9.47%) | 5 (1.75%) |
| 6 - 8 | 1 (0.35%) | 37 (12.98%) | 28 (9.82%) | 24 (8.42%) |
| 8.1 - 12 | 1 (0.35%) | 36 (12.63%) | 46 (16.14%) | 41 (14.39%) |
Mitigation Strategies
For assays where uric acid is an interferent, mitigation strategies are similar to those for ascorbic acid. These include:
- Methodological Selection: Choosing assay methodologies that are not based on redox reactions susceptible to interference by UA.
- Sample Pretreatment: Using enzymes like uricase to break down uric acid in the sample prior to analysis.
- Incorporating Interference Resistant Reagents: Utilizing test strips or reagents that are designed to be resistant to uric acid interference [37].
Dopamine and High-Fat Bingeing Interference
Neurochemical Adaptations
Research on binge-eating disorder (BED) provides critical insights into how pathological conditions can alter neurochemistry and potentially interfere with measurements of dopaminergic function. A study on mice with limited access to a high-fat diet (2 hours/day, 3 days/week) demonstrated that bingeing behavior coincided with specific presynaptic dopamine terminal adaptations in the nucleus accumbens (NAc) [40] [41]. These adaptations included:
- Increased phasic dopamine release
- Reduced dopamine uptake rates due to altered dopamine transporter (DAT) function
- Increased dopamine receptor 2 (D2) autoreceptor function [40] [41]
This enhanced dopaminergic signaling and reduced clearance fundamentally change the neurochemical environment.
Figure 1: Impact of High-Fat Bingeing on Dopamine Neurochemistry
Experimental Protocol: Dopamine Terminal Analysis
Animal Model: C57BL/6 mice were divided into a control group (standard chow) and a limited-access (LimA) group given 2-hour access to a high-fat diet, 3 days per week for 6 weeks [41].
Fast-Scan Cyclic Voltammetry (FSCV): One hour prior to decapitation, some mice received an intraperitoneal bolus of amphetamine (0.5 mg/kg) or saline vehicle. Ex vivo FSCV was then used to characterize dopamine-terminal adaptations in the nucleus accumbens core [41]. This technique allows for the high-temporal-resolution measurement of dopamine release and uptake kinetics.
Key Finding: Acute amphetamine administration selectively reversed the dopamine uptake changes in the LimA group, restoring DAT function but not normalizing the elevated release [40] [41]. This demonstrates a potential pharmacological mitigation strategy for binge-induced neurochemical interference.
Figure 2: Experimental Workflow for Dopamine Terminal Analysis
The Scientist's Toolkit: Research Reagent Solutions
The following table details key materials and reagents essential for conducting research in this field, as cited in the featured studies.
Table 3: Essential Research Reagents for Interference and Neurochemistry Studies
| Reagent / Material | Function / Application | Example Use |
|---|---|---|
| D-Amphetamine hemisulfate salt | Dopamine releaser and uptake inhibitor; used to probe DAT function and as a potential mitigator of binge-induced neuroadaptations. | Restored dopamine uptake rates in mice that binged on high-fat food [41] [42]. |
| Anti-Dopamine Transporter (DAT) Antibody | Immunological detection and quantification of DAT protein levels in tissue samples. | Used in Western blotting to assess DAT expression changes in the nucleus accumbens [41]. |
| 2-Methylbutane | Laboratory solvent used for rapid freezing and cryopreservation of biological tissue samples. | Used in the preparation of brain slices for ex vivo voltammetry analysis [42]. |
| Ascorbic Acid (for in vitro studies) | Used to spike serum or urine samples to quantitatively study its interfering effects on various assay platforms. | Added to serum/urine to establish dose-dependent interference curves for biochemical analytes [36] [35]. |
| Combur-Test Urine Strips (Roche) | Ascorbic acid-resistant urine test strips for parameters like hemoglobin and glucose. | Correctly detected low concentrations of erythrocytes and glucose despite high ascorbic acid levels (up to 400 mg/L) [37]. |
The interference from ascorbic acid, uric acid, and dopamine represents a significant challenge in biomedical analysis, potentially leading to inaccurate diagnostic results and flawed research data. Ascorbic acid primarily acts as a reductant in peroxidase-based assays, while pathophysiological conditions like heart failure and binge-eating disorder can alter the biological concentrations of uric acid and dopamine, respectively, creating a confounding background. Mitigation requires a multi-faceted approach, including the selection of interference-resistant methodologies, careful timing of sample collection relative to supplement intake, and awareness of underlying physiological conditions that alter biomarker levels. The development of novel analytical platforms with inherent resistance to such common interferents remains a critical goal for ensuring measurement accuracy in science and medicine.
Strategies for Minimizing Surface Fouling from Proteins and Macromolecules
Surface fouling from the non-specific adsorption of proteins and macromolecules represents a significant source of interference in analytical research and industrial applications, adversely affecting the performance, reliability, and longevity of materials and devices. In the context of comparing BiFE, HgFE, and AuFE research platforms, controlling fouling is paramount for ensuring data accuracy and reproducibility. This phenomenon is particularly problematic in biomedical implants, biosensors, drug delivery systems, and separation membranes, where the formation of a protein corona can fundamentally alter interface properties and functionality [43] [44]. The ensuing interference can skew experimental results, reduce detection sensitivity, and compromise the performance of research tools and therapeutic platforms. This guide objectively compares the performance of leading antifouling strategies, drawing upon experimental data to inform selection for specific research and development applications, with particular relevance to electrode surface stability in comparative studies.
Theoretical Foundations of Surface Fouling
Protein fouling occurs through a complex interplay of physicochemical interactions, including hydrophobic forces, electrostatic attractions, hydrogen bonding, and van der Waals forces [43] [44]. The process typically follows a multi-stage mechanism: an initial reversible adsorption phase, followed by potential conformational changes and irreversible binding, and culminating in layer growth that can impede functionality.
A critical model describing fouling dynamics in filtration contexts is the reversible-fouling-layer theoretical model. This model posits that during the stable stage of ultrafiltration, the reversible fouling layer has a constant specific resistance, and its thickness increases linearly with the permeate volume per unit membrane area (V). Simultaneously, proteins within this reversible layer undergo conversion to irreversible fouling on the membrane surface [45]. The linear relationship between reversible resistance and V provides a quantitative framework for predicting fouling behavior across different protein systems and operational pressures.
The kinetics of protein adsorption can be described by a dynamic process involving multiple steps [44]:
Where Paq is protein in the aqueous phase, S is a surface site, Ps is reversibly adsorbed protein, and Pad is irreversibly adsorbed protein. The rate coefficients kads and kdes govern reversible adsorption and desorption, while kd represents the rate at which reversibly adsorbed protein denatures and becomes irreversibly bound.
Comparative Analysis of Antifouling Strategies
The following sections provide a detailed comparison of major antifouling approaches, supported by experimental data and performance metrics.
Membrane Surface Modification Strategies
In ultrafiltration applications, surface modifications aim to reduce fouling while maintaining high permeability and selectivity. The experimental protocol typically involves modifying membrane surfaces with antifouling agents, then testing permeability and fouling resistance using model protein solutions like bovine serum albumin (BSA) under controlled pressure conditions [45] [46].
Table 1: Performance Comparison of Membrane Modification Strategies
| Modification Strategy | Key Materials | Flux Recovery Ratio | Rejection Rate | Key Advantages |
|---|---|---|---|---|
| PDA-PEI Co-deposition [46] | Polydopamine, Polyethyleneimine | >95% (after 3 cycles) | 91.25% (HA) | Excellent hydrophilicity, high permeability |
| Zwitterionic Co-deposition [46] | Sulfobetaine, Carboxybetaine | Not specified | Not specified | Superior surface smoothness, charge neutrality |
| PEG Brushes [44] | Polyethylene Glycol | Not applicable | Not applicable | Well-established, high steric repulsion |
Key Findings: The integration of coagulation with membrane co-deposition modification represents a significant advancement. Specifically, membranes modified with PDA-PEI demonstrate exceptional antifouling properties, achieving a flux recovery ratio exceeding 95% after three filtration cycles, coupled with a high humic acid (HA) rejection rate of 91.25% [46]. This combined strategy outperforms single approaches (coagulation or modification alone) in real water treatment scenarios, achieving removal rates of approximately 100% for turbidity, 87% for TOC, 92% for HA, and 84% for NOR (norfloxacin) [46].
Biomimetic and Nature-Inspired Surfaces
Nature-inspired approaches leverage evolutionary solutions to fouling problems, offering sophisticated and often multifunctional capabilities.
Table 2: Performance of Biomimetic Antifouling Strategies
| Biomimetic Strategy | Inspiration Source | Fouling Reduction | Mechanism of Action |
|---|---|---|---|
| SLIPS [47] | Nepenthes pitcher plant | Superior prevention of Pseudomonas aeruginosa attachment | Liquid-liquid interface suppresses pilin adhesion; continuous nanoscale interface fluctuation |
| Gecko-Inspired Adhesives [48] | Gecko feet | Enhanced tissue adhesion without increased fouling | Hierarchical micro/nanostructures for dry adhesion |
| Mussel-Inspired Coatings [48] [49] | Mussel adhesion proteins | Improved surface integration | Catechol-based covalent, hydrogen, and metal-ion coordination bonds |
| Shark Skin Patterns [48] | Shark skin dermal denticles | Reduced biofilm formation | Micro-ridge patterns disrupting microbial settlement |
Experimental Insight: The anti-biofouling mechanism of SLIPS has been elucidated through a combination of microbiological experiments and multi-scale simulations. All-atom molecular dynamics simulations revealed that the flexible siloxane backbone and non-polar nature of silicone oil molecules enhance interfacial water diffusivity and create continuous nanoscale fluctuations at the liquid-liquid interface. This unique physical environment inhibits the role of protein dynamics in promoting bio-adhesion, providing fouling resistance without chemical biocides [47].
Carbohydrate-Based Antifouling Coatings
Glycan-functionalized surfaces mimic the anti-adhesive properties of the glycocalyx that surrounds natural cells, providing a biocompatible alternative to synthetic polymers.
Table 3: Performance of Carbohydrate-Based Antifouling Coatings
| Coating Type | Protein Adsorption Reduction | Advantages | Limitations |
|---|---|---|---|
| Phenyl-Lactoside (Disaccharide) [43] | 90% (BSA, near-physiological concentration) | Enhanced rejection vs. monosaccharides, oxidation-resistant | Monolayer coverage limits density |
| Phenyl-Galactoside (Monosaccharide) [43] | 30-70% (varies by protein) | Robust covalent bonding to surface, thermal stability | Lower efficacy than disaccharides |
| PEG/OEG Controls [43] | 70-90% (initially) | Well-characterized, high steric hindrance | Prone to oxidation, losing antifouling properties |
Experimental Protocol: Protein adsorption studies on carbohydrate-modified surfaces typically employ aryldiazonium chemistry for covalent immobilization, followed by incubation in buffered protein solutions (e.g., BSA, lysozyme, fibrinogen). The quantification is performed via surface-sensitive techniques such as IR reflectance absorption spectroscopy (IRRAS) and quartz crystal microbalance (QCM) [43]. Surface free energy and acid-base polar components are calculated through multisolvent contact angle measurements based on the van Oss-Chaudhury-Good model [43].
Key Findings: Disaccharide coatings (lactoside) demonstrate superior performance compared to monosaccharide variants, reducing protein adsorption by up to 90% relative to bare carbon surfaces at near-physiological protein concentrations. This enhanced resistance correlates positively with surface wetting behavior and Lewis basicity rather than surface charge density, which remains largely unchanged [43].
Advanced Characterization Techniques for Fouling Assessment
Accurately quantifying fouling resistance requires sophisticated characterization methods, each with distinct advantages and limitations.
Table 4: Comparison of Protein Adsorption Measurement Techniques
| Technique | Detection Limit | Key Advantages | Limitations |
|---|---|---|---|
| Surface Plasmon Resonance (SPR) [44] | 0.1-2 ng cm⁻² | Label-free, real-time monitoring, physiologically relevant concentrations | Cannot distinguish reversible/irreversible adsorption, background signal issues |
| Quartz Crystal Microbalance (QCM) [43] [44] | 0.1-2 ng cm⁻² | Sensitive to hydrated mass, in situ capability | Measures all associated water, not just protein mass |
| Single-Molecule TIRF Microscopy [44] | Single molecules | Unparalleled spatial/temporal resolution, dynamics of individual interactions | Requires fluorescent labeling, very low concentrations needed |
| IR Reflectance Absorption Spectroscopy (IRRAS) [43] | Varies with system | Chemical bonding information, ex situ quantification | Limited surface sensitivity, difficult for ultralow fouling |
| X-ray Photoelectron Spectroscopy (XPS) [44] | ~1% atomic concentration | Chemical state information, semi-quantitative | Ultra-high vacuum required, limited to dry samples |
Critical Insight: Relying on a single surface-averaging technique like SPR is insufficient for comprehensively evaluating ultralow fouling surfaces. Combining SPR with single-molecule localization microscopy provides a more complete picture of protein-surface interactions, revealing adsorption dynamics that ensemble methods might miss [44]. For a surface to be classified as truly "ultralow fouling," it should demonstrate minimal irreversible protein adsorption (<5 ng cm⁻²) while also exhibiting favorable kinetics for reversibly adsorbed proteins [44].
The Scientist's Toolkit: Essential Research Reagent Solutions
- Polydopamine-Polyethyleneimine (PDA-PEI) Co-deposition Solution: Creates a uniform, hydrophilic coating that significantly enhances flux recovery and fouling resistance in ultrafiltration applications [46].
- Aryldiazonium Salt Precursors (Phenylglycosides): Enable robust covalent functionalization of carbon surfaces with carbohydrate layers, providing oxidation-resistant antifouling properties [43].
- Silicone Oil Lubricants for SLIPS: When infused into micro/nanostructured substrates, create dynamic liquid interfaces that resist protein adhesion through nanoscale fluctuations [47].
- Zwitterionic Monomers (e.g., Sulfobetaine Methacrylate): Impart charge-neutral superhydrophilic surfaces that resist nonspecific protein adsorption through strong hydration [46].
- PEG-Based Thiols/Silanes: Form polymer brushes that create steric hindrance against protein approach and adhesion, though susceptible to oxidation [44].
- Extracellular Matrix (ECM) Mimetic Peptides (e.g., RGD): Promote specific cell adhesion while potentially reducing nonspecific protein adsorption when properly engineered [48].
Decision Framework and Research Outlook
Selecting an optimal antifouling strategy requires consideration of multiple application-specific factors:
The field continues to evolve toward multifunctional, responsive, and biomimetic solutions. Future directions include the development of hybrid architectures that combine physical and chemical antifouling strategies, smart coatings that adapt to environmental changes, and more durable formulations capable of withstanding harsh operational conditions over extended periods [48] [49]. For researchers comparing specialized interfaces like BiFE, HgFE, and AuFE, selecting appropriate antifouling strategies becomes crucial for minimizing interference and ensuring accurate performance assessment across platforms.
Optimization of Electrolyte Composition and Applied Potential
The performance of electrochemical devices is fundamentally governed by the complex interplay between electrolyte composition and the applied operational potentials. Optimizing this relationship is critical for enhancing key metrics such as ionic conductivity, interfacial stability, cycling life, and energy density. This guide provides a comparative analysis of advanced electrolyte systems, including liquid-based, gel polymer, and specialized formulations, focusing on their performance under various electrochemical conditions. By presenting structured experimental data and detailed methodologies, this review serves as a reference for researchers designing next-generation energy storage systems, framing the discussion within the broader context of achieving interference-resistant electrochemical operation.
Comparative Performance of Electrolyte Systems
The design of an electrolyte system directly dictates its functional characteristics, including ionic conductivity, electrochemical stability window, and compatibility with electrodes. The table below provides a quantitative comparison of modern electrolyte strategies to establish a baseline for their performance envelopes.
Table 1: Performance Comparison of Advanced Electrolyte Systems
| Electrolyte System | Key Composition | Ionic Conductivity | Voltage Window | Performance Highlights | Key Challenges |
|---|---|---|---|---|---|
| Localized High-Concentration Electrolyte (LHCE) [50] | LiFSI–1.2DME–3TTE (Molar Ratio) | Data not specified | High (Compatible with NMC811) | 77% capacity retention after 483 cycles in Li||NMC811 pouch cell; High Li cycling CE (>99.5%) | Significant LiFSI salt consumption leads to ion depletion [50]. |
| Fluorine-Grafted Gel Polymer Electrolyte [51] | PAN-based with PETEA-HFA (3:2) graft | 4.06 × 10⁻³ S cm⁻¹ @ 30°C | 5.35 V (vs. Li/Li⁺) | Li⁺ transference no. (tₗᵢ₊) of 0.87; NCM811|GPE|Li cycle life: 300 cycles with 84% retention @ 2C [51]. | Restrictive electrochemical stability at high potentials; Side reactions with transition metal ions [51]. |
| High-Concentration Potassium Electrolyte [52] | 5.0 M KFSI in EC/DMC (1:1 v/v) | Data not specified | 5.25 V (vs. K⁺/K) | Enables full FSI⁻ intercalation/de-intercalation in graphite cathode (3.2–5.25 V) [52]. | High viscosity; Limited salt solubility (e.g., KPF₆) in carbonates [52]. |
| Dual-Unsaturated Additive Electrolyte [53] | 1.15 M LiPF₆ + 0.5% VC + 0.3% PES | Data not specified | Suitable for high-voltage NCM523 | 97.54% capacity retention after 500 cycles at 45°C in Gr|NMC523 pouch cell; Mitigates Li plating [53]. | Optimization required for additive concentration and compatibility. |
Experimental Protocols for Electrolyte Evaluation
A critical comparison requires an understanding of the experimental methods used to generate performance data. The following protocols are standardized approaches for quantifying electrolyte properties and cell performance.
Protocol for Ionic Conductivity and Transference Number Measurement
The ionic conductivity (σ) of an electrolyte is a fundamental property indicating its ability to conduct ions. It can be calculated using the equation σ = ΣiniμiZie, where ni is the number of free ions, μi is the ion mobility, Zi is the charge number, and e is the elementary charge [54]. Experimentally, this is often determined using electrochemical impedance spectroscopy (EIS) on a symmetric cell (e.g., stainless steel\|electrolyte\|stainless steel) to measure bulk resistance. The lithium-ion transference number (tₗᵢ₊), which represents the fraction of total current carried by Li⁺ ions, is crucial for mitigating concentration polarization. It can be determined using a combination of DC polarization and AC impedance methods (e.g., Bruce-Vincent method) on a Li\|electrolyte\|Li symmetric cell [51]. For instance, the high tₗᵢ₊ of 0.87 in the fluorine-grafted GPE was confirmed through such a protocol [51].
Protocol for Electrochemical Stability Window (ESW) Assessment
The ESW defines the voltage range within which the electrolyte is thermodynamically stable against oxidation and reduction. It is typically evaluated using linear sweep voltammetry (LSV) or cyclic voltammetry (CV) in a three-electrode cell, using an inert working electrode (e.g., platinum or glassy carbon), a lithium metal counter electrode, and a lithium metal reference electrode. The anodic stability limit is identified by the onset current of a rapid increase in current, indicating electrolyte oxidation. As an example, the fluorine-grafted GPE demonstrated stability up to 5.35 V vs. Li/Li⁺ using such a method [51]. The stability is intrinsically linked to the electrolyte's highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels [52].
Protocol for Long-Term Cycling and Failure Analysis
This protocol evaluates the practical performance and failure mechanisms of an electrolyte in a full-cell configuration, such as Li\|NMC811.
- Cell Assembly: Assemble single-layer pouch cells (e.g., with a 20 μm thick Li anode and a high-mass-loading NMC811 cathode ~17.1 mg cm⁻²) under lean electrolyte conditions (e.g., 2.1 g Ah⁻¹) [50].
- Electrochemical Cycling: Cycle the cells under specified conditions (e.g., charge at 0.2 C / discharge at 1 C) at a controlled temperature (e.g., 25°C) until failure (e.g., <80% capacity retention) [50].
- Post-Mortem Analysis:
- Titration-Differential Electrochemical Mass Spectrometry (T-DEMS): Quantify the consumption of active lithium and the formation of specific species like LiH and Li₂CO₃ in the solid-electrolyte interphase (SEI) after cycling [50].
- Extraction-Gas & Ion Chromatography (E-G&IC): Measure the irreversible consumption of lithium salt and organic solvents from the electrolyte after long-term cycling [50]. This protocol revealed that nearly 60% of LiFSI salt was consumed in an LHCE system after 100 cycles, leading to Li⁺ depletion and cell failure [50].
Electrolyte Optimization Pathways and Interfacial Interactions
The relationship between electrolyte composition, the resulting interfacial chemistry, and overall cell performance is complex. The following diagram illustrates the primary optimization pathways and their impacts on electrode interfaces.
Diagram 1: Electrolyte optimization pathways and their impacts on device performance. SEI: Solid-Electrolyte Interphase; CEI: Cathode-Electrolyte Interphase; VC: Vinylene Carbonate; PES: Prop-1-ene-1,3-sultone.
The Scientist's Toolkit: Key Research Reagents and Materials
Developing high-performance electrolytes requires a suite of specialized materials. The table below details key components, their functions, and representative examples from recent research.
Table 2: Essential Materials for Electrolyte Research and Development
| Material Category | Specific Example | Primary Function | Research Context / Rationale |
|---|---|---|---|
| Lithium Salts | Lithium bis(fluorosulfonyl)imide (LiFSI) | Provides Li⁺ ions for conduction; anion-derived SEI formation. | Used in LHCEs for high anodic stability; however, continuous decomposition can lead to Li⁺ depletion [50]. |
| Lithium Salts | Lithium hexafluorophosphate (LiPF₆) | Standard Li⁺ source; forms LiF in SEI/CEI. | Used with dual-unsaturated additives (VC/PES) to create a LiF-rich interphase and deactivate harmful PF₅ [53]. |
| Solvents | Dimethoxyethane (DME) | Ether-based solvent with low viscosity; good solvating power. | Used in LHCE formulations with LiFSI; participates in solvation structure but is less consumed than LiFSI during cycling [50]. |
| Solvents | Ethylene Carbonate (EC) | Carbonate solvent with high dielectric constant; aids salt dissociation. | Provides good anti-oxidation capability (high HOMO); fundamental component in many carbonate-based electrolytes [52]. |
| Diluents | 1,1,2,2-Tetrafluoroethyl-2,2,3,3-tetrafluoropropyl ether (TTE) | Non-solvating, fluorinated diluent; reduces viscosity and cost of HCEs. | Forms LHCEs; its mass remains constant during cycling, indicating non-participation in interfacial reactions [50]. |
| Polymer Matrices | Polyacrylonitrile (PAN) | Provides mechanical support and creates Li⁺ transport channels in GPEs. | Used as a porous membrane support in fluorine-grafted GPEs to form a continuous ion transport network [51]. |
| Additives | Vinylene Carbonate (VC) | Polymerizable film-forming additive. | Co-polymerizes with PES to form a spatially adaptable polymeric SEI on graphite anodes, mitigating Li plating [53]. |
| Additives | Prop-1-ene-1,3-sultone (PES) | Sulfur-containing, film-forming additive. | Works synergistically with VC; introduces S–F species into the CEI, enhancing high-temperature stability [53]. |
| Monomers for GPEs | Pentaerythritol tetraacrylate (PETEA) & 2,2,3,4,4,4-hexafluorobutyl acrylate (HFA) | Forms cross-linked polymer network for GPE; HFA provides fluorine groups. | Grafting HFA onto PETEA helps capture anions, improves Li⁺ transference number, and enhances interfacial stability [51]. |
Electrodes are fundamental components in various scientific and industrial applications, from electrochemical energy storage to advanced biosensing. However, their performance often degrades over time due to surface contamination, fouling, or structural changes. Regeneration protocols are essential for restoring electrode functionality, ensuring experimental consistency, and reducing costs. This guide objectively compares regeneration methods for different electrode types, with a specific focus on interference resistance within the context of bismuth film electrode (BiFE), mercury film electrode (HgFE), and gold film electrode (AuFE) research. The protocols and data presented herein provide researchers with validated methodologies for maintaining electrode performance.
Comparative Analysis of Electrode Regeneration Methods
The choice of regeneration protocol is highly dependent on the electrode material and its specific application. The following table summarizes the key characteristics of established regeneration methods.
Table 1: Comparison of Electrode Regeneration Protocols
| Regeneration Method | Target Electrode Materials | Core Mechanism | Typical Processing Conditions | Regeneration Efficiency | Reported Cycle Life | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|---|
| High-Temperature Pulse Annealing [55] | Catalytic electrodes (e.g., Ru-loaded), various carbonaceous & metallic substrates | Rapid thermal decomposition of byproducts & restoration of catalyst properties | High-temperature, rapid pulses | Restores original electrochemical performance | 10 full cycles [55] | Non-destructive; direct electrode reuse; versatile application [55] | Requires specialized equipment for high-temperature control |
| Double-Step Electrochemical (DSEE) Etching [56] | Gold Screen-Printed Electrodes (Au-SPEs) | Sequential electrochemical cleaning in H₂SO₄ and K₃Fe(CN)₆ to remove SAMs and bio-layers | CV in 10 mM H₂SO₄ (-1.8 V to 0 V); then CV in 50 mM K₃Fe(CN)₆ (-1.2 V to 1.2 V) [56] | ~95% current recovery vs. bare electrode [56] | 5 cycles (immunosensor & cytosensor) [56] | Nontoxic, simple protocol; complete organic layer removal; maintains Au surface integrity [56] | Primarily optimized for Au surfaces; performance declines after multiple cycles |
| Chemical Etching (Piranha Solution) [56] | Gold Electrodes | Strong oxidation to remove organic residues | Immersion in H₂SO₄ + H₂O₂ mixture | Effective residue removal | Limited data on repeated use | Fast and effective for stubborn contaminants [56] | Highly toxic and hazardous; can damage the underlying substrate [56] |
Detailed Experimental Protocols for Electrode Regeneration
High-Temperature Pulse Annealing for Catalytic Electrodes
This method is designed for regenerating catalytic electrodes used in energy devices, such as Li-air batteries.
- Principle: Short, high-temperature pulses decompose accumulated byproducts (e.g., Li₂O₂, Li₂C₂) on the electrode surface without altering the underlying catalyst's physiochemical properties. [55]
- Materials & Equipment:
- Used catalytic electrode (e.g., Ru-loaded carbon)
- Pulse annealing furnace with rapid heating/cooling capabilities
- Inert atmosphere gas supply (e.g., Argon)
- Step-by-Step Procedure:
- Remove the used electrode from the electrochemical cell.
- Place the electrode in the pulse annealing furnace.
- Purge the chamber with an inert gas to prevent unwanted oxidation.
- Apply a series of high-temperature rapid pulses. The exact temperature and duration are application-specific but are designed to be sufficiently high to decompose byproducts while being brief enough to preserve the catalyst nanostructure. [55]
- After treatment, cool the electrode to room temperature under an inert atmosphere.
- The regenerated electrode is now ready for reassembly and testing.
Double-Step Electrochemical Etching (DSEE) for Gold Electrodes
This protocol is specifically designed for regenerating gold-based biosensors by removing self-assembled monolayers (SAMs) and bio-affinity layers.
- Principle: The first step in dilute sulfuric acid helps desorb and break down organic layers. The subsequent step in potassium ferricyanide further oxidizes and removes any residual contaminants, restoring the electroactive surface area. [56]
- Materials & Equipment:
- Used gold screen-printed electrode (Au-SPE)
- Potentiostat
- 10 mM H₂SO₄ solution
- 50 mM K₃Fe(CN)₆ solution
- Phosphate-buffered saline (PBS) or deionized water for rinsing
- Step-by-Step Procedure:
- First Step (H₂SO₄ Treatment): Immerse the used Au-SPE in a 10 mM H₂SO₄ solution. Perform cyclic voltammetry (CV) by scanning the potential from -1.8 V to 0 V (vs. the electrode's internal reference) at a scan rate of 100 mV/s for a predetermined number of cycles. [56]
- Rinse: Rinse the electrode thoroughly with deionized water or PBS.
- Second Step (K₃Fe(CN)₆ Treatment): Immerse the electrode in a 50 mM K₃Fe(CN)₆ solution. Perform CV by scanning from -1.2 V to 1.2 V at a scan rate of 200 mV/s. [56]
- Final Rinse and Dry: Rinse the electrode again with deionized water and dry under a gentle stream of nitrogen.
- Validation: The regeneration efficiency can be validated by comparing the CV response of the etched electrode in a standard redox probe (e.g., [Fe(CN)₆]³⁻/⁴⁻) to that of a new, bare Au-SPE. A recovery of ~95% in current response indicates successful regeneration. [56]
Gold Electrode DSEE Regeneration Workflow
The Scientist's Toolkit: Essential Reagents & Materials
Successful electrode regeneration relies on specific laboratory reagents and equipment. The following table details key items for implementing the discussed protocols.
Table 2: Essential Research Reagent Solutions for Electrode Regeneration
| Item Name | Function/Application | Key Considerations |
|---|---|---|
| Potassium Ferricyanide (K₃Fe(CN)₆) | Electrochemical etchant for gold electrodes; oxidizes and removes residual organic layers in the DSEE protocol. [56] | Use an aqueous solution (e.g., 50 mM); part of a non-toxic alternative to piranha solution. [56] |
| Dilute Sulfuric Acid (H₂SO₄) | First-step etchant in DSEE protocol; helps desorb and break down self-assembled monolayers (SAMs) on Au surfaces. [56] | Low concentration (e.g., 10 mM) is sufficient, enhancing safety compared to concentrated acid. [56] |
| Piranha Solution | Powerful oxidizing agent for removing organic residues from gold and other metal surfaces. [56] | Extremely hazardous; requires specialized training and handling; can damage delicate electrode substrates. [56] |
| Inert Atmosphere (Argon/N₂) | Provides an oxygen-free environment during high-temperature annealing to prevent oxidation of the catalytic electrode. [55] | Essential for preserving the chemical state and structure of sensitive catalyst materials during thermal regeneration. [55] |
| Phosphate Buffered Saline (PBS) | A common rinsing solution for biosensors; used to remove salts and biological residuals between etching steps or after regeneration. | Ensures a neutral pH and isotonic environment, preventing damage to the electrode or interference with subsequent surface chemistry. |
Interference Resistance in the Context of BiFE, HgFE, and AuFE Research
The regeneration of film electrodes (BiFE, HgFE, AuFE) is intrinsically linked to their interference resistance. A freshly prepared or properly regenerated electrode surface is crucial for reproducible and accurate analysis, particularly in complex biological matrices where surface fouling by proteins or other macromolecules is a major concern.
Surface Renewal vs. Interference Resistance: HgFEs have historically been valued for their ability to be easily renewed by displacing the old film and forming a new one, which helps mitigate fouling. However, the toxicity of mercury is a significant drawback. [57] BiFEs are often presented as a non-toxic alternative to HgFEs, and their interference resistance can be maintained through electrochemical regeneration protocols that redeposit a fresh, uniform bismuth film. AuFEs, while biocompatible, are highly susceptible to surface fouling and require robust cleaning methods like DSEE to restore their active surface and, consequently, their analytical performance. [56] [57]
Impact of Regeneration on Analytical Performance: The primary goal of regenerating film electrodes is to restore a pristine, electroactive surface. This directly enhances interference resistance by:
- Reducing Surface Fouling: A clean surface with well-defined properties is less prone to non-specific adsorption of interfering compounds.
- Restoring Electron Transfer Kinetics: Regeneration removes insulating layers, ensuring fast and efficient electron transfer, which is vital for distinguishing the target analyte's signal from interferents.
- Maintaining Signal-to-Noise Ratio: A consistent, regenerated surface provides a stable baseline (low noise) and a high Faradaic response (strong signal), which is critical for low-detection limits in the presence of complex backgrounds.
How Regeneration Enhances Interference Resistance
Selecting the appropriate regeneration protocol is paramount for sustainable and reliable electrochemical research. Methods like high-temperature pulse annealing offer a non-destructive path for complex catalytic electrodes, while electrochemical techniques like DSEE provide a safer, effective alternative for rejuvenating gold biosensors. The choice between BiFE, HgFE, and AuFE involves a careful balance between toxicity, ease of regeneration, and inherent interference resistance. The protocols and data compared in this guide provide a foundation for researchers to make informed decisions, ensuring long-term electrode performance and the generation of high-quality, reproducible data.
Head-to-Head Validation: A Systematic Comparison of Analytical Performance
In the rigorous field of analytical science, particularly within electrochemical biosensor research, the performance of a sensing platform is quantitatively benchmarked using a set of well-defined metrics. Limit of Detection (LoD), sensitivity, and linear range are three fundamental parameters that collectively describe the lowest concentration an assay can identify, its ability to distinguish between small concentration differences, and the span over which quantitative results are reliable. For researchers comparing different electrode materials, such as bismuth-film (BiFE), mercury-film (HgFE), and gold-film (AuFE) electrodes, within the specific context of interference resistance, a deep understanding of these metrics is paramount. These parameters are not merely theoretical constructs but are determined through standardized experimental protocols that allow for direct, objective comparison between technologies. Accurate characterization of these metrics enables scientists to select the optimal sensing material for applications ranging from environmental monitoring to drug development and clinical diagnostics, ensuring that the chosen system is "fit for purpose" [58].
The following sections will delineate the formal definitions and standard methods for determining these critical metrics, provide detailed experimental protocols for their benchmarking, present a comparative analysis of electrode performances, and explore advanced sensing technologies that define the current state-of-the-art.
Defining the Core Metrics
Limit of Detection (LoD) and Limit of Quantification (LoQ)
The Limit of Detection (LoD) is the lowest concentration of an analyte that can be reliably distinguished from a blank sample containing no analyte. It is a critical threshold for confirming the presence of a target substance. Closely related is the Limit of Quantification (LoQ), which is the lowest concentration at which the analyte can not only be detected but also quantified with acceptable precision and trueness (bias) [58] [59]. The LoQ is always greater than or equal to the LoD.
The establishment of LoD and LoQ involves a structured statistical approach, often based on the properties of blank samples and low-concentration analytes. The Clinical and Laboratory Standards Institute (CLSI) guideline EP17 provides a standard method for this determination [58].
Limit of Blank (LoB): This is a foundational concept defined as the highest apparent analyte concentration expected to be found when replicates of a blank sample are tested. It is calculated as: LoB = meanblank + 1.645(SDblank) This formula assumes a Gaussian distribution, setting the LoB at the 95th percentile of blank measurements to minimize false positives (Type I error) [58].
Limit of Detection (LoD): The LoD is determined by utilizing both the measured LoB and test replicates of a sample containing a low concentration of analyte. It represents the concentration at which detection is feasible and is calculated as: LoD = LoB + 1.645(SD_low concentration sample) This ensures that 95% of measurements from a sample at the LoD concentration will exceed the LoB, minimizing false negatives (Type II error) [58].
Alternative Calculation Methods: Other common approaches include:
- Signal-to-Noise Ratio (S/N): Typically, a S/N ratio of 3:1 is acceptable for LoD, and 10:1 for LoQ [59].
- Standard Deviation and Slope: Using a calibration curve, LoD and LoQ can be calculated as LoD = 3.3 * σ / S and LoQ = 10 * σ / S, where σ is the standard deviation of the response and S is the slope of the calibration curve [59].
Table 1: Summary of LoB, LoD, and LoQ
| Parameter | Definition | Sample Type | Key Statistical Basis |
|---|---|---|---|
| Limit of Blank (LoB) | Highest apparent concentration expected from a blank sample. | Sample containing no analyte. | 95th percentile of blank sample distribution [58]. |
| Limit of Detection (LoD) | Lowest concentration reliably distinguished from the LoB. | Sample with low concentration of analyte. | LoB + 1.645(SD of low concentration sample) [58]. |
| Limit of Quantification (LoQ) | Lowest concentration quantified with acceptable precision and trueness. | Sample with low concentration of analyte at or above LoD. | Predefined goals for bias and imprecision are met; ≥ LoD [58]. |
Sensitivity
In an analytical context, sensitivity has two distinct but important meanings, which should not be confused. First, it can refer to the analytical sensitivity, which is the slope of the calibration curve. This indicates how much the analytical signal changes per unit change in analyte concentration; a steeper slope signifies a more sensitive assay [58]. Second, in clinical or diagnostic settings, sensitivity refers to the ability of a test to correctly identify positive samples. For this comparison, we focus on the former, analytical sensitivity.
For nanoplasmonic biosensors, a related metric is bulk sensitivity, expressed as resonance shifts per unit change in the bulk refractive index (e.g., nm/RIU) [60]. A higher sensitivity means the sensor can detect smaller changes in analyte concentration.
Linear Range
The linear range, or analytical measurement range, is the concentration interval over which the analytical response has a linear relationship with the analyte concentration. Within this range, the sensor can provide accurate quantitative data. The lower end of the linear range is bounded by the LoQ, as quantification below this point becomes unreliable. The upper end is marked by a plateau or deviation from linearity in the signal response. A wide linear range is desirable for analyzing samples with varying analyte concentrations without requiring dilution.
Experimental Protocols for Metric Determination
General Workflow for LoD and LoQ Determination
A standardized protocol is essential for generating comparable data. The following workflow, based on CLSI EP17 guidelines, outlines the key steps for determining LoD and LoQ [58].
Detailed Methodologies
1. Reagent and Sensor Preparation:
- Blank Solution: A matrix-matched solution containing all components except the target analyte.
- Low-Concentration Analyte Samples: Samples prepared in the same matrix as the blank, with analyte concentrations near the expected LoD. Serial dilutions from a stock solution are typically used.
- Sensor Conditioning: For electrode-based sensors (BiFE, HgFE, AuFE), preconditioning via electrochemical cycling in a suitable electrolyte is often required to stabilize the electrode surface and ensure reproducible performance.
2. Data Acquisition and Analysis:
- Measurement: Analyze at least 20 (for verification) to 60 (for establishment) independent replicates of both the blank and low-concentration samples [58]. Measurements should be randomized to avoid systematic bias.
- Calculation: Compute the mean and standard deviation (SD) for both the blank and low-concentration sample sets. Apply the formulas for LoB and LoD as defined in Section 2.1.
- Verification: Confirm the provisional LoD by testing a sample with analyte concentration at the calculated LoD. If more than 5% of the results fall below the LoB, the LoD must be re-estimated using a higher concentration sample [58].
- LoQ Determination: Measure samples at various concentrations above the LoD in multiple replicates. The LoQ is the lowest concentration where the relative standard deviation (RSD, or CV%) and bias (deviation from the true value) fall within pre-defined acceptable limits (e.g., CV ≤ 20% for functional sensitivity) [58].
3. Interference Testing: To benchmark interference resistance, the above protocols should be repeated in the presence of potential interferents at physiologically or environmentally relevant concentrations. The change in LoD, LoQ, and sensitivity is then recorded. A robust sensor will show minimal variation in these key metrics when interferents are present.
Comparative Performance of Sensing Platforms
Comparison of Electrode Materials
Different electrode materials offer distinct advantages and disadvantages in terms of key metrics, cost, and toxicity, which influences their application in interference-prone environments.
Table 2: Benchmarking Key Metrics for Electrode Materials
| Feature | Bismuth-Film Electrode (BiFE) | Mercury-Film Electrode (HgFE) | Gold-Film Electrode (AuFE) |
|---|---|---|---|
| Typical LoD Range | Low nM to pM | Very low pM to fM (Excellent) | nM to pM |
| Sensitivity | High | Very High (Excellent) | High |
| Linear Range | Wide (4-5 orders) | Wide (4-6 orders) | Moderate to Wide |
| Interference Resistance | Good (resistant to O₂, metal ions) | Moderate (excellent cathodic window) | Poor (susceptible to fouling, anion adsorption) |
| Key Advantages | Non-toxic, renewable surface, works in air-saturated solutions. | Superior sensitivity, wide cathodic potential window, high-quality films. | Biocompatible, easy functionalization, good for anodic detection. |
| Key Disadvantages | Narrow anodic potential window. | Toxic, surface can be unstable with time. | Expensive, prone to fouling and interference. |
| Ideal Application Context | Environmental heavy metal sensing in the field. | Ultra-trace lab analysis of metals and organics. | Biosensing, label-free affinity assays (SPR, electrochemical) [60]. |
Advanced Biosensing Platforms
Beyond traditional electrodes, newer biosensing technologies offer unique performance profiles.
- Nanoplasmonic Biosensors: These sensors, including conventional Surface Plasmon Resonance (SPR), confine light to probe biomolecular interactions. They boast a high bulk sensitivity (up to ~10,000 nm/RIU) and can achieve a detection limit of a few picograms of protein per cm² [60]. Their key strength is in label-free, real-time monitoring of binding kinetics.
- Biosensor-Integrated Drug Delivery Systems: These are closed-loop systems that combine continuous biomarker monitoring with automated drug administration. For example, glucose-responsive systems mimic pancreatic beta cells to release insulin in response to plasma glucose levels. The sensitivity and LoD of the integrated biosensor component are critical for triggering timely and accurate drug release [61].
- Silicon Nanowire Sensors: Emerging technologies like those from Advanced Silicon Group combine silicon nanowires with antibodies to create highly sensitive protein sensors. These platforms claim to be 15 times faster and lower cost than traditional ELISA tests, making them promising for affordable diagnostics and bioprocessing quality control [62].
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key reagents and materials required for the experimental characterization of biosensors and electrodes.
Table 3: Essential Research Reagent Solutions for Sensor Benchmarking
| Reagent/Material | Function and Description | Application Example |
|---|---|---|
| Blank Matrix Solution | A solution mimicking the sample matrix (e.g., buffer, serum) without the analyte. Serves as the negative control for LoB and baseline signal determination. | Phosphate Buffered Saline (PBS) for biological assays; synthetic water for environmental samples. |
| Analyte Stock Solution | A high-purity, accurately known concentration of the target analyte. Used to prepare serial dilutions for calibration curves, LoD/LoQ studies, and sensitivity determination. | A certified standard solution of a heavy metal ion for electrode testing; a purified protein for immunoassay development. |
| Supporting Electrolyte | A salt added in high concentration to the measurement solution to ensure ionic conductivity and minimize ohmic drop in electrochemical experiments. | Potassium chloride (KCl) or nitric acid (HNO₃) for stripping voltammetry of metals. |
| Interferent Stock Solutions | Solutions of chemically similar species or common foulants expected in real samples. Used to challenge the sensor and benchmark its interference resistance. | Solutions of Cu²⁺, Zn²⁺ for a Pb²⁺ sensor; albumin for a blood glucose sensor. |
| Surface Functionalization Reagents | Chemicals used to modify the sensor surface to make it selective for the target analyte. | Thiols (for AuFE), silanes (for oxide surfaces), antibodies, enzymes (e.g., Glucose Oxidase) [61]. |
| Regeneration Buffer | A solution that disrupts the binding between the analyte and the immobilized receptor without damaging the sensor surface. Allows for re-use of the sensor. | Low pH buffer (e.g., Glycine-HCl) or a surfactant solution for SPR and other affinity biosensors. |
The rigorous benchmarking of Limit of Detection, Sensitivity, and Linear Range is a non-negotiable process in the development and selection of biosensing platforms. As demonstrated, materials like HgFE offer unrivalled sensitivity for trace analysis, while BiFE presents a robust, non-toxic alternative for field-use, and AuFE is well-suited for functionalized biosensors. The choice of platform is a trade-off between these performance metrics, interference resistance, cost, and practicality. Emerging technologies like nanoplasmonic sensors and silicon nanowire devices continue to push the boundaries of these key metrics, offering enhanced sensitivity, lower cost, and new functionalities such as single-molecule detection and integrated drug delivery. For researchers in drug development and diagnostics, a thorough understanding of these metrics and the methodologies for their determination is fundamental to driving innovation and ensuring the reliability of analytical data.
Comparative Analysis of Interference Resistance in Standardized Tests
The accurate detection of heavy metals in various samples is crucial for environmental monitoring, pharmaceutical quality control, and toxicological research. Electrochemical sensors, particularly those utilizing bismuth-film electrodes (BiFE), mercury-film electrodes (HgFE), and gold-film electrodes (AuFE), have emerged as prominent tools for this purpose due to their sensitivity and potential for miniaturization. However, a significant challenge in their practical application is interference resistance—the ability to maintain accurate detection of target analytes in the presence of interfering species. This analysis provides a comprehensive comparison of the interference resistance properties of BiFE, HgFE, and AuFE platforms, drawing upon experimental data and methodological approaches from current research. The evaluation is contextualized within the broader framework of developing robust, reliable sensing platforms for researchers and drug development professionals who require precise metal quantification in complex matrices.
Performance Data Comparison
The analytical performance of electrode materials varies significantly based on their composition, surface characteristics, and the experimental conditions employed. The following tables summarize key performance metrics and interference resistance data for BiFE, HgFE, and AuFE systems, compiled from experimental studies.
Table 1: Key Analytical Performance Metrics for Film Electrodes
| Performance Parameter | BiFE | HgFE | AuFE |
|---|---|---|---|
| Limit of Detection (Cd²⁺) | ~nM range | ~nM range | Varies with design |
| Linear Dynamic Range | Wide | Wide | Moderate to Wide |
| Electrode Renewability | Good | Excellent (flowing drops) | Fair (requires polishing) |
| Reproducibility (% RSD) | <5% | <3% | ~5-10% |
| Optimal pH Range | Near-neutral | Wide, often acidic | Acidic to neutral |
Table 2: Interference Resistance Profile Against Common Ions
| Interfering Species | BiFE Response | HgFE Response | AuFE Response |
|---|---|---|---|
| Dissolved Oxygen | Moderate tolerance | High tolerance | Low tolerance (often requires deaeration) |
| Surfactants | Moderately susceptible | Highly susceptible | Highly susceptible |
| Cu²⁺ | Significant interference | Amalgamation formation | Surface alloying, major interference |
| Zn²⁺ | Minor interference | Co-amalgamation | Minor interference |
| Organic Ligands (e.g., Humics) | Moderate signal suppression | Significant signal suppression | Varies with ligand |
Experimental Protocols for Interference Assessment
A standardized approach to evaluating interference resistance is critical for obtaining comparable data. The following protocols detail common methodologies used to generate the comparative data.
Anodic Stripping Voltammetry (ASV) with Standard Additions
This is a cornerstone technique for quantifying heavy metals and assessing interference.
- Principle: The target metal ion is electrochemically reduced and pre-concentrated onto the electrode surface as an amalgam or film, followed by anodic re-dissolution (stripping). The resulting current peak is proportional to concentration.
- Procedure:
- Electrode Preparation: The working electrode (BiFE, HgFE, or AuFE) is prepared. BiFE is often electroplated in situ from a Bi(III) solution. HgFE is formed on a suitable substrate. AuFE requires careful pre-cleaning.
- Supporting Electrolyte: A known volume of the sample or standard solution is placed in the cell with a suitable supporting electrolyte (e.g., acetate buffer for BiFE, HCl for HgFE/AuFE).
- Deaeration (if required): For AuFE, dissolved oxygen is removed by purging with an inert gas (e.g., N₂ or Ar) for 8-10 minutes.
- Pre-concentration: The potential is held at a reducing value (e.g., -1.2 V vs. Ag/AgCl) for a fixed time (60-120 s) with solution stirring.
- Equilibration: Stirring is stopped, and the solution is allowed to equilibrate for 15-30 s.
- Stripping Scan: The potential is scanned anodically (e.g., from -1.2 V to 0 V) using a sensitive technique like Square-Wave Voltammetry (SWV) or Differential Pulse Voltammetry (DPV) to dissolve the metals.
- Standard Addition: Known aliquots of a standard metal solution are added to the cell, and steps 4-6 are repeated after each addition to build a calibration curve and account for matrix effects [63].
Interference Tolerance Test
This protocol specifically quantifies the impact of interferents.
- Principle: The signal for a fixed concentration of the target analyte is measured in the absence and presence of increasing concentrations of an interfering species.
- Procedure:
- Baseline Signal Measurement: Using the ASV protocol above, measure the stripping peak current (Ip) for the target analyte (e.g., 10 µg/L Cd²⁺) in a clean matrix.
- Introduction of Interferent: Add a known concentration of the interferent (e.g., Cu²⁺) to the cell.
- Signal Re-measurement: Repeat the ASV measurement and record the new peak current (Ip').
- Tolerance Calculation: The signal suppression/enhancement is calculated as (Ip' / Ip) × 100%. The interferent concentration causing a ±10% change in signal is often reported as the tolerance limit.
- Surface Inspection: For intermetallic interference (e.g., Cu-Zn on AuFE), the electrode surface is often inspected post-experiment using techniques like SEM/EDS to confirm alloy formation.
Visualization of Experimental Workflow
The following diagram illustrates the logical sequence and decision points in the standard experimental workflow for evaluating electrode interference resistance.
Figure 1. Interference Resistance Evaluation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Successful experimentation in this field relies on a set of essential materials and reagents. The following table details key items and their functions in the context of preparing and testing these electrochemical sensors.
Table 3: Essential Reagents and Materials for Electrode Testing
| Item | Function/Description | Application Notes |
|---|---|---|
| Bismuth Salt (e.g., Bi(NO₃)₃) | Precursor for in-situ or ex-situ electroplating of BiFE films. | An environmentally friendly ("green") alternative to mercury [63]. |
| Gold Electrode (µ-Disc or Sputtered) | Substrate for AuFE studies or for BiFE plating. Requires meticulous mechanical and electrochemical cleaning. | Prone to intermetallic compound formation with metals like copper [63]. |
| Mercury Film Electrode (MFE) | Traditional electrode formed by electrodepositing Hg on a carbon substrate. | Offers a wide cathodic potential window and high hydrogen overpotential [63]. |
| Supporting Electrolyte (e.g., Acetate Buffer, HCl) | Provides ionic conductivity, fixes pH, and can influence metal speciation and deposition efficiency. | BiFE performs well in acetate buffers; HgFE and AuFE are often used in HCl media. |
| Metal Standard Solutions | High-purity single or multi-element solutions for calibration and interference studies. | Used for standard addition methods to compensate for complex matrix effects. |
| Square-Wave Voltammetry (SWV) | A sensitive electrochemical technique used for the stripping step, offering effective background current suppression. | Allows for fast scanning and low detection limits, commonly used with all three electrodes [63]. |
The comparative analysis reveals a nuanced landscape for interference resistance among BiFE, HgFE, and AuFE. BiFE presents a compelling, environmentally sustainable alternative to HgFE, with generally good interference resistance, particularly in the presence of oxygen, and robust performance in near-neutral pH conditions suitable for many biological and environmental samples. However, it can be susceptible to certain metal interferences like copper. The traditional HgFE remains a benchmark for sensitivity and a wide potential window, offering excellent renewalability in flowing systems, but its susceptibility to surfactants and organic ligands, combined with toxicity concerns, limits its application. AuFE provides a valuable platform but often requires rigorous deoxygenation and is highly susceptible to intermetallic interference, which can be mitigated through surface modification. The choice of electrode material is, therefore, application-dependent. Researchers must weigh factors such as toxicity, required detection limits, the specific sample matrix, and the nature of potential interferents when selecting a sensor platform for standardized heavy metal testing.
Stability and Reproducibility Assessment Under Long-Term Operation
The assessment of stability and reproducibility under long-term operation is a critical consideration in electrochemical sensor research, directly impacting the reliability and real-world applicability of sensing platforms. Within the specific context of interference resistance, comparing sensor performance over extended periods provides essential data for selecting appropriate electrode materials. This guide objectively compares the long-term operational stability and reproducibility of Bismuth-Film Electrodes (BiFE), Mercury-Film Electrodes (HgFE), and Gold-Film Electrodes (AuFE) based on current experimental findings, with particular emphasis on their resilience to interfering substances and environmental factors. The evaluation framework focuses on quantitative performance metrics, including detection sensitivity, signal consistency, and resistance to fouling across multiple operational cycles, providing researchers and drug development professionals with evidence-based criteria for sensor selection and development.
Comparative Performance Analysis of Electrode Materials
Quantitative Performance Metrics Under Long-Term Operation
Table 1: Comprehensive performance comparison of BiFE, HgFE, and AuFE for long-term operation
| Performance Parameter | Bismuth-Film Electrodes (BiFE) | Mercury-Film Electrodes (HgFE) | Gold-Film Electrodes (AuFE) |
|---|---|---|---|
| Detection Limit (Sb(III)) | 3.6 ng L⁻¹ (t_ads: 60 s) [64] | Historical benchmark | Not specified in results |
| Optimal pH Range | 5.5 [64] | Varies | Varies |
| Reproducibility (RSD) | 1.7-2.5% [64] | Historical data shows degradation | Subject to fouling |
| Interference Resistance | High (with QSA) [64] | Moderate | Low for certain analytes |
| Environmental Impact | Low toxicity [64] | High toxicity | Non-toxic |
| Long-term Signal Stability | >95% over 50 cycles (in optimal conditions) | Gradual deterioration | Dependent on surface regeneration |
| Fouling Resistance | High with proper modification | Moderate | Low in complex matrices |
Interference Resistance Profiles
Table 2: Interference resistance characteristics in complex matrices
| Interference Type | BiFE Performance | HgFE Performance | AuFE Performance |
|---|---|---|---|
| Organic Compounds | Moderate resistance with QSA modification [64] | High historical performance | Prone to fouling |
| Heavy Metal Ions | Selective with proper complexation [64] | Broad spectrum detection | Selective response |
| Surface-Active Substances | Moderate tolerance | High tolerance | Low tolerance |
| pH Variations | Stable in acidic to neutral range [64] | Broad range | Medium-dependent |
| Chloride Ions | Minimal interference | Significant interference | Moderate interference |
Experimental Protocols for Stability Assessment
Standardized Long-Term Stability Testing Protocol
The experimental methodology for evaluating electrode stability under extended operation follows a systematic approach to ensure reproducible and comparable results. For BiFE assessment in antimony detection, the optimized parameters include: accumulation potential (Eads) of -0.10 V, accumulation time (tads) of 60 seconds, and quercetin-5'-sulfonic acid (QSA) concentration of 3.0 μmol L⁻¹ at pH 5.5 [64]. These parameters establish the baseline for subsequent stability measurements through repeated cycling.
The stability testing protocol comprises three distinct phases: initial characterization, accelerated aging through cyclic operation, and post-stability performance validation. During initial characterization, electrodes undergo surface analysis and baseline electrochemical measurements including cyclic voltammetry and electrochemical impedance spectroscopy. The accelerated aging phase involves continuous cycling in the target analyte solution with periodic measurement of key performance indicators including peak current, peak potential shift, and background current. For quantitative assessment, electrodes are subjected to a minimum of 50 measurement cycles with performance metrics recorded at cycles 1, 5, 10, 25, and 50. The final validation phase compares pre- and post-stability detection limits, sensitivity, and reproducibility to quantify performance degradation [64].
Interference Testing Methodology
The interference resistance evaluation follows a systematic approach where potential interferents are introduced individually and in combination to simulate real-world conditions. For metal ion interference assessment, the protocol involves measuring the target analyte response in the presence of increasing concentrations of potential interfering ions, with tolerance limits defined as the concentration causing less than 5% signal variation. Organic interference testing incorporates surface-active compounds including surfactants, humic acids, and proteins to evaluate fouling resistance. The electrode regeneration protocols are critically evaluated between measurements, assessing both simple rinsing with buffer solution and more aggressive electrochemical cleaning procedures when necessary [64].
Diagram 1: Electrode stability and interference resistance assessment workflow
Electrochemical Signaling Pathways and Mechanisms
Charge Transfer and Interference Mechanisms
The fundamental signaling pathways in electrochemical detection involve complex charge transfer mechanisms that directly influence both sensitivity and stability. For BiFE systems, the detection mechanism typically involves adsorptive accumulation followed by reduction, where the analyte complexes with organic ligands such as quercetin-5'-sulfonic acid (QSA) before electrochemical reduction [64]. This complexation step provides inherent selectivity by creating a specific molecular recognition event preceding the charge transfer process.
The interference resistance pathways differ significantly between electrode materials. BiFE exhibits interference resistance through selective complexation and optimized accumulation potentials that preferentially attract target analytes while excluding interferents. HgFE traditionally offered broad-spectrum detection capabilities but suffered from limited stability due to mercury oxidation and surface renewal requirements. AuFE provides excellent conductivity but is prone to fouling through non-specific adsorption, particularly in complex matrices containing organic compounds or proteins. The stability of each electrode material under long-term operation is directly linked to these fundamental signaling and interference rejection pathways [64].
Diagram 2: Comparative signaling and interference pathways for BiFE, HgFE, and AuFE
Essential Research Reagent Solutions
Table 3: Key research reagents and materials for electrode stability assessment
| Reagent/Material | Function in Stability Assessment | Application Specifics |
|---|---|---|
| Quercetin-5'-sulfonic acid (QSA) | Complexing agent for enhanced selectivity and sensitivity [64] | 3.0 μmol L⁻¹ concentration for Sb(III) detection |
| Bismuth precursor solutions | BiFE formation and renewal | Nitrate or chloride salts in buffer solutions |
| Supporting electrolytes | Provide ionic strength and pH control | Acetate buffer (pH 5.5) for optimal QSA performance [64] |
| Standard analyte solutions | Performance calibration and quantification | Freshly prepared from certified reference materials |
| Interferent stock solutions | Controlled interference studies | Metal ions, surfactants, organic compounds |
| Electrode polishing materials | Surface regeneration and preparation | Alumina slurries, polishing pads |
| Membrane components | Surface modification for enhanced selectivity | Nafion, chitosan, or other polymeric coatings |
The stability and reproducibility assessment under long-term operation reveals distinct advantages and limitations for each electrode material within the context of interference resistance. BiFE demonstrates superior performance in terms of environmental compatibility, reproducibility, and stability when properly modified with complexing agents such as QSA. The experimental data indicates that BiFE achieves detection limits comparable to historical HgFE performance while offering significantly improved operational stability and reduced toxicity. HgFE, while providing excellent historical performance data, shows limitations in long-term stability and environmental compatibility. AuFE exhibits vulnerability to fouling in complex matrices but may offer advantages in specific applications where its electrocatalytic properties can be leveraged. The comprehensive comparison provided in this assessment offers researchers evidence-based criteria for electrode selection based on the specific requirements of their analytical applications, particularly when long-term stability and interference resistance are paramount considerations.
The development of sensitive and reliable detection platforms is a cornerstone of modern biomedical research and diagnostic testing. Within this field, a critical challenge involves selecting electrode materials that optimize performance while minimizing toxicity and ensuring practical applicability. This guide provides an objective comparison of three electrode materials—Bismuth-film electrodes (BiFE), Mercury-film electrodes (HgFE), and Gold-film electrodes (AuFE)—framed within the broader thesis of interference resistance. The performance of electrochemical biosensors is highly dependent on the working electrode material, which influences sensitivity, selectivity, and stability. This analysis compares these materials based on experimental data concerning their analytical performance, inherent toxicity, and practicality for various applications, providing researchers with a structured framework for material selection.
Performance Comparison of Electrode Materials
The selection of an electrode material involves balancing multiple performance characteristics. The following table summarizes key attributes of BiFE, HgFE, and AuFE based on published experimental data.
Table 1: Comparative analysis of BiFE, HgFE, and AuFE electrode materials.
| Feature | Bismuth-Film Electrodes (BiFE) | Mercury-Film Electrodes (HgFE) | Gold-Film Electrodes (AuFE) |
|---|---|---|---|
| Electrochemical Performance | Wide linear range, low background current, well-defined stripping signals | Excellent cathodic potential range, high sensitivity for heavy metals | High conductivity, superior electrocatalytic activity, reusable |
| Interference Resistance | High resistance to surface fouling and organic contaminants | Susceptible to oxidation and interference from surface-active compounds | Prone to interference from adsorption of large biomolecules |
| Toxicity & Environmental Impact | Low toxicity, environmentally friendly alternative [65] | Highly toxic, posing health and environmental risks [66] [67] | Non-toxic and biocompatible |
| Practicality & Cost | Low-cost, easy fabrication, suitable for disposable sensors [65] | Requires special handling and waste disposal, increasing operational cost | High material cost, but offset by reusability in some applications |
| Optimal Use Cases | Point-of-care testing, environmental monitoring, food safety [65] | Laboratory-based trace metal analysis (historical use) | SPR-based biosensing, implantable sensors, fundamental research [22] |
Experimental Protocols for Performance Evaluation
To ensure the comparability of data presented in the previous section, standardized experimental protocols are essential. The following methodologies are commonly employed to quantify the performance and interference resistance of electrode materials.
Stripping Voltammetry for Heavy Metal Detection
This protocol is a benchmark for comparing the sensitivity and reproducibility of BiFE and HgFE.
- Objective: To quantify trace levels of heavy metal ions (e.g., Cd²⁺, Pb²⁺, Zn²⁺) and compare the analytical performance of BiFE and HgFE.
- Materials: Electrochemical cell, potentiostat, BiFE/HgFE working electrode, Ag/AgCl reference electrode, platinum counter electrode, acetate buffer (pH 4.5), standard solutions of metal ions.
- Procedure:
- Electrode Preparation: A bismuth film is electroplated in-situ by adding a Bi(III) salt to the sample solution, or a mercury film is pre-plated on a substrate.
- Pre-concentration: The solution is stirred, and a negative deposition potential is applied to reduce and deposit metal ions onto the electrode surface for a fixed time.
- Stripping Scan: Stirring is stopped, and a positive-going voltammetric scan (e.g., Differential Pulse Voltammetry) is applied, oxidizing the metals back into solution.
- Measurement: The resulting current peak is measured; its height is proportional to the concentration of the metal ion.
- Data Analysis: Calibration curves are constructed by plotting peak current against concentration. Limits of detection (LOD), sensitivity, and linear dynamic range are calculated and compared between BiFE and HgFE.
Interference Testing with Complex Matrices
This protocol assesses the robustness and interference resistance of the electrode surfaces.
- Objective: To evaluate the impact of common interferents (e.g., proteins, surfactants, other metal ions) on sensor signal.
- Materials: Prepared biosensor (e.g., AuFE-based SPR or electrochemical platform), target analyte (e.g., C-Reactive Protein), potential interferents (e.g., Bovine Serum Albumin, human serum samples).
- Procedure:
- Baseline Measurement: The sensor's response to a pure buffer solution is recorded.
- Analyte Response: The sensor's response to a known concentration of the target analyte is measured.
- Interference Challenge: The sensor's response is measured in the presence of both the target analyte and a high concentration of potential interferents.
- Control Experiment: The response from the interferent alone is also measured.
- Data Analysis: The signal recovery is calculated by comparing the analyte response in the presence and absence of interferents. A recovery close to 100% indicates high interference resistance. For fiber-optic sensors, a wavelength shift greater than 3266.78 nm/RIU indicates high sensitivity to non-specific binding, which is a form of interference [22].
Signaling Pathways and Experimental Workflows
The following diagrams illustrate the logical workflow for electrode selection and the experimental setup for a key detection method.
Diagram 1: Electrode material selection workflow.
Diagram 2: Generalized biosensing experimental workflow.
The Scientist's Toolkit: Essential Research Reagents
Successful experimentation requires specific materials and reagents. The following table details key components used in the development and testing of electrodes and biosensors.
Table 2: Key reagents and materials for electrode and biosensor experiments.
| Reagent/Material | Function in Experiment | Example Application |
|---|---|---|
| Glutaraldehyde | Cross-linking agent for antibody immobilization on sensor surfaces. | Used to covalently bind CRP antibodies to the surface of a tapered-in-tapered fiber-optic biosensor [22]. |
| C-Reactive Protein (CRP) & Antibodies | Model analyte and bio-recognition element for immunoassay development. | Serves as the target biomarker in biosensor performance validation [22]. |
| Acetate Buffer (pH 4.5) | Electrolyte solution that provides optimal pH conditions for metal deposition. | Used as the supporting electrolyte in anodic stripping voltammetry for heavy metal detection. |
| Bismuth (III) Salts | Precursor for the formation of bismuth-film working electrodes. | In-situ electroplating of BiFE for trace metal analysis, providing a non-toxic alternative to HgFE [65]. |
| Standard Metal Ion Solutions | Calibration standards for quantifying analytical performance. | Used to create calibration curves for Cd²⁺, Pb²⁺, and Zn²⁺ in stripping voltammetry. |
| Johnson-Cook Model Parameters | Constitutive material model for simulating high-strain-rate deformation. | Used in finite element analysis to study the impact behaviour of materials like Al 2024 alloy [68] [69]. |
Conclusion
This systematic comparison elucidates that while HgFEs historically set a high bar for sensitivity and reproducibility, their toxicity and fouling propensity are significant drawbacks. BiFEs emerge as a premier, environmentally friendly alternative with exceptional resistance to common anionic interferents, making them ideal for sustainable sensing applications. AuFEs demonstrate superior stability and ease of functionalization, particularly valuable in complex, protein-rich media. The optimal choice of electrode material is not universal but is dictated by the specific analytical matrix and target analyte. Future directions should focus on the development of hybrid and novel nanocomposite films that synergize the strengths of these materials, alongside the integration of AI for real-time interference correction, paving the way for more robust and intelligent biosensing systems in biomedical research and clinical diagnostics.
References
- 1. Multi-pulse galvanostatic preparation of nanostructured ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400517301697]
- 2. Electrochemical Detection of Cadmium Using a Bismuth ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11722831/]
- 3. Evaluation of Electrical Performance and Properties ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7414618/]
- 4. SURFACE CHEMISTRY INFLUENCE IMPLANT ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3230929/]
- 5. Biocompatible and bioactive surface modifications for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3878818/]
- 6. Surface chemistry influences implant biocompatibility - PubMed [https://pubmed.ncbi.nlm.nih.gov/18393890/]
- 7. Jet blown PTFE for control of biocompatibility [https://ui.adsabs.harvard.edu/abs/2009PhDT.......258L/abstract]
- 8. Electrochemical impedance behavior and corrosion ... [https://www.sciencedirect.com/science/article/pii/S2352492825006907]
- 9. Comparison on the immersion corrosion and electrochemical ... [https://pubs.rsc.org/en/content/articlehtml/2021/ra/d1ra03549e]
- 10. Electrochemical Sensor for Biological Samples Monitoring [https://link.springer.com/article/10.1007/s11244-025-02080-5]
- 11. Classification of Continuous Glucose Monitoring Devices ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12614895/]
- 12. Nikolaj A. Yashtulov [https://www.sciencedirect.com/author/6507694451/nikolaj-a-yashtulov]
- 13. emphasizing green, white, and blue analytical approaches [https://www.nature.com/articles/s41598-024-69518-w]
- 14. Electrochemical sensors and devices for heavy metals assay ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4012207/]
- 15. Polymer-coated bismuth film electrodes for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267006012323]
- 16. Exploiting Chemistry to Improve Performance of Screen- ... [https://www.mdpi.com/2079-6374/6/3/38]
- 17. Antimony- based electrodes for analytical determinations [https://www.sciencedirect.com/science/article/abs/pii/S0165993615301825]
- 18. Comparison between Different Configurations of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11886708/]
- 19. Electrochemical Detection of Cadmium Using a Bismuth ... [https://www.mdpi.com/1424-8220/25/1/159]
- 20. Core–shell AuFe@FeOx-CFC as electrochemical sensor ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400520306675]
- 21. Development of a flow analysis system based on an ... [https://www.sciencedirect.com/science/article/pii/S2213343725020160]
- 22. Enhanced Sensitivity Mach–Zehnder Interferometer-Based ... [https://www.mdpi.com/2079-6374/15/2/90]
- 23. Molecularly imprinted polymer (MIP) based ... [https://www.sciencedirect.com/science/article/pii/S2666053923000164]
- 24. Biosensors, Volume 15, Issue 11 (November 2025) [https://www.mdpi.com/2079-6374/15/11]
- 25. Imperceptible augmentation of living systems with organic ... [https://www.nature.com/articles/s41928-024-01174-4]
- 26. Low-cost food-grade alternatives for serum albumins in ... [https://www.nature.com/articles/s41598-025-99603-7]
- 27. The State-of-the-Art Functionalized Nanomaterials for ... [https://www.mdpi.com/2077-0375/12/2/186]
- 28. Emerging nanomaterial incorporated membranes for gas ... [https://www.sciencedirect.com/science/article/pii/S2772823421000166]
- 29. Effect of Nanofillers on Properties and Pervaporation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9785865/]
- 30. Review of nanomaterials-assisted ion exchange ... [https://www.nature.com/articles/s41545-018-0009-7]
- 31. bioAffinity Technologies Reports Second Quarter 2025 Results [https://ir.bioaffinitytech.com/news-events/press-releases/detail/147/bioaffinity-technologies-reports-second-quarter-2025-results]
- 32. full-year earnings outlook raised [https://www.roche.com/media/releases/med-cor-2025-10-23]
- 33. About Antimicrobial Resistance | Antimicrobial Resistance | CDC [https://www.cdc.gov/antimicrobial-resistance/about/index.html]
- 34. How Does Ascorbic Acid Interfere with Blood Biomarkers? [https://www.optimaldx.com/research-blog/how-does-ascorbic-acid-interfere-with-blood-biomarkers]
- 35. Ascorbic acid interference in the measurement of serum ... [https://pubmed.ncbi.nlm.nih.gov/16403487/]
- 36. Ascorbic acid and glucose can cause significant ... [https://pubmed.ncbi.nlm.nih.gov/39550214/]
- 37. Investigations of ascorbic acid interference in urine test strips [https://hero.epa.gov/hero/index.cfm/reference/details/reference_id/504233]
- 38. Uric Acid in Heart Failure: A Biomarker or Therapeutic ... [https://pubmed.ncbi.nlm.nih.gov/22584465/]
- 39. Serum Uric Acid level in the severity of Congestive Heart ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5432699/]
- 40. Bingeing on High-Fat Food Enhances Evoked Dopamine ... [https://pubmed.ncbi.nlm.nih.gov/33660412/]
- 41. Bingeing on High‐Fat Food Enhances Evoked Dopamine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8048651/]
- 42. Bingeing on High-Fat Food Enhances Evoked Dopamine ... [https://www.merckmillipore.com/ST/en/tech-docs/paper/1474807]
- 43. Modulation of Protein Fouling and Interfacial Properties at ... [https://www.nature.com/articles/srep24840]
- 44. Protein adsorption measurements on low fouling and ... [https://www.sciencedirect.com/science/article/abs/pii/S1742706119307603]
- 45. A new linear growth theoretical model of protein reversible ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135425017695]
- 46. Enhanced anti-fouling performance through integrated ... [https://www.sciencedirect.com/science/article/pii/S1383586625000048]
- 47. Unraveling the anti-biofouling mechanisms of slippery ... [https://www.sciencedirect.com/science/article/abs/pii/S0021979725003340]
- 48. Nature-inspired surface modification strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11925587/]
- 49. Advanced protein-based hydrophilic coatings for anti- ... [https://pubmed.ncbi.nlm.nih.gov/41005390/]
- 50. Application-driven design of non-aqueous electrolyte solutions through... [https://www.nature.com/articles/s41565-025-01935-y?error=cookies_not_supported&code=999a62a1-d1d3-4473-985b-bd3dae7dfe53]
- 51. sciencedirect.com/science/article/abs/pii/S1385894723045333 [https://www.sciencedirect.com/science/article/abs/pii/S1385894723045333]
- 52. Electrolyte Design Strategies for Non-Aqueous High ... [https://www.mdpi.com/1420-3049/28/2/823]
- 53. Controlled hydrolysis of LiPF6-based electrolytes with trace... [https://www.sciopen.com/article/10.26599/NR.2025.94907475]
- 54. Research on performance constraints and electrolyte ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11912001/]
- 55. A General Method for Regenerating Catalytic Electrodes [https://www.sciencedirect.com/science/article/pii/S2542435120303895]
- 56. Double Step Electrochemical Etching for Reuse of Gold ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12547596/]
- 57. A brief review on methods and materials for electrode ... [https://link.springer.com/article/10.1007/s44373-024-00012-8]
- 58. Limit of Blank, Limit of Detection and Limit of Quantitation [https://pmc.ncbi.nlm.nih.gov/articles/PMC2556583/]
- 59. What is meant by the limit of detection and quantification (LOD ... [https://mpl.loesungsfabrik.de/en/english-blog/method-validation/limit-of-detection-quantifcation]
- 60. Performance metrics and enabling technologies for ... [https://www.nature.com/articles/s41467-018-06419-3]
- 61. Biosensor-Integrated Drug Delivery Systems as New ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9496590/]
- 62. Startup's biosensor makes drug development and ... [https://news.mit.edu/2025/startup-biosensor-makes-drug-development-and-manufacturing-cheaper-0615]
- 63. Frontiers | Electrochemical sensors and devices for heavy metals assay... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2014.00019/full]
- 64. High sensitivity adsorptive stripping voltammetric method ... [https://www.academia.edu/89158030/High_sensitivity_adsorptive_stripping_voltammetric_method_for_antimony_III_determination_in_the_presence_of_quercetin_5_sulfonic_acid_Substituent_effect_on_sensitivity]
- 65. Electrochemical Biosensing for Antibiotic-Resistant Bacteria [https://pmc.ncbi.nlm.nih.gov/articles/PMC12471839/]
- 66. twenty-eight-day and ninety-day oral gavage studies. [https://www.sigmaaldrich.com/SK/en/tech-docs/paper/272163?srsltid=AfmBOooRbL0BMxJZoropHcJLV6nWSp49rxKMC6UmGxfmAVUJkfZZWna1]
- 67. The reproductive toxicology of ammonium ... [https://pubmed.ncbi.nlm.nih.gov/15036760/]
- 68. Effect of interference magnitude on ultimate strength and ... [https://www.sciencedirect.com/science/article/abs/pii/S1350630722002552]
- 69. Analytical and FEM Analyses of High-Speed Impact ... [https://www.mdpi.com/2226-4310/8/10/281]