A Practical Guide to Validating Electrochemical Methods for USP and EP Compliance
This article provides a comprehensive guide for researchers and drug development professionals on validating electrochemical methods to meet United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards.
A Practical Guide to Validating Electrochemical Methods for USP and EP Compliance
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on validating electrochemical methods to meet United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards. It covers foundational regulatory principles, methodological applications for drug substance and product analysis, strategies for troubleshooting and optimization, and the execution of rigorous validation protocols. By synthesizing current guidelines and practical case studies, this resource aims to equip scientists with the knowledge to implement robust, compliant electrochemical procedures that ensure drug safety, quality, and efficacy from development through post-marketing surveillance.
Understanding the Regulatory Landscape: USP and EP Fundamentals for Electrochemical Analysis
The Roles of USP and EP in Defining Pharmaceutical Quality Standards
In the global pharmaceutical industry, the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP) serve as the foundational pillars for ensuring drug quality, safety, and efficacy. These compendial standards provide the scientific and regulatory frameworks that govern pharmaceutical development, manufacturing, and quality control worldwide. Regulatory authorities, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), enforce compliance with these standards as part of Good Manufacturing Practice (GMP) requirements [1]. For researchers and drug development professionals, understanding the distinct roles, similarities, and differences between USP and EP is crucial for navigating international regulatory landscapes and establishing robust quality systems.
Both pharmacopeias establish legally enforceable standards that define pharmaceutical quality through detailed monographs, general chapters, and reference materials. While USP standards are enforceable under U.S. federal law, EP standards form the legal foundation for product approval within the European Union and its member states [1]. This article provides a comprehensive comparative analysis of USP and EP, with particular focus on their application in analytical method validation, to guide scientific professionals in maintaining compliance across international markets.
Comparative Analysis: USP vs. EP Organizational Structures and Governance
Organizational Backgrounds and Legal Status
The following table summarizes the fundamental organizational characteristics of USP and EP:
Table 1: Organizational Profiles of USP and EP
| Characteristic | United States Pharmacopeia (USP) | European Pharmacopoeia (EP) |
|---|---|---|
| Governing Body | Independent, non-profit scientific organization | European Directorate for the Quality of Medicines & HealthCare (EDQM) |
| Establishment | 1820 [2] | Published by EDQM [2] |
| Legal Status | Enforceable by FDA under U.S. federal law [1] | Legally binding in EU member states under EMA [1] |
| Primary Document | United States Pharmacopeia-National Formulary (USP-NF) | European Pharmacopoeia (Ph. Eur.) |
| Geographic Scope | Primarily United States, with global influence [1] | European Union and member states, with international recognition [1] |
| Reference Standards | Highly characterized specimens for drug substances, excipients, impurities, etc. [3] | Highly purified reference substances for quality testing [2] |
Scope and Coverage in Pharmaceutical Testing
Both pharmacopeias provide comprehensive quality specifications covering:
- Monographs: Detailed specifications for drug substances, excipients, and finished products defining identity, strength, purity, and quality [1].
- General Chapters: Mandatory and informational texts describing analytical procedures and methods [4] [2].
- Reference Standards: Physical reference materials essential for conducting compendial tests and verifying method performance [3] [2].
While their fundamental objectives align, differences emerge in specific methodological requirements, acceptance criteria, and implementation guidelines that laboratories must navigate for products marketed in multiple regions.
Analytical Method Validation: A Comparative Framework
Core Validation Parameters and Requirements
Analytical method validation establishes evidence that a method is fit for its intended purpose. Both USP and EP provide detailed guidance on validation parameters, though with some distinctions in emphasis and implementation:
Table 2: Comparison of Analytical Method Validation Parameters
| Validation Parameter | USP Requirements | EP Requirements | Application in Electroanalytical Methods |
|---|---|---|---|
| Specificity | Required to demonstrate ability to assess analyte in presence of matrix | Similarly required with focus on impurity interference | Essential for electrode selectivity in complex matrices [5] |
| Accuracy | Required through recovery studies | Required with statistical significance | Demonstrated via spike recovery in biological matrices [5] |
| Precision | Repeatability and intermediate precision | Repeatability and reproducibility | Cumulative standard addition method for reproducible electrode response [5] |
| Linearity | Required with statistical correlation | Required with defined concentration ranges | Calibration curves with defined linear ranges for electroanalytical quantification [5] |
| Range | Specified relative to linearity | Specified relative to intended use | Dependent on electrode modification and detection limits [5] |
| Detection Limit (LOD) | Signal-to-noise or statistical approaches | Similar approaches with possible methodological differences | Critical for sensing applications like hydrochlorothiazide detection [5] |
| Quantitation Limit (LOQ) | Signal-to-noise or precision-based approaches | Similar approaches with defined precision | Monte Carlo Method for uncertainty evaluation at low concentrations [5] |
| Robustness | Often evaluated through experimental design | Systematically evaluated with parameter variations | Particularly important for modified electrodes and sensor platforms [5] |
USP General Chapter <1225>, "Validation of Compendial Procedures," provides the foundational framework for validation in the United States, classifying methods based on required validation data and outlining specific criteria for each parameter [4]. Similarly, EP includes comprehensive guidelines on method validation within its general chapters, with ongoing harmonization efforts between the two pharmacopeias to reduce regulatory divergence [6].
System Suitability Testing and Allowable Adjustments
System suitability tests verify that the analytical system is functioning correctly at the time of analysis. Both pharmacopeias require these tests to ensure method validity [2]. However, differences exist in allowable adjustments to compendial methods, particularly for chromatographic techniques like HPLC.
As noted in a webcast comparing these systems, "There is an on-going effort between USP and Ph. Eur. to harmonize the allowable adjustments for HPLC methods. However, at this time some differences do exist that can make it challenging for users" [6]. These differences can impact laboratory efficiency, particularly for organizations operating in both regulatory jurisdictions. Understanding permitted modifications without requiring full revalidation is essential for optimizing analytical workflows while maintaining compliance.
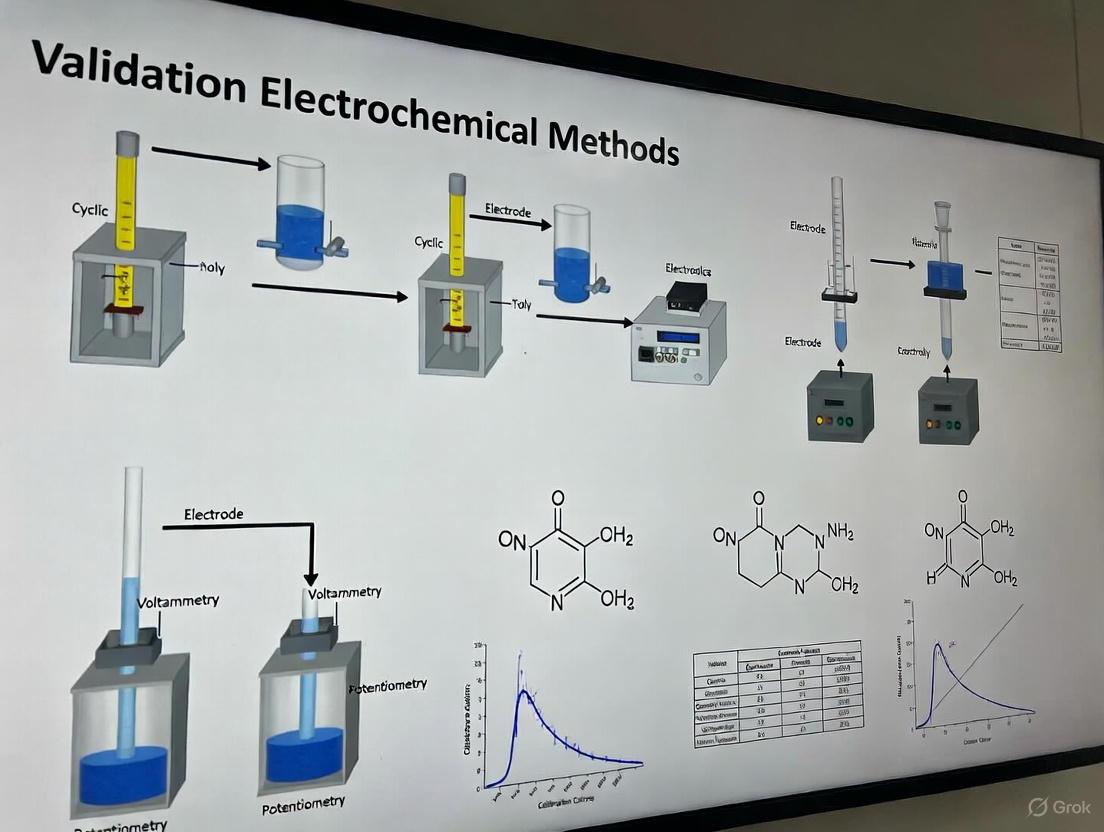
Experimental Protocols for Electroanalytical Method Validation
Development and Optimization of Electrochemical Sensors
Electroanalytical methods play a significant role in pharmaceutical analysis, with both USP and EP providing specific guidance on their implementation [7]. The development of advanced sensors, such as those for determining hydrochlorothiazide in urine, follows a systematic protocol:
- Electrode Modification: A glassy carbon electrode is modified with multiwall carbon nanotubes (MWCNT) and gold nanoparticles to enhance sensitivity and selectivity [5].
- Method Optimization: Experimental parameters including pH, buffer composition, accumulation time, and pulse conditions are optimized using statistical design of experiments.
- Calibration Approach: The sensor is calibrated in the sample matrix using the cumulative standard addition method to account for matrix effects [5].
- Measurement Technique: Differential pulse voltammetry (DPV) is employed for quantitative measurements due to its enhanced sensitivity and resolution.
Validation of Electroanalytical Procedures
The validation of electroanalytical methods follows a structured approach aligned with pharmacopeial requirements:
Diagram 1: Electroanalytical Method Validation Workflow
The validation incorporates several critical steps:
- Specificity: Demonstrated by analyzing potential interfering substances and confirming no significant response compared to the analyte [5].
- Linearity and Range: Established using a minimum of five concentration levels across the claimed range, with statistical evaluation of the calibration model.
- Accuracy: Assessed through spike recovery studies in the actual sample matrix (e.g., urine), with target recoveries typically between 95-105% [5].
- Precision: Evaluated through repeated analysis of quality control samples at multiple concentrations, expressed as relative standard deviation (RSD).
- Detection and Quantitation Limits: For advanced electrochemical sensors, LOD and LOQ are determined based on signal-to-noise ratios (3:1 for LOD, 10:1 for LOQ) or statistical approaches [5].
- Measurement Uncertainty: The Monte Carlo Method (MCM) can be applied for comprehensive uncertainty evaluation, applicable regardless of measurement function linearity [5].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of pharmacopeial standards requires specific reference materials and laboratory resources. The following table details essential research reagents and their functions in pharmaceutical quality control:
Table 3: Essential Research Reagents and Materials for USP/EP Compliance
| Reagent/Material | Function | Pharmacopeial Application |
|---|---|---|
| USP Reference Standards | Highly characterized specimens for drug identification, impurity analysis, and quality testing [3] | Required for compendial assays and method validation; supports ~3,500 available standards [3] |
| EP Reference Standards | Highly purified substances for qualitative and quantitative drug composition analysis [2] | Essential for identity testing, purity assessment, and analytical procedure verification |
| EP Impurity Standards | Certified reference materials for impurity identification and quantification [2] | Critical for assessing drug purity and identifying potentially harmful substances |
| Nitrosamine Impurities | Specific impurity standards for genotoxic compound detection [3] | Supports testing for potentially carcinogenic impurities in pharmaceutical products |
| Performance Verification Standards | Materials for verifying analytical instrument performance [3] | Used in system suitability testing and equipment qualification per USP <1058> [2] |
| Multiwall Carbon Nanotubes | Electrode modification material for enhanced sensitivity [5] | Used in developing advanced electrochemical sensors for drug quantification |
| Gold Nanoparticles | Nanomaterial for electrode surface modification [5] | Enhances electron transfer and catalytic activity in electroanalytical methods |
| Compendial Reagents | Qualified chemicals and solutions specified in monographs [3] | Ensure accuracy and reproducibility of compendial testing methods |
Regulatory Integration and Compliance Strategies
Implementation in Quality Control Laboratories
Effective integration of USP and EP standards into laboratory operations requires systematic approaches:
- Method Selection and Verification: Laboratories must use compendial methods as default procedures. When adapting methods for internal use, they must validate or verify them according to pharmacopeial guidelines [1].
- Instrument Qualification: USP General Chapter <1058> requires laboratories to conduct qualification and calibration of analytical instruments to ensure proper performance [2].
- Documentation Practices: Standard Operating Procedures (SOPs) must mirror USP/EP requirements, with complete and traceable experimental records maintained for audit purposes [1] [2].
- Change Control Systems: Laboratories must establish robust processes to manage updates to pharmacopeial standards, including timely revision of SOPs and retraining of personnel [1].
Addressing Global Harmonization Challenges
Despite ongoing harmonization efforts, differences between USP and EP present challenges for international operations. These include variations in allowable method adjustments, acceptance criteria, and validation requirements [6]. Laboratories operating in multiple regions should:
- Implement a compendial liaison to monitor and interpret updates from both organizations [1].
- Develop method transfer protocols that address requirements of both pharmacopeias [6].
- Conduct comparative assessments during method development to identify potential compliance issues early.
- Participate in industry working groups that contribute to harmonization initiatives between USP and EP.
USP and EP play indispensable, complementary roles in defining and maintaining pharmaceutical quality standards globally. While both share the common goal of ensuring drug safety and efficacy, differences in their specific requirements necessitate careful navigation by researchers and quality professionals. The ongoing harmonization efforts between these organizations promise to reduce regulatory divergence and enhance global cooperation.
For the scientific community, success in this evolving landscape requires not only strict adherence to current standards but also active engagement with the scientific principles underpinning them. By embracing the robust frameworks provided by both pharmacopeias, particularly in emerging areas like advanced electroanalytical methods, researchers can contribute to the continuous advancement of pharmaceutical quality while accelerating the development of vital medicines for patients worldwide.
For pharmaceutical researchers and drug development professionals, the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) represent the cornerstone of quality control standards. These documents provide the legally binding requirements for the identity, strength, quality, and purity of medicines, ensuring consistent quality and safety for patients worldwide [8]. The validation of analytical procedures, including electrochemical methods, is a fundamental requirement within both frameworks, demonstrating that methods are fit for their intended purpose and yield reliable results [9].
This guide provides a comparative analysis of key documents, focusing on the evolving USP General Chapter <1225> and relevant EP General Texts. With the recent publication of a proposed revision for USP <1225>, understanding the alignment with international guidelines and the shift towards a lifecycle approach is crucial for modern laboratories [10] [11].
United States Pharmacopeia (USP) - General Chapter <1225>
USP <1225>, titled "Validation of Compendial Procedures," is the primary guideline for validating analytical methods in the United States. A significant proposed revision is currently open for comment until January 31, 2026, which will rename the chapter to "Validation of Analytical Procedures" to reflect its broader application for both compendial and non-compendial methods [10].
The revision aims to closely align the chapter with the principles of ICH Q2(R2) on analytical procedure validation and integrate it more clearly into the analytical procedure lifecycle described in USP <1220> [10] [11]. This represents a paradigm shift from treating validation as a one-time event to managing it as a dynamic, ongoing process [11].
Key Concepts in the Revised USP <1225>
- Reportable Result (RR): Emphasized as the definitive output supporting batch release and compliance decisions, shifting focus to the final reported value rather than individual measurements [10] [11].
- Fitness for Purpose: Positioned as the overarching goal of validation, focusing on the confidence in decision-making rather than merely checking isolated parameters [10].
- Replication Strategy: Now linked to controlling the uncertainty of the Reportable Result, moving beyond simple predefined numbers of injections [10].
- Combined Evaluation: Statistical intervals (confidence, prediction, tolerance) are introduced as tools for evaluating precision and accuracy in relation to decision risk [10].
European Pharmacopoeia (EP) - General Texts
The European Pharmacopoeia (Ph. Eur.) is the official pharmacopoeia of the European Union and is legally binding in its 38 member states and beyond [12]. The current edition is the 11th Edition, which became effective on January 1, 2023 [13]. The EP consists of monographs and general chapters. Monographs specify quality requirements for specific active substances and finished products, while general chapters provide horizontal standards applicable to multiple products, including methods of analysis and requirements for quality control [12].
The EP does not have a single, direct equivalent to USP <1225>. Instead, its requirements for analytical procedure validation are embedded within its general chapters. The EP's general texts are critical for ensuring that the quality requirements in the monographs are met [12]. The EP is regularly updated, with the 10th Edition featuring over 2,420 monographs and 374 general texts [12].
Table: Comparison of USP and EP Key Documents and Concepts
| Feature | United States Pharmacopeia (USP) | European Pharmacopoeia (EP) |
|---|---|---|
| Primary Validation Chapter | <1225> Validation of Compendial Procedures (proposed: Validation of Analytical Procedures) [10] | Requirements are embedded across various general chapters [12]. |
| Current Edition/Status | Proposed revision in PF 51(6); comment period until Jan 31, 2026 [10] | 11th Edition (effective Jan 1, 2023) [13] |
| Legal Status | Legally recognized under U.S. Federal Food, Drug, and Cosmetic Act [9] | Legally binding in 38 member states and the European Union [12] [13] |
| Core Philosophy | Evolving towards a lifecycle approach, aligning with ICH Q2(R2) and Q14 [10] [11] | Provides a set of comprehensive, legally enforceable quality standards [12] |
| Key Emphasis | Reportable Result, Fitness for Purpose, Control Strategy [10] | Monograph compliance, harmonized standards across member states [12] |
| Related Chapters | <1220> Analytical Procedure Life Cycle, <1210> Statistical Tools [10] | General chapters on quality management, analytical methods, and validation [12] |
Experimental Protocols and Validation Parameters
The validation of an analytical method is the process that establishes, through laboratory studies, that its performance characteristics meet the requirements for the intended application [9]. The following protocols outline the core parameters required by pharmacopeial standards.
Standard Validation Protocol for an Assay Method
The workflow below illustrates the typical stages of an analytical procedure lifecycle, from development through to ongoing verification, as endorsed by modern regulatory thinking.
Accuracy
- Definition: The closeness of test results obtained by the method to the true value [9].
- Protocol: For drug substance assays, apply the method to an analyte of known purity (e.g., a Reference Standard). For drug products, use synthetic mixtures of the product components spiked with known amounts of the analyte [9].
- Experimental Data: Accuracy is calculated as the percentage of recovery of the known added amount. ICH recommends a minimum of nine determinations over a minimum of three concentration levels covering the specified range [9].
Precision
- Definition: The degree of agreement among individual test results when the method is applied repeatedly to multiple samplings of a homogeneous sample. Precision has three tiers: repeatability (same analyst, same day), intermediate precision (different days, different analysts), and reproducibility (between laboratories) [9].
- Protocol: Assay a sufficient number of aliquots of a homogeneous sample to calculate statistically valid estimates of standard deviation or relative standard deviation (coefficient of variation) [9].
- Experimental Data: For repeatability, ICH recommends a minimum of nine determinations covering the specified range (e.g., three concentrations and three replicates each) or a minimum of six determinations at 100% of the test concentration [9].
Specificity
- Definition: The ability to assess the analyte unequivocally in the presence of components that may be expected to be present, such as impurities, degradation products, and matrix components [9].
- Protocol: For assays, specificity is demonstrated by spiking the drug substance or product with appropriate levels of impurities or excipients and showing that the assay result is unaffected. If impurity standards are unavailable, results are compared to a second, well-characterized procedure [9].
Linearity and Range
- Definition:
- Linearity: The ability of the method to obtain test results proportional to the concentration of the analyte.
- Range: The interval between the upper and lower levels of analyte that have been demonstrated to be determined with suitable precision, accuracy, and linearity [9].
- Protocol: Prepare and analyze a series of standard solutions across the claimed range (e.g., 50% to 150% of the target concentration for assay). The response is plotted against concentration, and statistical methods are used to evaluate the linearity of the data [9].
Table: Validation Characteristics for Different Types of Analytical Procedures [9]
| Validation Characteristic | Identification | Testing for Impurities | Assay |
|---|---|---|---|
| Accuracy | - | + | + |
| Precision | - | + | + |
| Specificity | + | + | + |
| Detection Limit | - | +* | - |
| Quantitation Limit | - | + | - |
| Linearity | - | + | + |
| Range | - | + | + |
| Key: + signifies that characterization is generally needed; - signifies that it is generally not needed; * may be required if the test is used for quantitative purposes. |
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table details key materials and solutions essential for conducting validation studies per USP and EP standards.
Table: Essential Research Reagents and Materials for Pharmacopeial Validation
| Item | Function & Importance in Validation |
|---|---|
| USP/EP Reference Standards | Highly purified and characterized substances used to calibrate instruments and validate methods. They are essential for establishing accuracy, specificity, and for use in system suitability tests [8]. |
| Analytical Instruments (HPLC, UV-Vis) | Qualified instruments (per USP <1058>) are fundamental for generating reliable data. They are used across all validation parameters, from measuring linearity and range to determining precision [8]. |
| System Suitability Test (SST) Materials | Mixtures or solutions used to verify that the chromatographic or analytical system is adequate for the intended analysis. SST is a required check before validation runs and routine testing begin [8]. |
| Reagents and Solvents (Pharmacopeial Grade) | High-purity reagents, solvents, and water (per EP's monograph on pharmaceutical water) are critical to prevent interference, ensure method robustness, and achieve the required detection limits [12] [8]. |
| Impurity Standards | Isolated and qualified impurities, degradation products, or related compounds. These are vital for validating the specificity and quantitation limit of impurity methods, a key focus of both USP and EP [8]. |
The landscape of pharmacopeial standards is dynamic. The ongoing revision of USP <1225> marks a significant shift towards a more holistic, risk-based lifecycle approach, closely aligning with ICH Q2(R2) and emphasizing concepts like "Fitness for Purpose" and the control of the "Reportable Result" [10] [11]. In parallel, the European Pharmacopoeia continues to update its monographs and general texts to reflect scientific progress, as seen in its 11th Edition [13].
For researchers validating electrochemical methods or any analytical procedure, success hinges on a deep understanding of both frameworks. This involves not only rigorous adherence to experimental protocols for accuracy, precision, and specificity but also an embrace of knowledge management and ongoing performance verification. By leveraging the essential reagents and understanding the interconnectedness of development, validation, and control, scientists can ensure their methods are not only compliant but also robust and reliable throughout the product lifecycle.
Core Principles of Quality by Design (QbD) and its Impact on Method Validation
Quality by Design (QbD) is a systematic, scientific approach to pharmaceutical development and manufacturing that begins with predefined objectives and emphasizes product and process understanding and control [14]. In the context of analytical method validation, QbD shifts the paradigm from a one-time, documentation-focused exercise to a holistic lifecycle approach that ensures methods remain fit-for-purpose throughout their use [15]. This approach aligns with regulatory guidance from the FDA and ICH, which now encourages "the collection and evaluation of data and knowledge from the method design stage throughout its lifecycle of use, which establishes scientific evidence that a method is capable of consistently delivering quality data" [15].
The application of QbD principles to analytical method development is particularly crucial for modern techniques like electrochemical methods, which offer advantages in sensitivity, speed, and cost-effectiveness but require robust validation to gain regulatory acceptance [16] [17]. As pharmacopeial standards such as USP and EP evolve through harmonization efforts like the Pharmacopeial Discussion Group (PDG), understanding QbD's core principles becomes essential for researchers and drug development professionals aiming to develop reliable, validated analytical methods [18].
Core Principles of QbD
The Foundation of QbD
The QbD framework is built upon predefined objectives and emphasizes understanding based on sound science and quality risk management [14] [15]. This systematic approach to development ensures that quality is built into the product and process from the beginning, rather than merely tested at the end. The International Conference on Harmonisation (ICH) outlines the exact elements of QbD, which include [14]:
- A Quality Target Product Profile (QTPP) that sheds light on the product's Critical Quality Attributes (CQAs)
- Product design and understanding that includes identification and further exploration of the product's Critical Material Attributes (CMAs)
- Process design and research to identify Critical Process Parameters (CPPs)
- Control Strategies that include specifications for the product and target customer
- Product Lifecycle Management that includes process capability evaluations and continuous improvement
The QbD Implementation Process
Implementing QbD involves a structured process that can be conceptualized in five key stages [14]:
- Define: Summarize the market and target customers, including the product's unique measured goals such as market share, lead times, launch date, performance, price-point, or customer loyalty.
- Discover: Research the market and explore customer needs, connecting those needs to specific, measurable outcomes the product will help achieve.
- Design: Create product features to meet those needs through a creative process that encourages free thinking with appropriate risk controls.
- Develop: Build the processes needed to create those features, ensuring understanding of all variables and ways to ensure quality.
- Deliver: Identify and implement process controls as needed, featuring continuous improvement and careful planning.
This framework ensures that quality considerations are integrated at every stage of development, rather than being addressed as an afterthought.
QbD Implementation Workflow
QbD Versus Traditional Approaches to Method Validation
Fundamental Differences in Philosophy
Traditional method validation and QbD-based validation differ significantly in their fundamental approach and execution. Traditional method validation, typically performed in accordance with ICH Q2 guideline, is often treated as a one-time event with experts who have been involved in developing the method [15]. This approach carries several limitations, including a potential focus on producing validation documentation that will withstand regulatory scrutiny rather than ensuring the method will perform well during routine application. There is also a lack of guidance on how to demonstrate in practice that a method is fit-for-purpose, and the routine operating environment is not always considered during method-development and validation [15].
In contrast, the QbD approach to method validation embraces a lifecycle perspective that aligns with the FDA guidance for process validation. This approach can be conceptualized in three stages: method design, method qualification, and continued method verification [15]. The QbD philosophy recognizes that an analytical method is a process with an output of acceptable quality data, and therefore benefits from the same systematic approach applied to manufacturing processes.
Comparative Analysis: Traditional vs. QbD Approach
Table 1: Comparison of Traditional and QbD-Based Method Validation Approaches
| Aspect | Traditional Approach | QbD-Based Approach |
|---|---|---|
| Philosophy | One-time event; documentation-focused | Lifecycle management; science and risk-based |
| Focus | Regulatory compliance | Method robustness and reliability |
| Knowledge Management | Limited transfer of tacit knowledge | Systematic knowledge management throughout lifecycle |
| Control Strategy | Fixed method parameters | Method operable design space with proven acceptable ranges |
| Continuous Improvement | Limited ongoing verification | Continued method verification and monitoring |
| Regulatory Foundation | ICH Q2 | ICH Q8, Q9, Q10, Q11 |
| Risk Management | Implicit or minimal | Explicit, systematic, and proactive |
QbD Framework for Analytical Method Lifecycle
Stage 1: Method Design
The first stage in the QbD framework for analytical methods is method design, which involves defining method requirements and conditions according to the measurement requirements given in the Analytical Target Profile (ATP) [15]. The ATP acts as the focal point for all stages of the analytical lifecycle and describes the required performance characteristics of the method (e.g., accuracy, precision) and the target criteria for these characteristics [15]. To build the ATP, it is necessary to determine what characteristics will be indicators of method performance and define how accurate or precise the method needs to be, with a key factor being the overall manufacturing process capability [15].
Method design involves selecting appropriate technologies and developing a method that will meet the ATP requirements, then performing studies to understand the critical method variables that need to be controlled to ensure the method is robust and rugged [15]. This understanding phase employs risk assessment tools, such as fishbone diagrams and failure mode effects analysis (FMEA), to determine which variables need studying [15]. Robustness experiments are typically performed on method factors using design of experiments (DoE) to ensure maximum understanding is gained from a minimum number of experiments [15].
Stage 2: Method Qualification
Having determined a set of operational method controls during the design phase, the next step is to qualify that the method will operate in its routine environment as intended [15]. Method qualification involves demonstrating that the defined method, including specified sample and standard replication levels and calibration approaches, will under routine operating conditions produce data that meet the precision and accuracy requirements defined in the ATP [15].
This stage may involve performing a number of replicate measurements of the same sample to confirm that the precision of the method is adequate and to demonstrate that any potential interferences do not introduce an unacceptable bias by comparing results with a sample of known quality [15]. If the respective experimental results have already been obtained during Stage 1, they only need to be summarized for the final evaluation, making the process more efficient.
Stage 3: Continued Method Verification
The goal of the third stage in the method lifecycle is to continually ensure that the method remains in a state of control during routine use [15]. This includes both continuous method performance monitoring of the routine application of the method as well as performance verification following any changes [15]. Continued method performance monitoring should include an ongoing program to collect and analyze data that relate to method performance, such as from replication of samples or standards, by trending system suitability data, assessing precision from stability studies, or by trending data from regular analysis of a reference lot [15].
This activity aligns with the guidance in USP Chapter <1010> on system performance verification and represents a significant advancement over traditional approaches, where method validation was often considered complete after the initial validation exercise [15]. The continued verification stage ensures that methods remain capable and reliable throughout their operational lifetime.
Analytical Method Lifecycle
Impact of QbD on Validation of Electrochemical Methods
Advantages of Electrochemical Methods in Pharmaceutical Analysis
Electrochemical methods offer significant advantages for pharmaceutical analysis, particularly when developed and validated using QbD principles. Recent studies comparing analytical techniques have demonstrated that electroanalytical methods provide rapid response, simple operation, time efficiency, high selectivity, and sensitivity [17]. For example, in the quantification of hydrogen sulfide (H₂S) in aqueous solutions, electrochemical methods were able to quantify H₂S in the nanomole and picomole ranges and were less time-consuming compared to colorimetric and chromatographic methods [16].
Similarly, in the detection and quantification of sunscreen agents like octocrylene (OC) in water matrices, electroanalysis using a glassy carbon sensor (GCS) showed superior performance compared to HPLC, with lower limits of detection (LOD) and quantification (LOQ) [17]. The LOD and LOQ for OC were approximately 0.11 ± 0.01 mg L⁻¹ and 0.86 ± 0.04 mg L⁻¹ by electroanalysis, compared to 0.35 ± 0.02 mg L⁻¹ and 2.86 ± 0.12 mg L⁻¹ by HPLC [17]. These performance characteristics make electrochemical methods particularly attractive for pharmaceutical applications where sensitivity, speed, and cost-effectiveness are critical.
Experimental Data: Method Comparison Studies
Table 2: Comparison of Analytical Techniques for Hydrogen Sulfide Quantification
| Technique | Detection Range | Sample Volume | Time Requirements | Key Advantages |
|---|---|---|---|---|
| Colorimetric | Millimolar to micromolar | 1 ml | Requires 10+ minutes for color development | Relatively simple and inexpensive |
| Chromatographic (HPLC) | Micromolar | 25 μl | ~6 minutes run time | Much greater sensitivity than colorimetric |
| Voltametric | Nanomolar | Varies (electrode dependent) | Rapid measurement after stabilization | High sensitivity, direct measurement |
| Amperometric | Picomolar to nanomolar | 20 ml | Requires polarization (12h) but rapid measurement | Highest sensitivity, direct measurement |
Table 3: Performance Comparison of Electroanalysis vs. HPLC for Octocrylene Detection
| Parameter | Electroanalysis (GCS) | HPLC |
|---|---|---|
| Limit of Detection (LOD) | 0.11 ± 0.01 mg L⁻¹ | 0.35 ± 0.02 mg L⁻¹ |
| Limit of Quantification (LOQ) | 0.86 ± 0.04 mg L⁻¹ | 2.86 ± 0.12 mg L⁻¹ |
| Operational Complexity | Simple and cost-effective | Complex and costly maintenance |
| Sample Pre-treatment | Minimal | Often required |
| Time Efficiency | High | Moderate |
QbD-Enhanced Validation Protocols for Electrochemical Methods
Applying QbD principles to electrochemical method validation involves a systematic approach to establishing the ATP, identifying critical method parameters, and defining the method operable design space. For electrochemical methods, the ATP should define requirements for sensitivity, selectivity, accuracy, precision, and robustness appropriate to the intended application, whether for drug substance testing, impurity profiling, or dissolution testing.
The method design stage for electrochemical techniques should include studies to understand the impact of critical parameters such as electrode type and surface condition, electrolyte composition and pH, temperature, and potential scan rates. Using DoE approaches, the relationship between these input variables and method performance characteristics can be mapped to establish a design space within which the method will reliably meet ATP requirements [15]. Method qualification then verifies that the method operates as intended in the routine environment, while continued verification involves ongoing monitoring of method performance through system suitability tests and quality control samples.
Essential Research Reagents and Materials
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for QbD-Based Electrochemical Method Development
| Reagent/Material | Function | Application Example |
|---|---|---|
| Glassy Carbon Electrode | Working electrode providing wide potential range, low background current, and good surface reproducibility | Quantification of octocrylene in sunscreen products [17] |
| Ag/AgCl Reference Electrode | Provides stable reference potential for accurate potential control and measurement | Essential for all quantitative electrochemical measurements |
| Britton-Robinson Buffer | Versatile buffer system covering wide pH range for studying pH effects on electrochemical processes | Optimization of analytical methods for compounds with pH-dependent electroactivity |
| Antioxidant Buffer (e.g., DAOB) | Prevents oxidation of analytes susceptible to oxidative degradation during analysis | Stabilization of hydrogen sulfide standards for voltametric measurement [16] |
| Electrochemical Impedance Spectroscopy Setup | Characterizes electrode-solution interface properties and electron transfer kinetics | Method understanding and troubleshooting for complex matrices |
| Design of Experiments Software | Enables efficient optimization of multiple method parameters simultaneously | Systematic method development and robustness testing |
Regulatory Perspectives and Harmonization
Current Regulatory Landscape
The regulatory landscape for pharmaceutical analysis continues to evolve with an increasing emphasis on QbD principles. The Pharmacopeial Discussion Group (PDG), which harmonizes general chapters across the United States Pharmacopeia (USP), European Pharmacopoeia (EP), and Japanese Pharmacopoeia (JP), has made significant progress in harmonizing analytical methods [18]. Recent harmonization activities include methods for Dissolution <711>, Disintegration <701>, Uniformity of Content/Mass <905>, Microbial Enumeration <61>, and Particulate Contamination <788>, among others [18].
This harmonization effort supports the implementation of QbD by providing consistent standards and methods across regulatory jurisdictions. For electrochemical methods, which may not yet be extensively covered in pharmacopeial monographs, the QbD approach provides a solid scientific foundation for demonstrating method validity and reliability to regulatory authorities.
Future Directions
As regulatory agencies increasingly embrace QbD principles, the validation of analytical methods is expected to continue shifting toward the lifecycle approach. The adoption of new technologies, including artificial intelligence and machine learning for method development and optimization, is likely to accelerate this trend [19]. Additionally, the growing acceptance of real-time release testing and continuous manufacturing in the pharmaceutical industry will further drive the need for robust, reliable analytical methods developed and validated using QbD principles.
For electrochemical methods specifically, the demonstrated advantages in sensitivity, speed, and cost-effectiveness position them for increased adoption in pharmaceutical analysis, particularly when coupled with the rigorous, science-based approach offered by QbD.
The application of Quality by Design principles to analytical method validation represents a significant advancement over traditional approaches, offering a systematic, science-based framework for ensuring methods remain fit-for-purpose throughout their lifecycle. For electrochemical methods, which offer distinct advantages in sensitivity and efficiency, the QbD approach provides a pathway to robust validation and regulatory acceptance. As the pharmaceutical industry continues to evolve toward more efficient and quality-focused manufacturing paradigms, the integration of QbD into analytical method development and validation will become increasingly essential for researchers and drug development professionals.
Within pharmaceutical research and development, the validation of analytical methods is not merely a regulatory formality but a fundamental scientific requirement to ensure the reliability, consistency, and accuracy of data. For researchers and scientists working with electrochemical methods, establishing that a procedure is "fit for its intended purpose" is paramount [20]. This process provides documented evidence that the analytical test system is capable of delivering useful and valid analytical data, forming the bedrock upon which critical decisions regarding drug safety, efficacy, and quality are made [21]. The United States Pharmacopeia (USP) and European Pharmacopoeia (EP) provide the essential regulatory frameworks that govern these validation activities, offering strict scientific standards that ensure consistency across pharmaceutical testing practices from drug discovery through to post-marketing surveillance [22].
Adherence to USP and EP standards is a mandatory aspect of the drug development lifecycle. These standards define comprehensive requirements for identity, strength, purity, and consistency of pharmaceutical products, creating a common language for quality that facilitates international collaboration and regulatory harmonization [22]. For electrochemical methods, which are employed in diverse applications from quantifying active ingredients to detecting impurities, validation within these pharmacopeial contexts guarantees that the generated data meets global regulatory expectations. This guide provides a detailed comparison of the four critical validation parameters—Accuracy, Precision, Specificity, and Linearity—within the specific context of electrochemical methods, complete with experimental protocols and data to support robust method development.
Core Validation Parameters: Definitions and Experimental Protocols
The following parameters represent the cornerstone of any analytical method validation. The definitions and experimental approaches outlined below are aligned with general pharmacopeial principles and their specific application to electrochemical techniques.
Accuracy
Accuracy is defined as the degree of agreement between the test result generated by the method and the true value or an accepted reference value [21] [20]. It measures the correctness of your data.
Experimental Protocol for Determining Accuracy: A common approach for determining accuracy in electrochemical methods is through recovery studies using spiked samples [21] [20]. The general procedure is as follows:
- Sample Preparation: Begin with a blank matrix (the sample without the analyte, e.g., a placebo formulation or the supporting electrolyte) that is representative of the actual sample.
- Spiking: Spike the blank matrix with known quantities of the analyte standard. A minimum of three concentration levels (e.g., 80%, 100%, and 120% of the target concentration) should be prepared, with each level analyzed in triplicate.
- Analysis and Calculation: Analyze the spiked samples using the developed electrochemical method. The accuracy is calculated as the percentage recovery of the measured concentration versus the known, spiked concentration:
- % Recovery = (Measured Concentration / Known Concentration) × 100
The acceptable criteria for recovery can vary based on the sample matrix and the analyte level but are often set within 98–102% for the drug substance [20].
Precision
Precision expresses the closeness of agreement (degree of scatter) between a series of measurements obtained from multiple sampling of the same homogeneous sample under the prescribed conditions [21]. It measures the reproducibility of your data. Precision is further subdivided into repeatability (intra-assay precision) and intermediate precision (inter-assay precision, inter-analyst, inter-instrument).
Experimental Protocol for Determining Repeatability:
- Sample Preparation: Prepare a homogeneous sample at 100% of the test concentration.
- Replicate Analysis: Analyze a minimum of five, and preferably six, independent preparations of this sample [20].
- Calculation: Calculate the mean, standard deviation (SD), and relative standard deviation (%RSD or CV) of the results.
- %RSD = (Standard Deviation / Mean) × 100
The Horwitz equation is sometimes used as a guide for expected precision, where the acceptable %RSD for repeatability can be estimated as Horwitz value × 0.67 [21]. For an analyte at 100% concentration, the proposed acceptable %RSD is 1.34%.
Table 1: Example Precision Data from a Cyclic Voltammetry Study of Paracetamol
| Concentration of Standard Solution (μg/ml) | Mean Peak Area (n=3) | Standard Deviation (SD) | %RSD |
|---|---|---|---|
| 0.1956 | 32,952 | 304 | 0.923 |
| 0.9780 | 175,972 | 1265 | 0.719 |
| 1.956 | 318,025 | 2358 | 0.741 |
| 2.934 | 414,746 | 3827 | 0.923 |
This table, adapted from a study on paracetamol, demonstrates excellent repeatability with %RSD values well below 1% across a wide concentration range, which is indicative of a precise analytical method [23].
Specificity
Specificity is the ability of the method to assess unequivocally the analyte in the presence of components that may be expected to be present, such as impurities, degradation products, and matrix components [20]. In electrochemical terms, it ensures that the voltammetric peak or amperometric signal is unequivocally attributable to the target analyte.
Experimental Protocol for Determining Specificity:
- Analysis of Interferents: Individually analyze the blank matrix, the analyte standard, and potential interferents (e.g., degradation products induced by stress conditions like heat or light, known impurities, or excipients).
- Comparison of Signals: Compare the resulting electrochemical signals (e.g., voltammograms). The method is considered specific if the signal from the analyte is resolved and unaffected by the presence of interferents, and no significant signal is observed from the blank or interferents alone at the retention time/peak potential of the analyte.
- For a stability-indicating method, forced degradation studies are performed, and the analyte response should be unaffected by the degradation products and able to be accurately quantified.
Linearity
Linearity of an analytical method is its ability (within a given range) to obtain test results that are directly proportional to the concentration (amount) of analyte in the sample [21] [20].
Experimental Protocol for Determining Linearity:
- Standard Preparation: Prepare a series of standard solutions at a minimum of five, and preferably six, different concentrations. The range should ideally span from 80% to 120% of the expected working range [20].
- Analysis and Plotting: Analyze each standard solution and plot the instrumental response (e.g., peak current in voltammetry) versus the concentration of the analyte.
- Statistical Analysis: Perform a linear regression analysis on the data. The correlation coefficient (r), slope, and y-intercept of the line are reported. The correlation coefficient should generally be greater than or equal to 0.990 [20].
Table 2: Comparison of Key Validation Parameters and Typical Acceptance Criteria
| Parameter | Definition | Typical Experimental Approach | Common Acceptance Criteria (Example) |
|---|---|---|---|
| Accuracy | Closeness to the true value [21] | Recovery study using spiked samples [20] | % Recovery of 98-102% |
| Precision | Closeness of a series of measurements [21] | Multiple injections of a homogeneous sample | %RSD < 2% (for assay) [21] |
| Specificity | Ability to measure analyte unequivocally in the presence of interferents [20] | Analysis of analyte, blank, and potential interferents | No interference observed; analyte peak is resolved. |
| Linearity | Proportionality of response to concentration [20] | Analysis of standards across a concentration range | Correlation coefficient (r) ≥ 0.990 [20] |
Method Comparison and Application in Electrochemical Analysis
Electrochemical methods offer a versatile toolkit for pharmaceutical analysis, but their validation requires careful consideration of the specific technique and application. A comparative study of different electrochemical methods for characterizing microbial fuel cells highlighted the importance of method selection, showing that while techniques like Electrochemical Impedance Spectroscopy (EIS) and Current Interruption (CI) provided highly concordant results for ohmic resistance, real-time optimization methods like Pulse Width Modulation (R-PWM) could achieve power densities up to 95% of the maximum [24]. This underscores that the choice of method impacts not just the validity of the parameters, but also the practical efficiency of the analysis.
Furthermore, the required sensitivity of a method is a critical factor in its selection and validation. A comparison of hydrogen sulfide (H₂S) quantification methods demonstrated a stark contrast in performance between different analytical families. While colorimetric and chromatographic methods quantified H₂S in millimolar and micromolar ranges respectively, electrochemical methods (voltametric and amperometric) successfully quantified H₂S in the nanomole and picomole ranges, while also being less time-consuming [16]. This illustrates that for low-concentration analytes, electrochemical methods can offer distinct advantages in sensitivity and speed, parameters that must be validated through the LOD, LOQ, and precision experiments.
Practical Implementation and Workflow
Successfully implementing a validated electrochemical method requires a structured workflow and an understanding of the necessary reagents and materials. The following diagram and table outline the key components.
Diagram 1: Analytical Method Validation and Implementation Workflow. This chart outlines the key stages from method selection through to ongoing monitoring, highlighting that validation is a cyclical process requiring revalidation when conditions change [20].
Table 3: Essential Research Reagent Solutions for Electrochemical Method Validation
| Reagent / Material | Function in Validation | Example / Notes |
|---|---|---|
| Primary Reference Standard | To establish accuracy and calibration; provides the "true value" [22]. | High-purity analyte from USP/EP or other certified source. |
| Supporting Electrolyte | To provide ionic conductivity and control the electrochemical environment. | Phosphate Buffered Saline (PBS) [16] or LiClO₄ in organic solvents [23]. |
| System Suitability Test Solutions | To verify the system's performance before or during analysis [22]. | A mixture containing the analyte at a known concentration to check precision and resolution. |
| Blank Matrix | To demonstrate specificity and assess potential interference [20]. | Placebo formulation or simulated physiological fluid (e.g., Simulated Tear Fluid) [16]. |
| Standard Solutions for Linearity | To construct the calibration curve and validate the linear range. | A series of solutions at minimum 5 concentrations from 80-120% of target [20]. |
The rigorous validation of electrochemical methods, centered on the core parameters of accuracy, precision, specificity, and linearity, is a non-negotiable standard in pharmaceutical research and development. As demonstrated through comparative studies, the choice of electrochemical technique directly influences the quality and applicability of the generated data. By adhering to the structured experimental protocols and workflows outlined in this guide—which are aligned with USP and EP standards—scientists and researchers can ensure their analytical methods are truly fit for purpose. This diligence provides the foundation for reliable data, which in turn supports the development of safe, effective, and high-quality pharmaceutical products, ultimately fulfilling the regulatory and ethical imperatives of the drug development profession.
For researchers and scientists in drug development, the establishment of robust analytical methods is not merely a regulatory obligation but a scientific imperative that ensures the safety, efficacy, and quality of pharmaceutical products. The lifecycle of an analytical method—encompassing development, validation, and continued verification—forms the backbone of reliable data generation in compliance with global standards such as the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) [25] [26]. Within this framework, electrochemical methods have emerged as powerful analytical techniques offering distinct advantages for specific applications, though they must be objectively evaluated against established alternatives like chromatographic methods.
This guide provides a systematic comparison of analytical techniques, with a focused examination of electrochemical methods relative to chromatographic approaches, underpinned by experimental data and structured within pharmacopeial requirements. The objective is to equip drug development professionals with evidence-based insights for selecting, validating, and implementing analytical methods appropriate for their specific analytical challenges.
The Analytical Method Lifecycle
The journey of an analytical method from conception to routine use is a continuous, interdependent process managed by research and development, quality control, and quality assurance departments [25]. This lifecycle ensures methods remain fit-for-purpose throughout their operational use.
Phase 1: Method Development
Method development begins with a clear definition of analytical requirements. Scientists collect all known information about the analyte's physical and chemical properties and obtain a pure standard for initial studies [25]. A comprehensive literature survey examines existing methodologies for the analyte or structurally similar compounds [25].
Key considerations during development include:
- Analyte Characterization: Understanding stability, solubility, and structural properties.
- Instrument Selection: Choosing techniques compatible with sample stability and analytical requirements.
- Initial Optimization: Systematically changing one parameter at a time while documenting every step.
- Preliminary Figures of Merit: Establishing initial estimates for detection limits, linearity, and precision [25].
The output of this phase is a prototype method ready for formal validation.
Phase 2: Method Validation
Method validation provides documented evidence that the analytical procedure is suitable for its intended purpose. According to USP and EP standards, validation confirms that a method consistently yields results that accurately reflect the quality of the product under assessment [25] [26].
The table below summarizes the core validation parameters and their acceptance criteria as defined by pharmacopeial guidelines [25] [27] [28].
Table 1: Key Analytical Method Validation Parameters and Acceptance Criteria
| Validation Parameter | Definition | Typical Acceptance Criteria |
|---|---|---|
| Specificity | Ability to assess analyte unequivocally in the presence of interferences | Forced degradation & peak purity ≥0.99 [28] |
| Accuracy | Closeness between measured value and true value | Impurities: 80-120% recovery [28] |
| Precision | Degree of agreement among individual test results | Stricter repeatability limits [28] |
| Linearity | Ability to obtain results proportional to analyte concentration | Assay ≥0.9999, Impurities ≥0.9995 [28] |
| Range | Interval between upper and lower analyte concentrations | Demonstrates accuracy, precision, and linearity |
| LOD/LOQ | Limit of Detection/Quantitation | Signal-to-noise ratios of 3:1 for LOD, 10:1 for LOQ |
| Robustness | Capacity to remain unaffected by small, deliberate parameter variations | Now mandatory per ICH Q2(R2) [28] |
Phase 3: Continued Verification
Continued verification, often termed ongoing performance verification, ensures the method remains in a state of control throughout its operational life. This phase includes regular system suitability testing, calibration verification, and periodic revalidation as required by USP <621> [26]. Laboratories must establish rigorous calibration schedules and perform regular equipment inspections to maintain data integrity [26]. Significant changes in conditions or instrumentation necessitate method revalidation to confirm ongoing suitability [25].
Comparative Analysis: Electrochemical vs. Chromatographic Methods
To objectively evaluate analytical performance, we examine experimental data from direct comparisons of electrochemical and chromatographic methods across different applications.
Case Study 1: Quantification of Hydrogen Sulfide (H₂S)
A 2023 study systematically compared four analytical techniques for quantifying H₂S in simulated physiological solutions, with key performance metrics summarized below [16].
Table 2: Performance Comparison for H₂S Quantification in Aqueous Media
| Method | Principle | Quantification Range | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Colorimetric | Mixed diamine reagent forming colored complex | Millimolar (mM) range | Relatively simple and inexpensive | Requires large sample volume (1 ml) and more time |
| Chromatographic (HPLC) | Separation with C-18 column, detection at 670 nm | Micromolar (μM) range | Greater sensitivity than colorimetry; minimal sample (25 μl) | More expensive; requires specialized instrumentation |
| Voltametric | Measurement of electrical signal change | Nanomolar (nM) range | High sensitivity; less time-consuming | Requires electrode preparation and stabilization |
| Amperometric | Current measurement from H₂S oxidation | Picomolar (pM) range | Extremely high sensitivity; rapid response | Requires argon atmosphere; specialized sensor |
The study concluded that electrochemical methods (voltametric and amperometric) offered superior sensitivity, detecting H₂S in nanomolar to picomolar ranges, and were less time-consuming compared to other techniques [16]. The choice of method should be based on the specific project requirements for sensitivity, response time, and cost-effectiveness.
Case Study 2: Detection of Octocrylene in Water Matrices
A 2025 study directly compared electroanalysis using a glassy carbon sensor (GCS) with high-performance liquid chromatography (HPLC) for detecting and quantifying octocrylene (OC), a persistent organic pollutant from sunscreens, in water matrices [17].
Table 3: Method Comparison for Octocrylene Quantification
| Performance Metric | Electroanalysis (GCS) | HPLC |
|---|---|---|
| Limit of Detection (LOD) | 0.11 ± 0.01 mg L⁻¹ | 0.35 ± 0.02 mg L⁻¹ |
| Limit of Quantification (LOQ) | 0.86 ± 0.04 mg L⁻¹ | 2.86 ± 0.12 mg L⁻¹ |
| Sample Preparation | Minimal | More extensive |
| Operational Costs | Lower | Higher |
| Analysis Time | Faster | Slower |
| Instrument Portability | Potential for portable systems | Laboratory-bound |
The research demonstrated that electroanalysis provided lower detection and quantification limits while successfully quantifying OC in real sunscreen samples and various water matrices, proving it a reliable and efficient alternative for environmental monitoring [17].
Case Study 3: Analysis of Natural Food Preservatives
A comprehensive review of methods for determining natural food preservatives (nisin and natamycin) highlighted complementary strengths of electrochemical and chromatographic techniques [29].
Electrochemical techniques offer fast detection time, low cost, high sensitivity, selectivity, and portability, making them suitable for rapid screening and field applications [29]. However, they can suffer from interference in complex food matrices and require regular sensor calibration [29].
Chromatographic techniques remain the most commonly employed methods for determining nisin and natamycin, providing high sensitivity, precision, and robust separation capabilities [29]. Their limitations include expensive instrumentation, need for skilled operators, and more time-consuming procedures [29].
Experimental Protocols for Method Comparison
To ensure reproducible and comparable results when evaluating analytical methods, standardized experimental protocols are essential.
Protocol for Electrochemical Determination of Octocrylene
Materials and Equipment:
- Autolab PGSTAT302N potentiostat/galvanostat (Metrohm)
- Three-electrode cell: Glassy carbon working electrode, Ag/AgCl reference electrode, Platinum counter electrode
- Britton-Robinson (BR) buffer solution (0.04 M, pH 6)
- Octocrylene standard solutions (prepared in ethyl alcohol/water)
Methodology:
- Polish the glassy carbon working electrode before each measurement.
- Prepare 10 mL of BR buffer solution (pH 6) as the electrolyte.
- Set differential pulse voltammetry parameters:
- Initial potential: -0.8 V
- Final potential: -1.5 V
- Step potential: +0.005 V
- Modulation amplitude: +0.1 V
- Modulation time: 0.02 s
- Time interval: 0.5 s
- Equilibrium time: 10 s
- Construct analytical curve by correlating OC concentration with voltammetric current response.
- For real sample analysis, spike water matrices (pool water, distilled water) with sunscreen formulations.
- Periodically renew the sensor surface to ensure selective and sensitive detection [17].
Protocol for Chromatographic Determination of H₂S
Materials and Equipment:
- HPLC system (Ultimate 3000, Thermo) with C18 column (150 mm × 4.6 mm, 5 μm)
- PDA UV-visible detector (670 nm)
- Mobile phase: Acetonitrile and Ammonium formate (15 mM; 70:30 v/v)
- Mixed diamine reagent
Methodology:
- Prepare standard NaSH solutions in simulated tear fluid (STF) or phosphate-buffered saline (PBS).
- Add 100 μl mixed diamine reagent to 5-ml aliquots of standard NaSH solution.
- Shake vigorously and allow to stand for 10 minutes for color development.
- Inject 20-μl aliquot into HPLC system.
- Use isocratic elution with flow rate set at 1.2 ml/min.
- Total run time: 6 minutes (retention time of H₂S: 3.3 minutes) [16].
System Suitability Testing
For both techniques, system suitability testing must be performed as required by USP <621> to verify that the analytical system is operating as intended [26]. This includes evaluating parameters such as precision, resolution, and sensitivity to ensure both the instrument and method are suitable for the intended analysis.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key reagents and materials essential for implementing the experimental protocols described above, with explanations of their functions in analytical method development and execution.
Table 4: Essential Research Reagents and Materials for Analytical Method Development
| Reagent/Material | Function | Application Examples |
|---|---|---|
| USP/EP Reference Standards | Highly purified and characterized substances for method calibration and validation | Drug identity testing, impurity analysis, quality control [26] |
| Glassy Carbon Electrode | Working electrode with low adsorption, high conductivity, and wide potential window | Voltametric detection of organic compounds (e.g., octocrylene) [17] |
| Mixed Diamine Reagent | Forms colored complex with sulfide for detection | Colorimetric and chromatographic H₂S quantification [16] |
| Britton-Robinson Buffer | Versatile buffer system adaptable to various pH values | Electroanalytical measurements requiring controlled pH [17] |
| C18 Chromatographic Column | Reversed-phase stationary phase for compound separation | HPLC analysis of H₂S and other pharmaceutical compounds [16] [17] |
| Electrochemical Cell | Three-electrode setup for controlled potential application | All voltametric and amperometric measurements [16] [17] |
Workflow Visualization
The following diagram illustrates the complete lifecycle of an analytical method, integrating development, validation, and continued verification phases within the regulatory framework of USP and EP standards.
Diagram 1: Analytical Method Lifecycle Workflow
The comparative analysis presented in this guide demonstrates that both electrochemical and chromatographic methods have distinct roles in the analytical scientist's toolkit. Electrochemical methods offer compelling advantages in sensitivity, speed, and cost-effectiveness for specific applications, particularly when measuring analytes in nanomolar to picomolar ranges or when portability is desirable [16] [17]. Chromatographic methods remain the gold standard for many applications due to their robust separation capabilities, established regulatory acceptance, and versatility across diverse analyte types [16] [29].
The choice between these techniques—or their complementary use—must be guided by the specific analytical requirements, sample matrix, regulatory context, and intended use of the data. What remains universal is the necessity of implementing any chosen technique within a rigorous lifecycle framework of development, validation, and continued verification, in compliance with USP, EP, and ICH guidelines [25] [26] [28]. This systematic approach ensures the generation of reliable, defensible data that ultimately protects patient safety and drug product quality throughout the pharmaceutical development process.
Implementing Compliant Electrochemical Methods: From Theory to Practice
Electrochemical methods are indispensable in modern pharmaceutical analysis, offering highly sensitive and selective means to ensure drug quality, safety, and efficacy. This guide provides an objective comparison of three core techniques—voltammetry, amperometry, and potentiometry—framed within the stringent requirements of United States Pharmacopeia (USP) and European Pharmacopoeia (EP) validation standards.
Fundamental Principles and Pharmacopeial Context
Electrochemical techniques measure electrical properties like current, potential, or charge to quantify analytes. The validation of these methods for pharmacopeial monographs demands that they are robust, reproducible, and fit-for-purpose [30] [31]. The drive to replace older methods like thin-layer chromatography (TLC) with more efficient and sensitive techniques, including electroanalysis, is a ongoing priority in international pharmacopoeias [30].
A standard experimental setup involves a three-electrode system: a Working Electrode where the redox reaction occurs, a Reference Electrode that provides a stable potential baseline, and a Counter Electrode to complete the electrical circuit [32]. The choice of technique depends on the analytical question, whether it is identifying and quantifying impurities, assaying active ingredients, or monitoring ions in formulations.
Technique Comparison: Operational Principles and Analytical Performance
The table below summarizes the core characteristics, applications, and key performance metrics of each technique, providing a basis for informed selection.
Table 1: Comparative overview of voltammetry, amperometry, and potentiometry
| Feature | Voltammetry | Amperometry | Potentiometry |
|---|---|---|---|
| Measured Quantity | Current as a function of applied potential [32] | Current at a constant applied potential [32] | Potential (voltage) at zero current [32] |
| Primary Information | Qualitative & Quantitative (redox potential, concentration, reaction kinetics) [32] | Quantitative (concentration of electroactive species) [32] | Quantitative (ion activity or concentration) [32] |
| Key USP/EP Applications | Drug purity, impurity profiling, dissolution testing, active ingredient quantification [31] [33] | Flow-injection analysis, detector for chromatography, biosensors (e.g., glucose) [34] [32] | pH measurement, ion analysis (Na+, K+, Ca2+), potentiometric titrations [35] [32] |
| Typical Detection Limits | Nanomolar to picomolar range (e.g., Lidocaine LOD: 0.29 µmol L⁻¹) [33] | Low micromolar to nanomolar range [34] | Micromolar range (ion-selective electrodes) [32] |
| Advantages | High sensitivity and selectivity; rich mechanistic information; various modes for different needs [31] [32] | Simple instrumentation; fast response; well-suited for continuous flow and detection systems [34] [32] | Simple, low-cost instrumentation; non-destructive; direct measurement of ionic activity [32] |
| Disadvantages | Can be more complex; electrode fouling can be an issue [31] | Less selective than voltammetry; requires a constant potential [32] | Can be affected by sample matrix; requires stable reference electrode [34] |
Quantitative data further highlights their performance differences. A study comparing solid-contact ion-selective electrodes (SC-ISEs) in potentiometric and amperometric (flow injection) modes demonstrated distinct advantages for amperometry, including significantly lower detection limits and faster response times [34].
Table 2: Experimental performance data for solid-contact ion-selective electrodes (SC-ISEs)
| Performance Metric | Potentiometric Mode | Amperometric Mode (Flow Injection) |
|---|---|---|
| Response Time | Slower | 15–30 seconds [34] |
| Detection Limit | Higher | Two orders of magnitude lower [34] |
| Signal-to-Noise Ratio | Standard | 5-6 fold increased [34] |
| Key Advantage | Direct ion activity measurement | Eliminates background drift and potential instability of SC-ISEs [34] |
Experimental Protocols and Methodologies
Detailed methodologies are critical for method validation and transfer between laboratories, as required by pharmacopeias [30].
Square-Wave Voltammetry (SWV) for Drug Compound Assay
This protocol outlines the determination of Lidocaine Hydrochloride (LH) using Square-Wave Voltammetry at a carbon-paste electrode (CPE), a method suitable for pharmaceutical preparations [33].
- Reagents and Solutions: Lidocaine Hydrochloride standard, potassium nitrate (KNO₃) as supporting electrolyte, and paraffin oil for electrode preparation. Use distilled/deionized water [33].
- Electrode System: Working Electrode: Bare carbon-paste electrode (70% graphite powder, 30% paraffin oil). Reference Electrode: Ag/AgCl (3M KCl). Counter Electrode: Platinum wire [33].
- Procedure:
- Electrode Preparation: Thoroughly mix graphite powder and paraffin oil. Pack the paste into a syringe body and insert a copper wire for electrical contact. Smooth the surface on weighing paper [33].
- Sample Preparation: Transfer an aliquot of the standard or sample LH solution into a 25 mL volumetric flask. Add 4 mL of 1 M KNO₃ solution and dilute to the mark with water [33].
- Accumulation: Pour the solution into the electrochemical cell. Set the system to open-circuit and allow an accumulation time of 120 seconds [33].
- Voltammetric Recording: Using the SWV technique, scan the potential from +0.5 V to +1.2 V. Apply a scan rate of 0.125 V/s. After each measurement, regenerate the CPE surface by polishing on weighing paper [33].
- Data Analysis: The oxidation peak of LH appears between +0.80 and +0.88 V. Plot the peak current against concentration to generate a calibration curve [33].
Flow Injection Amperometry for Ion Analysis
This protocol describes using amperometry with solid-contact ion-selective electrodes (SC-ISEs) in a flow system, which offers superior stability over traditional potentiometry [34].
- Reagents and Solutions: Standard solutions of the target ion (e.g., Na⁺, K⁺). A background electrolyte solution compatible with the flow system. The ion-selective membrane components (e.g., PVC, plasticizer, ionophore) [34].
- Electrode and System Setup: Working Electrode: Glassy carbon electrode coated with a conducting polymer (e.g., PEDOT) and the appropriate ion-selective membrane. Reference Electrode and Counter Electrode as per system requirements. Flow System: A flow injection analysis (FIA) apparatus with a pump and injection valve [34].
- Procedure:
- System Conditioning: Pass the background electrolyte carrier stream through the system at a constant flow rate until a stable baseline current is established [34].
- Sample Injection: Inject a defined volume of the standard or sample solution into the carrier stream [36].
- Amperometric Detection: Apply a constant potential to the SC-ISE and record the transient current peak as the sample plug passes over the electrode [34].
- Signal Measurement: The height or area of the current peak is proportional to the concentration of the target ion [34].
Visualizing Method Selection and Workflow
The following diagrams illustrate the logical process for selecting an electrochemical technique and a generalized workflow for voltammetric drug analysis.
Figure 1: Electrochemical technique selection logic
Figure 2: Voltammetric analysis workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key materials and reagents required for executing the electrochemical experiments described in this guide.
Table 3: Essential research reagents and materials for electrochemical analysis
| Item | Function / Application | Example / Specification |
|---|---|---|
| Three-Electrode Potentiostat | The core instrument for applying potentials and measuring currents in voltammetry and amperometry [32]. | Computer-controlled system with data acquisition software. |
| Working Electrodes | Surface where the redox reaction of the analyte occurs; material choice is critical [36] [33]. | Glassy Carbon Electrode (GCE), Carbon-Paste Electrode (CPE), Gold, Platinum, Mercury-based electrodes [36] [33]. |
| Reference Electrodes | Provides a stable, known potential for the working electrode to be measured against [32]. | Ag/AgCl (3M KCl) or Saturated Calomel Electrode (SCE) [33] [32]. |
| Counter Electrodes | Completes the electrical circuit in the electrochemical cell [32]. | Platinum wire or coil [33]. |
| Supporting Electrolyte | Carries current and minimizes resistive loss (iR drop) in solution; essential for clear voltammograms [33]. | Inert salts (e.g., KNO₃, KCl, phosphate buffers) at 0.1 - 1.0 M concentration [33]. |
| Ion-Selective Membrane Components | Used to fabricate sensors for potentiometry and amperometry targeting specific ions [34]. | Poly(vinyl chloride) PVC, plasticizers (e.g., o-NPOE), ionophores, ion-exchangers [34]. |
| Standard Analytical Reagents | Used for calibration curves and method validation [33]. | High-purity drug standards (e.g., Lidocaine HCl) and ionic standards (e.g., NaCl, KCl) [34] [33]. |
The simultaneous determination of epinephrine (EP) and uric acid (UA) presents a significant analytical challenge in pharmaceutical and clinical chemistry due to their similar electrochemical oxidation potentials at conventional electrodes. This case study examines the development, validation, and optimization of a Square-Wave Adsorptive Stripping Voltammetry (SWAdSV) method using a poly(L-cysteine) modified screen-printed carbon electrode (pLC-SPCE) for the simultaneous determination of EP and UA in real samples [37]. This methodology represents a significant advancement in electrochemical analysis, offering a validated approach that aligns with pharmacopeial standards for method validation as outlined in USP and EP guidelines [30].
The pLC-SPCE sensor was specifically designed to overcome the limitations of bare electrodes, which typically exhibit poor peak separation for EP and UA. The modification process involved electrophoretic polymerization of L-cysteine onto the carbon electrode surface, creating a functionalized interface that enhances both selectivity and sensitivity [37]. This sensor development is particularly relevant within the framework of pharmacopeial method validation, which emphasizes the need for robust, specific, and reproducible analytical methods to ensure drug quality and safety [30].
Experimental Protocols and Methodologies
Sensor Fabrication and Modification
The pLC-SPCE sensor was fabricated through a meticulous electrode modification process [37]:
- Electropolymerization: L-cysteine was deposited onto the screen-printed carbon electrode surface through electropolymerization within a specific potential window.
- Optimization: Critical parameters including L-cysteine concentration, number of polymerization cycles, and deposition potential were systematically optimized to achieve maximum sensor performance.
- Characterization: Successful deposition of the poly(L-cysteine) film was confirmed using Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS), providing surface analysis and confirmation of the modified layer [37].
The modification process significantly enhanced the electrode properties, resulting in improved peak-to-peak separation for EP and UA compared to bare SPCE, which is essential for reliable simultaneous determination [37].
SWAdSV Analytical Procedure
The optimized SWAdSV method incorporated the following experimental sequence [37]:
- Accumulation Step: Analytes were preconcentrated on the pLC-SPCE surface at an optimized deposition potential and time under stirring conditions.
- Equilibration Period: A brief quiet time (5-10 seconds) allowed for stabilization of the solution prior to measurement.
- Voltammetric Scan: Square-wave voltammetry was performed with carefully optimized parameters including amplitude, frequency, and potential step.
- Electrode Cleaning: A cleaning step between measurements ensured surface regeneration and reproducibility.
All measurements were conducted in 0.1 M phosphate buffer solution (PBS) at optimized pH, with the entire analysis performed at room temperature (23 ± 2°C) [37] [38].
Method Optimization Parameters
Comprehensive optimization of the SWAdSV method was conducted to enhance analytical performance [37]:
Table 1: Optimized Parameters for SWAdSV Method using pLC-SPCE
| Parameter Category | Specific Parameters | Optimized Conditions |
|---|---|---|
| Electrode Modification | L-cysteine concentration | Specifically optimized |
| Electropolymerization cycles | Specifically optimized | |
| SWV Parameters | Amplitude | 25 mV |
| Frequency | 15 Hz | |
| Potential step | 5 mV | |
| Accumulation Conditions | Deposition potential | Optimized for analytes |
| Deposition time | 60-120 seconds | |
| Solution Conditions | Supporting electrolyte | 0.1 M PBS (pH 7.4) |
| pH | Optimized for separation |
Performance Comparison with Alternative Methods
The validation data for the SWAdSV method using pLC-SPCE demonstrates significant advantages over other electrochemical and conventional techniques for simultaneous determination of EP and UA.
Analytical Figures of Merit
Table 2: Performance Comparison of Analytical Methods for EP and UA Determination
| Method | Sensor/Technique | Linear Range (EP/UA) | LOD (EP/UA) | Real Sample Application |
|---|---|---|---|---|
| SWAdSV | pLC-SPCE [37] | 49.0-887.1 μg/L (both) | 10.0 μg/L (both) | Pharmaceutical, urine |
| SWV | Bare SPCE (single-drop) [38] | Not specified | Not specified | Pharmaceutical, urine |
| SWAdSV | Cytosine/PGE (dopamine) [39] | 0.1 mM-0.5 μM / 0.1 μM-7.5 nM | 2.28 nM | Human plasma serum |
| HPLC-MS/MS | Dried blood spots [40] | Not specified | Not specified | Dried blood spots |
Selectivity and Interference Studies
The pLC-SPCE sensor demonstrated excellent selectivity in the presence of potentially interfering compounds [37]:
- No significant interference was observed from glucose, L-ascorbic acid, urea, and various ions (K+, Cl-, Ca2+, SO42-, Mg2+, NH4+, C2O42-).
- The modified electrode surface effectively minimized fouling effects, maintaining stability over multiple measurements.
- The method successfully resolved the oxidation peaks of EP and UA with sufficient peak-to-peak separation for reliable quantification in complex matrices.
Validation According to Pharmacopeial Standards
The SWAdSV method was rigorously validated following principles aligned with USP and EP guidelines for analytical method validation [30], addressing key parameters:
Accuracy and Precision
- Accuracy: Average recoveries ranged from 94.4% to 108.4% (n=6) across four concentration levels for both analytes [37].
- Precision: Relative standard deviations (RSD) ranged from 2.6% to 11.7% (n=6), demonstrating acceptable method precision [37].
- The recovery studies were conducted in both pharmaceutical formulations (EP auto-injector) and human urine samples, confirming method accuracy in different matrices.
Sensitivity and Linearity
- The method demonstrated exceptional sensitivity with LOD of 10.0 μg/L and LOQ of 19.8 μg/L for both EP and UA [37].
- Two linear concentration ranges were established: 49.0-326.1 μg/L and 326.1-887.1 μg/L for both analytes, with correlation coefficients exceeding 0.99 [37].
- Weighted linear regression models were employed to address heteroscedasticity, improving accuracy at lower concentration levels [38].
Robustness and Practical Application
- The method maintained performance under slight variations of operational parameters, confirming robustness.
- Successful application to real-world samples included analysis of EP in auto-injector formulations and UA in human urine, with accuracy comparable to established techniques [37].
- The single-drop analysis capability (50 μL volume) demonstrated particular advantage for samples with limited availability [38].
Signaling Pathways and Experimental Workflow
The experimental workflow for method development and validation encompasses multiple interconnected phases, from sensor design to practical application.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents and Materials for SWAdSV Method Development
| Reagent/Material | Function/Purpose | Application Example |
|---|---|---|
| L-cysteine | Electrode modifier for selectivity enhancement | pLC-SPCE sensor fabrication [37] |
| Screen-printed carbon electrodes | Disposable, reproducible electrode platforms | Single-drop analysis (50 μL) [38] |
| Phosphate buffer solution (PBS) | Supporting electrolyte, pH control | Optimal peak separation at pH 7.4 [37] |
| Stable isotope internal standards | Quantification accuracy in complex matrices | HPLC-MS/MS reference methods [40] |
| Cytosine-modified electrodes | Alternative modification for neurotransmitter detection | Dopamine determination in biological samples [39] |
The SWAdSV method using pLC-SPCE represents a significant advancement in electrochemical analysis for simultaneous determination of epinephrine and uric acid. The validated method demonstrates excellent sensitivity, selectivity, and reliability that meets pharmacopeial standards for analytical method validation [30]. With LOD of 10.0 μg/L for both analytes, wide linear range, and successful application to pharmaceutical and biological samples, this approach offers a robust alternative to conventional chromatographic methods [37]. The methodology aligns with current trends in analytical chemistry toward miniaturization, portability, and cost-effectiveness while maintaining rigorous performance standards required for pharmaceutical analysis and clinical diagnostics [38]. The incorporation of advanced data processing techniques, including weighted linear regression to address heteroscedasticity, further enhances the method's reliability for quantitative analysis at low concentration levels [38].
High-Performance Liquid Chromatography with Electrochemical Detection (HPLC-EC or HPLC-ECD) represents a cornerstone technique in neuroscience and pharmaceutical research for quantifying monoamine neurotransmitters and their metabolites. This method synergizes the superior separation capabilities of liquid chromatography with the exceptional sensitivity of electrochemical detection for analyzing electroactive compounds. HPLC-ECD enables precise measurement of key neurotransmitters such as dopamine (DA), serotonin (5-HT), and norepinephrine (NE), along with metabolites including 5-hydroxyindoleacetic acid (5-HIAA), 3,4-dihydroxyphenylacetic acid (DOPAC), and homovanillic acid (HVA) at concentrations as low as the nano-, pico-, and even femtomolar level [41] [42]. This sensitivity is paramount for studying the intricate dynamics of neurochemical transmission in both healthy and impaired nervous systems and for diagnosing neurotransmitter-related disorders [41] [43].
The technique is particularly vital for analyzing complex biological matrices like cerebrospinal fluid (CSF), brain tissue homogenates, and microdialysates, where analytes are present in trace amounts amidst a multitude of interfering substances [41] [42]. This case study will objectively compare HPLC-EC's performance against alternative techniques, detail experimental protocols, and frame the discussion within the context of validation as per United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards [44] [45].
Analytical Technique Comparison: HPLC-ECD vs. LC-MS/MS
While several analytical techniques are available for neurotransmitter analysis, HPLC-ECD and Liquid Chromatography with Tandem Mass Spectrometry (LC-MS/MS) are among the most prominent. The table below provides a structured, quantitative comparison of these two core techniques.
Table 1: Performance and Practical Comparison of HPLC-ECD and LC-MS/MS
| Aspect | HPLC-ECD | LC-MS/MS |
|---|---|---|
| Detection Principle | Measurement of current from oxidation/reduction of electroactive compounds [41] [46] | Mass-to-charge (m/z) ratio measurement [46] |
| Best For | Targeted analysis of electroactive monoamines and metabolites [41] [46] | Broad, exploratory analysis, non-electroactive compounds, isomers [46] |
| Sensitivity | High (pM / pg/µL range) [46] | Very High (sub-pg/µL) [46] |
| Sample Preparation | Simple (often only filtration; derivatization for some analytes) [42] [46] | Complex (protein precipitation, solid-phase extraction, derivatization) [46] |
| Typical Run Time | 5–30 minutes [46] | 15–45 minutes [46] |
| Instrument Cost | ~$45k–$80k [46] | ~$250k–$450k [46] |
| Cost per Sample | ~$2–$5 [46] | ~$10–$30 [46] |
| Ease of Use | User-friendly, simpler maintenance [46] | Requires significant technical expertise [46] |
HPLC-ECD is highly specific for electroactive compounds, making it excellent for routine, high-throughput monitoring of known monoamine targets. Its advantages include lower operational costs, rapid analysis times, and robust performance with minimal downtime [41] [46]. A key limitation is that it is generally unsuitable for non-electroactive compounds unless a derivatization step is incorporated [41] [46].
LC-MS/MS is recognized for its superior sensitivity and selectivity, often considered a gold standard for its ability to analyze a vast panel of analytes simultaneously, including non-electroactive substances and neuropeptides [41] [46]. It also provides structural confirmation for identifying unknown compounds. However, these benefits come with significantly higher costs, both for initial investment and per sample, and require more complex operation and maintenance [46].
Experimental Protocol for HPLC-EC Analysis of CSF
The following detailed methodology, adapted from a clinical study, outlines the quantification of serotonin and dopamine metabolites in human cerebrospinal fluid [43].
Materials and Reagents
Table 2: Essential Research Reagents and Materials
| Item | Function / Specification | Source Example |
|---|---|---|
| HPLC System | Pump, autosampler, column oven | Waters |
| Electrochemical Detector | Amperometric or coulometric detection | Waters 2465 |
| Analytical Column | Reverse-phase C18 (e.g., Symmetry C18, 4.6 × 250 mm, 5 µm) | Waters |
| Mobile Phase | 50 mM potassium dihydrogen phosphate, 0.99 mM sodium 1-octanesulfonate, 0.053 mM EDTA, 12% methanol, pH adjusted to 2.5 | Prepared in-lab [43] |
| Reference Standards | 5-HIAA, HVA, 3-OMD | SantaCruz Biotech |
| CSF Matrix Control | For preparing quality control samples | Randox |
Sample Collection and Preparation
- Collection: CSF samples (1–1.5 ml) are collected, snap-cooled, and immediately stored at -80 °C until analysis to preserve analyte integrity [43].
- Preparation: Upon analysis, samples are thawed and centrifuged at 3000 rpm for 10 minutes. The supernatant is transferred to an autosampler vial for injection. CSF typically requires minimal preparation, eliminating the need for protein precipitation or extraction [43].
Chromatographic and Detection Conditions
- Column: Reverse-phase C18 column maintained at 35 °C [43].
- Mobile Phase: Isocratic elution with a mixture of 50 mM potassium dihydrogen phosphate, 0.99 mM sodium 1-octanesulfonate (an ion-pairing agent), 0.053 mM EDTA (a chelating agent), and 12% methanol. The pH is critically adjusted to 2.5 using 85% O-phosphoric acid [43].
- Flow Rate: 1.3 mL/min [43].
- Injection Volume: 20 µL [43].
- Detection: Electrochemical detection using a glassy carbon (GC) working electrode and an in-situ Ag/AgCl (ISSAC) reference electrode. The potential is set at +0.80 V, with a range of 10 nA [43].
Validation Parameters per USP/EP Guidelines
To ensure reliability and regulatory compliance, the method is validated against key parameters [43] [45]:
- Linearity: Verified for 5-HIAA (65.35–2615.0 nmol/L), HVA (68.62–2745.0 nmol/L), and 3-OMD (236.5–4730.0 nmol/L) with a correlation coefficient (R²) > 0.98 [43].
- Precision and Accuracy: Inter-day and intra-day precision (Relative Standard Deviation, RSD) should be < 20%, and accuracy (mean relative error) within ±20% [43].
- Specificity: The method must resolve analytes from other components in the CSF matrix [43].
- Limit of Quantification (LOQ): The lowest standard with precision and accuracy within ±20% [43].
- System Suitability Testing (SST): Performed before and/or during analysis to ensure the chromatographic system is performing adequately, as mandated by USP <621> and EP chapters [44] [45].
Signaling Pathways and Metabolites
Understanding the metabolic pathways of neurotransmitters is crucial for interpreting analytical results. The following diagrams illustrate the biosynthesis and metabolism of the serotonergic and dopaminergic systems.
Application Workflow and Regulatory Framework
A typical workflow for a validated HPLC-EC study, from sample collection to data reporting, must operate within a stringent quality and regulatory framework.
Adherence to USP and EP Standards
The entire analytical process is governed by pharmacopeial standards to ensure data integrity and reliability [45]:
- Analytical Instrument Qualification (AIQ): USP <1058> requires that instruments like HPLC systems undergo rigorous qualification (Design, Installation, Operational, and Performance Qualification) to prove they are suitable for their intended use [45] [47]. Performance Qualification (PQ) can be efficiently integrated with System Suitability Testing (SST) from daily runs for continuous performance verification [47].
- System Suitability Testing (SST): USP <621> and corresponding EP chapters define SST parameters (e.g., signal-to-noise, peak symmetry) to confirm that the analytical system is under control at the time of testing [44].
- Method Validation: USP <1225> and ICH Q2(R1) provide guidelines for validating the analytical procedure itself, establishing documented evidence that the method is fit for purpose [45].
HPLC-EC remains a powerful, cost-effective, and robust solution for the targeted analysis of electroactive neurotransmitters in biological samples. Its strengths in sensitivity, speed, and operational economy make it ideal for high-throughput laboratories focused on monoamine research, such as longitudinal microdialysis studies or clinical diagnostics of neurotransmitter disorders [43] [46].
The choice between HPLC-EC and LC-MS/MS should be guided by specific research needs. For focused, high-volume analysis of classic monoamines, HPLC-EC is often the superior choice. For discovery-phase work requiring broad panels, analysis of non-electroactive compounds, or structural confirmation, LC-MS/MS is more appropriate [46]. A hybrid approach, using HPLC-EC for routine analysis and LC-MS/MS for validation or expanded profiling, is a powerful strategy employed in many modern neuroscience labs [46].
Ultimately, the credibility of data generated by either technique hinges on a foundation of rigorous method validation and ongoing instrument qualification as prescribed by USP and EP standards, ensuring that results are reliable, reproducible, and scientifically defensible [45] [47].
Instrument Qualification and Calibration as per USP <1058>
In the pharmaceutical industry, the validation of analytical methods per USP and EP pharmacopeia standards is a cornerstone of ensuring drug safety and efficacy. The lifecycle of an analytical instrument is integral to this process, providing the foundation for reliable and reproducible data. United States Pharmacopeia (USP) General Chapter <1058> provides the definitive framework for Analytical Instrument Qualification (AIQ), a critical compliance requirement for every drug development laboratory. A profound understanding of USP <1058> is non-negotiable for researchers and scientists tasked with method validation, as it directly impacts the integrity of all subsequent analytical results.
Staying current with regulatory guidance is a dynamic challenge. As of 2025, a significant proposed update to USP <1058> is under review, which aims to restructure the existing guidance and refine the scientific, risk-based approach to the Analytical Instrument and System Qualification (AISQ) journey [48]. This article provides a comparative guide to the established and proposed qualification models, equipping professionals with the knowledge to navigate this transition.
Traditional vs. Proposed Lifecycle Models: A Comparative Analysis
The familiar "4Qs" model has long been the bedrock of instrument qualification. The proposed update to USP <1058> seeks not to abolish this model but to integrate it into a more holistic, three-stage lifecycle that aligns with contemporary process validation and analytical procedure lifecycle concepts [48].
The Established 4Qs Model
The traditional model subdivides qualification into four distinct, sequential stages:
- Design Qualification (DQ): Documented verification that the instrument's design conforms to user requirements and intended purpose.
- Installation Qualification (IQ): Documented verification that the instrument is delivered and installed correctly according to specified requirements.
- Operational Qualification (OQ): Documented verification that the instrument operates according to its specifications across its intended operating ranges.
- Performance Qualification (PQ): Documented verification that the instrument consistently performs according to specifications for its intended application.
The Proposed Three-Stage Lifecycle Model
The proposed model reframes qualification into three overarching stages [48]:
- Stage 1: Specification and Selection: This stage encompasses the strategic planning and definition of requirements, effectively integrating the principles of DQ.
- Stage 2: Installation, Performance Qualification, and Validation: This stage consolidates the hands-on verification activities, combining IQ, OQ, and initial PQ.
- Stage 3: Ongoing Performance Verification: This stage emphasizes that qualification is not a one-time event, focusing on continuous monitoring and re-qualification throughout the instrument's operational life.
Table 1: Comparison of Traditional and Proposed USP <1058> Qualification Models
| Aspect | Traditional 4Qs Model | Proposed Three-Stage Lifecycle Model |
|---|---|---|
| Core Structure | Four sequential qualifications (DQ, IQ, OQ, PQ) [48] | Three integrated stages (Specification, Installation/PQ, Ongoing Verification) [48] |
| Governance Philosophy | Event-based qualification | Continuous assurance journey over the entire operational lifecycle [48] |
| Primary Focus | Instrument-centric verification | System-level fitness for intended use and data integrity |
| Alignment with Other Standards | Standalone process | Integrated with analytical procedure lifecycle and process validation [48] |
| Change Management | Implied between stages | Explicitly called out as "Change Control over the Life Cycle" [48] |
| Formal Status | Currently official (as of Aug 2017) [48] | Proposed revision; comment period open until May 31, 2025 [48] |
The following workflow diagram illustrates the logical progression of the proposed three-stage lifecycle and its relationship to the traditional 4Qs, highlighting the continuous nature of the process.
Experimental Protocols for Instrument Qualification
A robust qualification protocol is characterized by its objectivity, reproducibility, and comprehensive documentation. The following methodologies detail the core experiments for the key verification stages.
Operational Qualification (OQ) Protocol for a High-Performance Liquid Chromatography (HPLC) System
OQ verifies that the instrument operates according to specifications across its entire operating range.
- 1. Objective: To verify the operational performance of the HPLC system's pump, autosampler, column oven, and detector against predefined specifications.
- 2. Materials and Reagents:
- HPLC system with DAD or UV-Vis detector
- Certified reference standard (e.g., caffeine, uracil)
- Mobile phase: HPLC-grade water and acetonitrile
- Calibrated thermometer and flow meter
- 3. Methodology:
- Pump Flow Rate Accuracy and Precision: Set the pump at various flow rates (e.g., 0.5, 1.0, 2.0 mL/min). Use a calibrated flow meter or gravimetrically measure the eluent output over a set time. Calculate accuracy (% of set point) and precision (%RSD).
- Pump Composition Accuracy: Perform a step-gradient (e.g., 10% to 90% organic phase) while monitoring the detector baseline. The observed steps should correlate with the programmed composition.
- Autosampler Temperature Accuracy: Place a temperature probe in the sample vial compartment and compare the reading to the set temperature.
- Autosampler Precision (Injection Volume Carryover): Make six consecutive injections of a high-concentration standard followed by a blank. Calculate the %RSD of the peak areas for precision and the percentage carryover in the blank injection.
- Column Oven Temperature Accuracy: Place a calibrated thermometer inside the column compartment and compare the reading to the set temperature.
- Detector Wavelength Accuracy: Inject a standard with known spectral maxima (e.g., caffeine). The observed wavelength maximum should be within ± a specified tolerance (e.g., ±2 nm) of the certified value.
- Detector Linearity and Stray Light: Inject a series of standards of increasing concentration and plot response versus concentration. Calculate the correlation coefficient (R²) and evaluate for deviations from linearity.
- 4. Acceptance Criteria: All measured parameters must fall within the manufacturer's specifications or tighter, internally defined limits.
Performance Qualification (PQ) / Ongoing Performance Verification Protocol
PQ demonstrates that the system performs consistently for its intended application under actual conditions of use.
- 1. Objective: To verify that the integrated instrument system is suitable for its intended analytical application on a day-to-day basis and over time.
- 2. Materials and Reagents:
- A system suitability test (SST) mixture specific to the application (e.g., a drug and its related compounds).
- Established chromatographic method.
- 3. Methodology: Perform the analysis of the SST mixture according to the validated method. Key parameters are measured and tracked.
- 4. Acceptance Criteria: System suitability parameters as defined in the analytical method must be met. The results should be documented and tracked via control charts to monitor long-term performance.
Table 2: Key System Suitability Tests for HPLC Performance Qualification
| Test Parameter | Experimental Measurement | Typical Acceptance Criteria | Function in Data Integrity |
|---|---|---|---|
| Retention Time | Time from injection to peak apex | %RSD < 1.0% for replicate injections | Verifies system stability and method reproducibility |
| Peak Area | Integrated area under the chromatographic peak | %RSD < 2.0% for replicate injections | Confirms injection precision and detector stability |
| Theoretical Plates | Calculation from peak width and retention time (N > 2000) | Meets or exceeds method specification | Assesses column performance and separation efficiency |
| Tailing Factor | Measurement of peak symmetry (T ≤ 2.0) | Meets or exceeds method specification | Indicates appropriate column-peak interaction |
| Resolution | Separation between two adjacent peaks (Rs > 1.5) | Meets or exceeds method specification | Demonstrates the method's ability to separate critical pairs |
The Scientist's Toolkit: Essential Research Reagent Solutions
The following reagents and materials are fundamental for executing the qualification and calibration protocols for electrochemical and chromatographic instruments.
Table 3: Essential Reagents and Materials for Instrument Qualification
| Item | Function & Application | Critical Specification Notes |
|---|---|---|
| Certified Reference Standards | Calibration of detector response (wavelength, linearity); used in OQ/PQ. | Must be traceable to a national metrology institute (e.g., NIST). Purity and stability are critical. |
| HPLC-Grade Solvents | Mobile phase preparation for chromatographic systems. | Low UV absorbance, high purity, minimal particulates to prevent system damage and baseline noise. |
| System Suitability Test (SST) Mixtures | Verification of integrated system performance for PQ/Ongoing Verification. | A mixture of analytes that challenge the method's critical parameters (resolution, peak shape, etc.). |
| Buffer Salts & Reagents | Mobile phase preparation for methods requiring pH control. | HPLC-grade purity, prepared with ultra-pure water, and filtered through 0.45µm or 0.22µm membranes. |
| Qualification Kits | Manufacturer-provided tools for specific OQ tests (e.g., flow meters, thermometers). | Often include certified hardware and software for automated execution and data collection. |
The proposed update to USP <1058> marks a significant evolution from a static, instrument-centric checklist to a dynamic, system-oriented lifecycle assurance journey [48]. For researchers and drug development professionals, this shift underscores the importance of continuous data monitoring and robust change control management over the entire operational life of an instrument. While the traditional 4Qs model provides a familiar structure, its integration into the broader three-stage model promises greater alignment with modern quality-by-design principles and analytical procedure lifecycles. As the comment period for this proposal remains open until May 31, 2025, the scientific community has a critical window to engage with and shape the future of analytical instrument qualification, ensuring it remains a robust foundation for pharmaceutical research and public health.
The development of a pharmaceutical product is a complex journey from synthesizing a new drug substance to delivering a safe and effective formulated product to patients. Throughout this lifecycle, robust analytical methods are critical for ensuring identity, strength, quality, purity, and potency. The validation of these methods, particularly modern techniques like electrochemical methods, against globally recognized pharmacopeial standards such as the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) is not merely a regulatory formality but a fundamental scientific requirement [49]. This guide provides a comparative analysis of product performance and methodological applications across the key stages of drug development: drug substance, formulated product, and biomarker analysis, providing a framework for scientists to make informed decisions based on experimental data and regulatory guidelines.
Analytical Method Validation per USP/EP
Core Principles and Regulatory Significance
Adherence to USP and EP standards provides a consistent, globally recognized framework for quality. These standards are integral to every stage of drug development, from discovery and preclinical research to regulatory approval and post-marketing surveillance [49]. Compliance ensures improved research and development efficiency, robust data integrity, and facilitates international collaboration and regulatory harmonization [49]. The validation process for any analytical method, including electrochemical methods, must assess critical performance parameters as outlined in general chapters like USP <1225> to ensure the method is fit for its intended purpose [49].
Essential Research Reagent Solutions
The following toolkit details reagents and materials essential for conducting validated analyses, particularly in a pharmacopeial context.
Table: Research Reagent Toolkit for Pharmacopeial Analysis
| Reagent/Material | Primary Function in Analysis |
|---|---|
| USP/EP Reference Standards | Highly characterized specimens used for instrument calibration, method qualification, and determining accuracy, specificity, and linearity [3] [49]. |
| Compendial Reagents | Reagents specified in USP-NF or EP monographs, ensuring consistency and reproducibility of official analytical procedures [3]. |
| Impurity Standards | Critical for assessing drug substance and product purity; used for identification and quantification of potentially harmful impurities [49]. |
| Performance Verification Standards | Used to qualify analytical instruments and ensure systems are operating within specified parameters before analysis [3] [49]. |
Drug Substance Analysis
Performance Comparison: Batch vs. Continuous Manufacturing
The shift from traditional batch processing to continuous manufacturing for drug substances is driven by potential gains in quality, safety, and efficiency [50]. Modeling and simulation (M&S) tools are vital for informing this decision, allowing for the comparison of process performance before significant capital investment.
Table: Performance Comparison of Batch vs. Continuous Drug Substance Manufacturing
| Performance Metric | Batch Process | Continuous Process | Experimental/Modeling Data |
|---|---|---|---|
| Process Footprint | Large (e.g., 2000-gallon reactor) | Small (e.g., 2.5 L tubular reactor) | Mechanistic model for a reduction reaction [50]. |
| Cycle Time | Long (e.g., ~19 hours for addition steps) | Short (e.g., 1 min reactor residence time) | Model based on heat transfer and kinetics [50]. |
| Heat Transfer Control | Challenging at large scale; slow addition to manage exothermicity | Excellent due to high surface-area-to-volume ratio | Data from calorimetric studies and safety assessments [50]. |
| Facility Occupancy | High (Large reactor halls) | Low (e.g., ~2 fume hoods for a reaction/workup train) | Equipment sizing and facility assessment [51]. |
Experimental Protocol: Informing Process Decisions with M&S
Objective: To evaluate the feasibility of converting a reduction reaction and workup from semi-batch to continuous mode using a mechanistic model. Methodology:
- Data Collection: Gather kinetic and calorimetric data (heat of reaction, heat of quench) and partition coefficients for extraction steps [50].
- Model Development: Construct a mechanistic model based on mass and energy balances. For the semi-batch process, model the slow addition of reagents to control temperature [50].
- Simulation of Alternatives:
- Hybrid Process: Model the reaction in a plug flow reactor (PFR) while retaining semi-batch workup.
- Fully Continuous Process: Model the reaction in a PFR and the workup in a series of continuous stirred-tank reactors (CSTRs) and mixer-settlers [50].
- Performance Comparison: Use the model to predict and compare key performance indicators (KPIs) such as cycle time, equipment size, and facility occupancy between the different process configurations [50].
Formulated Product Analysis
Performance Comparison: In Vivo vs. In Vitro Bioequivalence Methods
For formulated products, demonstrating bioequivalence (BE) is critical. While human pharmacokinetic studies are often used, in vitro methods can be superior in specific contexts, offering ethical and economic advantages [51].
Table: Performance Comparison of BE Assessment Methods for Solid Oral Dosage Forms
| Performance Metric | In Vivo Pharmacokinetic Study | In Vitro Biowaiver (e.g., Dissolution) | Context of Use |
|---|---|---|---|
| Direct Cost | High (e.g., $22-38 million/year industry-wide for Class I drugs) [51] | Low | Applies to BCS Class I and III drugs with rapid dissolution [51]. |
| Ethical Consideration | Involves human testing | No human testing | Aligns with "No unnecessary human testing" principle [51]. |
| Primary Measured Endpoint | Systemic drug exposure (indirect) | Drug product dissolution (direct performance) | Directly assesses product performance [51]. |
| Regulatory Acceptance | Common for most products | For BCS Class I (high solubility/high permeability) and some Class III (high solubility/low permeability) with very rapid dissolution [51] | Refer to FDA/EMA BCS guidance [51]. |
Experimental Protocol: BCS-Based Biowaiver
Objective: To justify a biowaiver using the Biopharmaceutics Classification System (BCS) and in vitro dissolution testing, avoiding an in vivo BE study. Methodology:
- BCS Classification: Determine the drug's solubility and permeability to classify it as BCS Class I (high solubility, high permeability) or Class III (high solubility, low permeability) [51].
- Dissolution Profile Comparison: Perform a comparative dissolution study of the test and reference products in three media: pH 1.2, 4.5, and 6.8, using the USP apparatus specified in the product monograph [51] [49].
- Acceptance Criteria: The test product is considered rapidly dissolving if ≥85% dissolves in ≤30 minutes (Class I) or ≤15 minutes (Class III) in all three media. The dissolution profiles of test and reference products must be similar [51].
Biomarker Analysis
Performance Comparison: Biomarker Qualification Levels
The integration of biomarkers into drug development requires a clear understanding of their validation and qualification status, which exists on a spectrum of scientific acceptance [52].
Table: Performance and Acceptance Levels of Biomarkers in Drug Development
| Biomarker Category | Analytical Validation | Clinical Significance | Regulatory Acceptance | Example |
|---|---|---|---|---|
| Exploratory Biomarker | May be in development | Used for internal decision-making; links animal to human data | Not submitted for regulatory decision-making [52] | VEGF evaluation for angiogenesis inhibitors [52]. |
| Probable Valid Biomarker | Measured with well-established performance characteristics | Established scientific framework exists; predictive value not widely accepted [52] | Can be submitted in investigational applications [52] | Examples often represent the stage between exploratory and known valid. |
| Known Valid Biomarker | Measured with well-established performance characteristics | Widespread agreement in the scientific community [52] | Can be used for regulatory decision-making; may be on drug label [52] | HER2/neu for breast cancer; K-RAS mutations for colorectal cancer [52]. |
Experimental Protocol: Analytical Method Validation for a Biomarker Assay
Objective: To validate an analytical method (e.g., an electrochemical immunoassay) for quantifying a protein biomarker according to USP/EP principles, ensuring it is fit for purpose [52] [53]. Methodology:
- Define Context of Use: Specify the biomarker's role and the required assay performance [54] [52].
- Select Validation Parameters: Assess key parameters per USP <1225>:
- Specificity: Ability to measure analyte in the presence of matrix components.
- Accuracy/Recovery: Agreement between measured and true value.
- Precision: Repeatability and intermediate precision.
- Linearity & Range: Directly proportional relationship between response and concentration.
- Limit of Detection (LOD) & Quantification (LOQ) [52] [49].
- System Suitability Testing: Before each analysis run, perform tests to verify that the analytical system (electrochemical instrument, electrodes, software) is functioning appropriately [49].
Integrated Workflow and Visualization
The following diagram illustrates the integrated logical relationship between pharmacopeial standards, analytical method validation, and their application across the drug product lifecycle.
Solving Common Challenges and Optimizing Electrochemical Assay Performance
For researchers and scientists in drug development, the Limit of Detection (LOD) represents a fundamental figure of merit that determines the lowest concentration of an analyte that can be reliably distinguished from a blank sample. Within the framework of pharmacopeial standards such as USP and EP, establishing a robust and validated LOD is not merely a technical exercise but a regulatory requirement that ensures analytical methods are fit-for-purpose in pharmaceutical quality control [55]. The LOD is formally defined as the smallest amount of an analyte in a sample that can be detected, though not necessarily quantified, with a reasonable statistical certainty [56]. This parameter becomes particularly crucial for methods detecting low levels of impurities, degradation products, or potent active compounds where trace-level detection directly impacts product safety and efficacy.
A persistent challenge across analytical chemistry, however, lies in the multitude of definitions and calculation methods for LOD, leading to potential inconsistencies in method validation and comparison [55]. The situation is further complicated by the "LOD paradox," where the relentless pursuit of lower detection limits does not always translate to improved practical utility for a given application [57]. This article provides a comprehensive comparison of strategies to overcome sensitivity limitations, focusing on experimentally-proven approaches while contextualizing them within the rigorous demands of pharmacopeial validation.
Foundational Concepts: Distinguishing Sensitivity from Detection Limit
A critical conceptual foundation for any improvement strategy is understanding that instrument sensitivity is not equivalent to detection limit [58]. Sensitivity refers to the magnitude of a signal change per unit change in analyte concentration (the slope of the calibration curve), while the LOD is determined by the signal-to-noise ratio (SNR) [58].
The LOD is fundamentally governed by the equation: LOD = k × (Noise / Sensitivity) Where 'k' is a statistical factor (typically 3 or 3.3), 'Noise' represents the variability of the blank signal, and 'Sensitivity' is the calibration slope [55] [59]. Consequently, improving the LOD can be achieved by either increasing the signal (improving sensitivity), reducing noise, or both simultaneously [56]. This relationship forms the basis for all optimization strategies discussed in subsequent sections.
Figure 1: Conceptual framework for LOD improvement. Effective strategies target either signal enhancement, noise reduction, or both, while considering their impact on practical sensitivity.
Systematic Strategies for LOD Improvement
Signal Enhancement Approaches
Sample Preparation and Pre-concentration represent the most direct approaches for signal enhancement. Techniques such as solid-phase microextraction (SPME), liquid-liquid extraction, and derivatization can significantly increase the effective concentration of the analyte presented to the analytical system [60]. In chromatographic applications, employing splitless injection instead of split injection directs nearly the entire sample onto the column, minimizing sample loss and enhancing detectability for trace components [60].
Detection System Optimization offers another pathway for signal enhancement. In HPLC with UV detection, ensuring detection occurs at the analyte's wavelength of maximum absorption (λmax) provides the strongest possible signal [56]. For multiple analytes, selecting a compromise wavelength or implementing multi-wavelength detection can optimize response across components. In electrochemical systems, selecting working electrode materials with favorable electrocatalytic properties toward the target analyte, such as boron-doped diamond (BDD) electrodes, can significantly enhance electron transfer kinetics and subsequent signal response [61].
Separation Efficiency Improvements indirectly enhance signals by producing sharper, taller chromatographic peaks. This can be achieved through column selection (e.g., smaller particle sizes), optimized mobile phase composition, and temperature control [56] [60]. In gas chromatography, using capillary columns with smaller inner diameters (e.g., 0.25 mm instead of 0.32 mm) enhances efficiency and produces sharper peaks, thereby increasing peak height and improving the signal-to-noise ratio [60].
Noise Reduction Strategies
Mobile Phase and Solvent Optimization can substantially reduce baseline noise. For LC-MS applications, selecting volatile solvents and additives that produce minimal ionization suppression reduces chemical noise [56]. In UV detection, using solvents with low UV cutoff values (e.g., acetonitrile instead of acetone) minimizes background absorbance [56]. Ensuring high purity of reagents and solvents prevents contaminant-related noise interference.
Instrument Maintenance and System Suitability practices are often overlooked but critically important. Regular replacement of chromatographic system components (septa, liners, ferrules) prevents contamination-related noise and ghost peaks [60]. Using high-purity carrier gases (≥99.999%) with proper purification traps in gas chromatography minimizes baseline noise and improves detector stability [60]. Establishing robust system suitability tests ensures the analytical system is operating within specified noise parameters before method execution.
Signal Processing and Data Treatment approaches can mathematically improve SNR. Modern analytical instruments often incorporate digital filtering algorithms that reduce high-frequency random noise without significantly distorting the analytical signal [60]. Adjusting integration parameters (peak width, threshold) appropriately can help distinguish small analyte peaks from background fluctuations, though such approaches require validation to ensure they don't introduce bias [60].
Table 1: Comprehensive LOD Improvement Strategies Across Analytical Techniques
| Approach Category | Specific Strategy | Applicable Techniques | Expected Improvement | Key Considerations |
|---|---|---|---|---|
| Sample Preparation | Solid-phase microextraction | GC, HPLC, Electroanalysis | 5-100x | Potential for analyte loss |
| Derivatization | GC, HPLC, CE | 3-50x | Reaction completeness critical | |
| Separation Optimization | Smaller ID columns | GC, HPLC | 2-5x | May increase backpressure |
| Temperature programming | GC, HPLC | 1-3x | Resolution trade-offs | |
| Detection Enhancement | λmax optimization | UV-Vis, FL | 2-10x | May not suit multi-analyte |
| Electrode material selection | Electrochemical | 5-100x | Matrix-dependent | |
| Noise Reduction | High-purity solvents | All | 1-3x | Cost consideration |
| System maintenance | All | 1-2x | Required for reproducibility | |
| Signal processing | All | 1-2x | Risk of signal distortion |
Methodological and Computational Approaches
Advanced Calibration Strategies can improve LOD reliability. Traditional approaches based on blank signal statistics (LOD = mean blank + 3.3 × standard deviation of blank) provide a foundation, but more sophisticated approaches like uncertainty profiling offer enhanced reliability, particularly for complex matrices [62]. This method combines tolerance intervals and measurement uncertainty to define a validity domain for the analytical method, providing a more realistic assessment of detection capabilities [62].
Experimental Design for LOD Determination significantly impacts the reliability of the calculated detection limit. The American Standards Board Standard 036 for forensic toxicology recommends analyzing three blank matrix samples in duplicate over three separate runs, allowing the LOD to be assessed as the mean blank signal plus 3.3 times the standard deviation of the blank signal [55]. This approach, performed under intermediate precision conditions, provides a more realistic estimation of method capabilities compared to single-day determinations.
Technique-Specific Optimization: Comparative Performance Data
Electrochemical Methods
Electrochemical techniques offer particular advantages for pharmaceutical analysis, including low reagent consumption, ease of operation, and rapid analysis times [61]. A comparative study of differential pulse voltammetry (DPV) using a boron-doped diamond electrode demonstrated LODs of 0.69 mg L⁻¹ for caffeine, 0.84 mg L⁻¹ for paracetamol, and 0.46 mg L⁻¹ for methyl orange in synthetic effluents and environmental water samples [61]. The study confirmed that electrochemical approaches can provide comparable performance to traditional techniques like HPLC and UV-vis spectrophotometry for certain applications, with the added benefits of simplicity and cost-effectiveness [61].
Electrode surface modification represents a powerful strategy for enhancing LOD in electrochemical methods. While not explicitly detailed in the search results, the principles can be inferred: modifying electrode surfaces with nanomaterials, molecularly imprinted polymers, or catalytic layers can significantly enhance electron transfer kinetics and selectively preconcentrate analytes at the electrode surface, thereby improving both sensitivity and LOD.
Chromatographic Techniques
For HPLC applications, comprehensive optimization involves multiple parameters. Mobile phase optimization with additives such as 0.1% formic acid (for amines) can reduce peak tailing, resulting in sharper, taller peaks and improved signal intensity [56]. Column selection also plays a crucial role; for example, hydride-based columns with Aqueous Normal Phase (ANP) chromatography have demonstrated superior performance for hydrophilic analytes compared to traditional reversed-phase approaches, often yielding improved peak shape and signal intensity [56].
In gas chromatography, detector selection and optimization significantly impact LOD. The typical sensitivity hierarchy for GC detectors is: MS > ECD > FPD > FID > TCD [60]. Selecting the detector most responsive to the target analyte class provides immediate LOD improvements. For instance, Electron Capture Detectors (ECD) offer exceptional sensitivity for halogenated compounds, while Flame Photometric Detectors (FPD) are ideal for sulfur and phosphorus compounds [60].
Table 2: Comparison of Analytical Techniques for Determining Emerging Contaminants
| Technique | Analyte | Reported LOD | Key Advantages | Limitations |
|---|---|---|---|---|
| Electrochemical (DPV with BDD) | Caffeine | 0.69 mg L⁻¹ | Low cost, rapid analysis | Matrix susceptibility |
| Paracetamol | 0.84 mg L⁻¹ | Portability potential | Limited multiplexing | |
| Methyl Orange | 0.46 mg L⁻¹ | Minimal sample prep | ||
| HPLC-UV | Not specified | Compound-dependent | High reproducibility | Higher solvent consumption |
| GC-MS | Not specified | Compound-dependent | Excellent identification | Sample derivatization often needed |
| UV-Vis Spectrophotometry | Not specified | Compound-dependent | Simplicity, low cost | Limited specificity |
Regulatory and Practical Considerations for Pharmacopeial Methods
Harmonization of LOD Determination Methods
The Pharmacopeial Discussion Group (PDG) works to harmonize general chapters across USP, EP, and JP, including those relevant to analytical method validation [18] [63]. For chromatography (General Chapter <621>), harmonization reached Stage 4 in September 2021, indicating official implementation across the participating pharmacopeias [63]. This harmonization provides a standardized framework for evaluating analytical methods, including LOD determination, facilitating global drug development and regulatory submissions.
Despite harmonization efforts, multiple calculation approaches for LOD persist in the scientific literature and practice. These include visual evaluation, signal-to-noise ratio, blank sample measurement, and calibration curve-based methods [55] [59]. Each approach has distinct advantages and limitations, and the choice of method should be justified based on the specific analytical technique, matrix, and intended application.
The "LOD Paradox": When Lower Isn't Always Better
A critical consideration in pharmaceutical analysis is that achieving ultra-low LOD values may not always be necessary or practical for the intended application [57]. The intense focus on pushing LOD boundaries can sometimes overlook other crucial factors such as detection range, ease of use, and market readiness, which are vital for analytical methods to be effective in real-world applications [57].
For clinical applications, the ability of a method to operate within the relevant biological range of a target analyte is often more critical than detecting trace levels well below the physiological concentrations [57]. A method capable of detecting picomolar concentrations of a biomarker represents an impressive technical achievement, but if the biomarker's clinical relevance occurs in the nanomolar range, such sensitivity becomes redundant and may complicate the method without adding practical value [57].
Figure 2: Balancing factors in LOD optimization for pharmacopeial methods. Successful method development requires equilibrium between technical capabilities, practical utility, and regulatory requirements.
Experimental Protocols for LOD Determination
Protocol for LOD Determination via Blank Standard Deviation
This protocol aligns with approaches recommended by multiple regulatory bodies and is particularly suitable for chromatographic and spectrophotometric methods [55] [59]:
- Prepare a minimum of 6 independent blank samples using the appropriate matrix (placebo formulation for drug product, biological fluid for bioanalysis, etc.)
- Process all blanks through the entire analytical procedure, including extraction, derivatization, and analysis
- Analyze blanks over at least three separate runs performed by different analysts on different days to incorporate intermediate precision
- Record the response for each blank at the retention time/migration time of the analyte
- Calculate the mean (X̄B) and standard deviation (σB) of the blank responses
- Compute the LOD using the formula: LOD = X̄B + 3.3 × σB
- Verify the calculated LOD experimentally by analyzing a minimum of 5 samples fortified at the calculated LOD concentration; the analyte should be detected in at least 90% of the samples
Protocol for LOD Determination via Uncertainty Profile
The uncertainty profile approach represents a more advanced graphical strategy for LOD and LOQ determination that incorporates measurement uncertainty [62]:
- Analyze validation standards at multiple concentration levels across the expected range, including levels near the anticipated LOD/LOQ
- Perform analyses over multiple series (e.g., different days, analysts, instruments) to capture method variability
- For each concentration level, compute the β-content tolerance interval using the formula: β-TI = Ȳ ± ktol × σ̂m where Ȳ is the mean result, ktol is the tolerance factor, and σ̂m is the estimate of reproducibility standard deviation [62]
- Calculate measurement uncertainty u(Y) for each level using the formula: u(Y) = (U - L) / [2 × t(ν)] where U and L are the upper and lower tolerance limits, and t(ν) is the Student t quantile [62]
- Construct the uncertainty profile by plotting |Ȳ ± k × u(Y)| against concentration, where k is a coverage factor (typically 2 for 95% confidence)
- Define acceptance limits (λ) based on the intended method purpose and regulatory requirements
- Determine the LOQ as the lowest concentration where the entire uncertainty interval falls within the acceptance limits
- Establish the LOD as the concentration corresponding to the intersection of the uncertainty profile with the acceptance limits, typically yielding an LOD approximately one-third of the LOQ
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents and Materials for LOD Optimization
| Reagent/Material | Function in LOD Improvement | Application Examples |
|---|---|---|
| High-Purity Solvents (HPLC/MS grade) | Reduces chemical noise and background interference | Mobile phase preparation, sample reconstitution |
| Formic Acid/Acetic Acid (0.1%) | Improves ionization efficiency and peak shape in LC-MS | Mobile phase additive for acidic compounds |
| TFA (Trifluoroacetic Acid) | Enhances UV detection sensitivity for peptides/proteins | LC-UV mobile phase additive (non-MS applications) |
| Solid-Phase Extraction Cartridges | Pre-concentrates analytes and cleans sample matrix | Biological sample preparation, environmental analysis |
| Derivatization Reagents | Enhances detectability of poorly responding analytes | GC analysis of polar compounds, fluorescence detection |
| Boron-Doped Diamond Electrodes | Provides low background current and wide potential window | Electrochemical detection of oxidizable compounds |
| Molecularly Imprinted Polymers | Selectively preconcentrates target analytes | Sensor development, sample clean-up |
Overcoming sensitivity limitations requires a systematic, multifaceted approach that balances technical capabilities with practical utility and regulatory requirements. The most effective strategies simultaneously address signal enhancement through sample pre-concentration, detection optimization, and separation efficiency improvements, while implementing noise reduction via solvent purity, system maintenance, and appropriate signal processing.
For researchers working within pharmacopeial frameworks, understanding the principles of LOD determination and the available computational approaches—from traditional blank-based calculations to advanced uncertainty profiling—enables informed selection of fit-for-purpose methodology. The comparison of analytical techniques presented in this guide provides a foundation for selecting and optimizing methods based on specific application requirements.
Ultimately, successful LOD improvement strategies must align with the intended use of the analytical method, recognizing that the pursuit of lower detection limits should be guided by practical necessity rather than technical possibility alone. By adopting the systematic approaches outlined in this guide, researchers can develop robust, sensitive methods that meet the rigorous demands of modern pharmaceutical analysis while remaining practical for routine application.
For researchers and scientists in drug development, ensuring the specificity of an analytical method is a critical benchmark in the validation process. Specificity is the ability to assess unequivocally the analyte in the presence of components that may be expected to be present, such as impurities, degradation products, and excipients. Within the framework of United States Pharmacopeia (USP) and European Pharmacopoeia (EP) guidelines, this parameter is not merely a recommendation but a mandatory requirement for method validation, ensuring that the measured signal originates solely from the active pharmaceutical ingredient (API) without interference. The United States Pharmacopeia–National Formulary (USP–NF) and the European Pharmacopoeia establish strict scientific standards for pharmaceutical product requirements concerning identity, strength, quality, and purity [64].
Electrochemical methods offer significant advantages for pharmaceutical analysis, including high sensitivity, cost-effectiveness, and the capacity for rapid, on-site analysis [65] [66]. However, their application in regulated environments is contingent on a rigorous demonstration of specificity. This guide provides a comparative analysis of strategies and experimental protocols, grounded in recent research, to manage interference effectively, ensuring that electrochemical methods meet the stringent criteria set forth by pharmacopeial standards.
Strategic Approaches to Specificity: A Comparative Guide
Managing interference requires a multi-faceted strategy, from the initial selection of electrode materials to the final data analysis. The following table compares core experimental components and their role in ensuring specificity.
Table 1: Comparison of Strategic Components for Managing Interference
| Strategic Component | Function in Ensuring Specificity | Key Comparisons and Experimental Findings |
|---|---|---|
| Electrode Material Selection | Different electrode materials offer varying electrocatalytic properties, potential windows, and susceptibilities to surface fouling, directly influencing selectivity for the target API. | - Borou-Doped Diamond (BDD): Exhibits a wide potential window and high stability. A study on abacavir showed effective API oxidation with minimal passivation [67].- Platinum (Pt): Effective for indirect oxidation via generated hydroxyl radicals. Research on venlafaxine demonstrated high degradation efficiency (94%) despite no direct oxidation peak, indicating utility for radical-mediated processes [68].- Glassy Carbon (GC): Allows for direct oxidation of electroactive compounds but may be prone to fouling. Voltammetry of venlafaxine on GC showed a distinct oxidation peak at ~0.74 V [68]. |
| Chemometric Analysis | Multivariate statistical tools like Principal Component Analysis (PCA) and Hierarchical Cluster Analysis (HCA) objectively classify degradation profiles and identify the electrochemical method that best mimics traditional forced degradation. | - A study on maraviroc used PCA and HCA to demonstrate that electrochemical degradation profiles on Pt and Au electrodes clustered closely with oxidative (H₂O₂) and photolytic stress samples, validating the electrochemical approach as a suitable mimic for forced degradation studies [69]. |
| Separation Coupled with Detection | Coupling an electrochemical flow cell (for degradation) directly to a separation technique like LC-MS isolates the API from its degradation products for individual identification and quantification. | - This approach was used for abacavir and maraviroc, where electrolysis products were directly analyzed by UHPLC-MS. This allows for the precise identification of degradation products (e.g., m/z 319.20 and 247.19 for abacavir) and confirmation that the API signal is resolved from impurity signals [67] [69]. |
| Method Optimization (pH/Electrolyte) | Fine-tuning the supporting electrolyte and pH can drastically alter the electrochemical behavior of the API versus potential interferents, enhancing selectivity. | - The electrochemical degradation of abacavir was strongly pH-dependent, with the fastest oxidation at pH 9. pH also affected the composition of the degradation products [67].- For venlafaxine, a 0.1 M Na₂SO₄ solution at pH 9 was optimal, generating hydroxyl and sulfate radicals responsible for degradation while avoiding chloride-based electrolytes that form harmful by-products [68]. |
The following diagram illustrates the logical workflow for developing and validating a specific electrochemical method, integrating the strategies outlined above.
Diagram 1: A strategic workflow for developing specific electrochemical methods, integrating material selection, condition optimization, and advanced data analysis to meet regulatory requirements.
Experimental Protocols for Specificity Assessment
The following section provides detailed methodologies for key experiments cited in the comparison guide, which can be adapted to assess the specificity of your electrochemical method for a target API.
Protocol: Electrochemical Oxidative Stability Testing with LC-MS Analysis
This protocol, adapted from a study on abacavir, is designed to generate and identify degradation products, thereby testing the method's ability to distinguish the API from its impurities [67].
1. Materials and Reagents:
- API standard (e.g., Abacavir Sulfate ≥ 99.0%)
- Supporting electrolyte (e.g., Ammonium acetate buffer, 0.1 M)
- pH adjustment solutions (e.g., Acetic acid, Ammonium hydroxide)
- LC-MS grade solvents (Acetonitrile, Water)
2. Instrumentation:
- Potentiostat/Galvanostat
- Electrochemical Cell: BDD or Pt working electrode, Pt counter electrode, Ag/AgCl reference electrode
- UHPLC system coupled to a Mass Spectrometer (e.g., QDa or Q-TOF)
- pH Meter
3. Procedure:
- Step 1: Solution Preparation. Prepare a solution of the API (e.g., 1 mmol L⁻¹) in the selected supporting electrolyte. Adjust the pH to the desired value (e.g., pH 3, 7, and 9 for a pH-dependency study).
- Step 2: Electrochemical Degradation. Transfer the solution to the electrochemical cell. Apply a constant potential (e.g., +1.15 V for a large-surface Pt electrode or +4.0 V for a BDD electrode [67]) or current density. Perform electrolysis for a predetermined time or until approximately 5-20% degradation is achieved, as recommended for pharmaceutical stability studies [67].
- Step 3: Sample Analysis. Inject samples (pre- and post-electrolysis) into the UHPLC-MS system.
- Chromatographic Conditions: Use a suitable column (e.g., C18). Employ a gradient elution with mobile phases such as 0.1% formic acid in water and acetonitrile to achieve separation of the API from its degradation products.
- Mass Spectrometry: Operate in positive or negative electrospray ionization (ESI) mode. Use full-scan and MS/MS modes to obtain the mass-to-charge ratio (m/z) of the API and its degradation products.
4. Data Interpretation:
- Identify the retention times of the API and any new peaks appearing after electrolysis.
- Use the MS and MS/MS data to elucidate the structure of the degradation products by determining their molecular formulas and fragment ions.
- Confirm specificity by demonstrating that the analytical method can baseline resolve the API peak from all degradation product peaks.
Protocol: Voltammetric Specificity Screening with Chemometric Analysis
This protocol, based on research with maraviroc, uses voltammetry on different electrodes combined with chemometrics to rapidly compare degradation profiles and select the optimal electrochemical system [69].
1. Materials and Reagents:
- API standard (e.g., Maraviroc)
- Buffer solution (e.g., Phosphate buffer, pH 7.4)
- Acetonitrile
- Screen-printed electrodes (SPE): Platinum (Pt), Gold (Au), and Glassy Carbon (GC)
2. Instrumentation:
- Voltammetric Analyzer
- Ultrasonic Bath
3. Procedure:
- Step 1: Solution Preparation. Prepare a solution of the API in a mixture of phosphate buffer (pH 7.4) and acetonitrile (50:50, v/v).
- Step 2: Electrochemical Transformation. On each type of SPE (Pt, Au, GC), perform an electrochemical experiment by applying a high constant potential (e.g., +1400 mV) for a fixed duration [69].
- Step 3: Chromatographic Profiling. Analyze the solutions from each electrode, along with samples from traditional forced degradation (oxidative, photolytic, hydrolytic), using UHPLC with high-resolution MS detection.
- Step 4: Chemometric Analysis.
- Data Alignment: Input the chromatographic data (e.g., peak abundances/areas of all detected ions) into chemometric software (e.g., Mass Profiler Professional).
- Multivariate Analysis: Perform Principal Component Analysis (PCA) to visualize the clustering of samples. Electrochemical samples that cluster with traditional forced degradation samples (e.g., H₂O₂ oxidation) are considered to effectively mimic the relevant degradation pathway.
- Hierarchical Cluster Analysis (HCA): Use HCA (Ward's linkage, Euclidean distance) to generate a dendrogram and heatmap, further confirming the relationships between different stress conditions.
4. Data Interpretation:
- The electrode material whose electrochemical profile is most similar to the traditional forced degradation profiles, as determined by PCA and HCA, is identified as the optimal choice for further method development.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Electrochemical Specificity Studies
| Item | Function in Research | Application Note |
|---|---|---|
| USP/EP Reference Standards | Highly purified and characterized substances used for system suitability testing, identification, and quantification to ensure data reliability and regulatory compliance [64]. | Critical for calibrating instruments and validating that the analytical method is suitable for its intended use, as required by USP 〈1225〉 and related chapters. |
| Boron-Doped Diamond (BDD) Electrode | An inert electrode with a wide potential window and low background current, ideal for generating hydroxyl radicals for indirect oxidation and minimizing electrode fouling [67] [68]. | Preferred for anodic oxidation processes where high potentials are needed and for generating consistent degradation profiles comparable to chemical oxidation. |
| Platinum Electrode | A stable electrode with a well-established surface cleaning procedure, ensuring high reproducibility. Effective for both direct and indirect oxidation mechanisms [68]. | A robust alternative to BDD, particularly when a reproducible surface is paramount. Its stability allows for extended use. |
| Screen-Printed Electrodes (SPEs) | Disposable, low-cost electrochemical cells integrated onto a small chip, enabling rapid, high-throughput screening of electrochemical behavior [69]. | Ideal for preliminary studies to quickly compare the performance of different electrode materials (Pt, Au, GC) and experimental conditions. |
| Chemometric Software | Software platforms enabling multivariate statistical analysis (PCA, HCA) of complex chromatographic and spectral data to objectively compare degradation profiles [69]. | Essential for providing data-driven evidence that an electrochemical degradation method adequately mimics a traditional forced degradation pathway. |
Ensuring specificity in electrochemical methods is an achievable goal through a strategic combination of modern materials, optimized experimental conditions, and advanced data analysis techniques. As demonstrated by recent research, the conscious selection of electrode materials like BDD or platinum, the careful optimization of pH and electrolyte, and the objective power of chemometrics provide a robust framework for managing interference from excipients and degradation products. By adhering to the detailed experimental protocols and utilizing the essential tools outlined in this guide, scientists and drug development professionals can confidently develop and validate electrochemical methods that not only meet the rigorous demands of USP and EP pharmacopeias but also offer efficient, cost-effective, and insightful alternatives to conventional analytical techniques.
Electrochemical methods have become indispensable in pharmaceutical analysis and drug development, playing a pivotal role in drug delivery systems, biomarker detection, and quality control. The precision of these methods hinges on the meticulous optimization of key operational parameters, including electrode modification strategies, solution pH, deposition time, and waveform settings. Within the framework of United States Pharmacopeia (USP) and European Pharmacopoeia (EP) validation guidelines, demonstrating control over these parameters is not merely a best practice but a regulatory requirement for ensuring the reliability, reproducibility, and accuracy of analytical data. This guide provides a comparative examination of how these parameters influence the performance of different electrochemical techniques and platforms, supported by experimental data, to aid researchers in making informed decisions for method development and validation.
Comparative Analysis of Electrode Modification Strategies
Electrode modification is a fundamental step for enhancing sensor selectivity, sensitivity, and stability. Different modification strategies cater to specific analytical challenges.
Table 1: Comparison of Electrode Modification Techniques
| Modification Technique | Material/Platform Used | Key Performance Outcomes | Optimal Parameters | Best For |
|---|---|---|---|---|
| Gold Nanoparticle Electrodeposition [70] | SPCE with [AuCl4]- solution | Stable, reproducible platform; LOD: 0.56 μg/mL for NGAL; Sensitivity: 21.8 μA mL/μg | Potential sweep: 1.1 V to -0.1 V; Optimization of Au concentration & scan number | Label-free immunosensors (e.g., AKI biomarker detection) |
| Polymer Size-Exclusion Membrane [71] | Carbon-fiber microelectrode with electrodeposited 1,3-phenylenediamine (mPD) | Selective H2O2 detection in brain tissue; exclusion of interferents (adenosine, histamine) | Electrodeposition of mPD to form a selective membrane | In vivo neurochemical monitoring (e.g., Parkinson's disease research) |
| Self-Assembled Monolayer (SAM) [70] | Gold electrodeposited platform with 3-mercaptopropionic acid (MPA) | Enabled antibody immobilization via EDC/NHS coupling | SAM formation followed by functionalization | Biosensor development for specific protein detection |
Experimental Protocols for Key Modifications
Protocol 1: Optimization of a Gold Electrodeposited Platform [70]
- Objective: To create a stable and reproducible platform for unlabelled immunosensors.
- Materials: Screen-printed carbon electrodes (SPCEs), gold chloride solution ([AuCl4]-), phosphate buffered saline (PBS).
- Methodology:
- The modification is performed in a [AuCl4]- solution by sweeping the electrode potential between 1.1 V and -0.1 V vs an Ag/AgCl reference electrode.
- The influence of critical parameters, including gold concentration and the number of electrodeposition scans, is systematically investigated.
- The optimized platform is characterized using Scanning Electron Microscopy (SEM) for surface morphology, Energy Dispersive X-ray (EDX) for elemental analysis, and Cyclic Voltammetry (CV) for electrochemical behavior.
- Outcome: The platform with the best electrochemical behavior, stability, and reproducibility is selected for immunosensor development.
Protocol 2: Fabrication of a Selective H₂O₂ Sensor [71]
- Objective: To develop a mechanically robust sensor for selective detection of H₂O₂ in the brain, excluding interferents like adenosine and histamine.
- Materials: Carbon-fiber microelectrodes, 1,3-phenylenediamine (mPD) solution, artificial cerebral spinal fluid (aCSF).
- Methodology:
- mPD is electrodeposited onto the carbon-fiber sensing surface from a freshly prepared solution.
- This process creates a size-exclusion polymer membrane that allows the smaller H₂O₂ molecule to pass while blocking larger interferents.
- The coated electrodes are calibrated using a flow-injection apparatus and validated in brain tissue.
- Outcome: A sensor capable of real-time, selective detection of H₂O₂ fluctuations in vivo, which is crucial for studying oxidative stress in neurodegenerative diseases.
The Influence of pH, Deposition Time, and Waveform Settings
Beyond the physical modification of the electrode, the conditions of the analytical procedure are equally critical.
Table 2: Impact of Key Experimental Parameters on Electrochemical Performance
| Parameter | Influence on Performance | Comparative Data from Studies |
|---|---|---|
| Solution pH | Affects redox reaction rates, stability of analytes, and charge state of functional groups on electrode surfaces. | A mobile phase for neurotransmitter analysis required a specific pH for optimal separation on a C18 column [72]. The stability of neurotransmitter standards was ensured using a perchloric acid solution [72]. |
| Deposition Time | Directly impacts the amount of analyte or modifying material on the electrode surface, influencing sensitivity and signal-to-noise ratio. | In gold electrodeposition, the number of scans (related to deposition time) significantly affected the average nanoparticle diameter and the platform's electrochemical properties [70]. |
| Waveform Settings | Determines the kinetic regime of the experiment, the selectivity, and the signal-to-background ratio, especially in techniques like FSCV. | For paracetamol analysis, a quasi-reversible system, the scan rate (ν) was critical for calculating kinetic parameters (α, D₀, k₀) [23]. For H₂O₂ detection, a waveform from -0.4 V to +1.4 V at 400 V/s was optimized [71]. |
- Objective: To accurately calculate the transfer coefficient (α), diffusion coefficient (D₀), and heterogeneous electron transfer rate constant (k₀) for an electroactive species.
- Materials: Electroactive species (e.g., paracetamol), supporting electrolyte (e.g., LiClO₄), three-electrode system (glassy carbon working electrode, Pt counter electrode, SCE reference electrode).
- Methodology:
- Cyclic Voltammetry: Run CV experiments at multiple scan rates (e.g., from 0.025 V/s to 0.300 V/s).
- Data Extraction: From each voltammogram, record the anodic and cathodic peak potentials (Epa, Epc) and peak currents (Ipa, Ipc).
- Parameter Calculation:
- Transfer Coefficient (α): Calculate using the equation derived from the peak potential and the half-peak potential (Ep − Ep/2).
- Diffusion Coefficient (D₀): Calculate using the modified Randles–Ševčík equation, which relates peak current to the scan rate and concentration.
- Heterogeneous Rate Constant (k₀): Calculate using reliable methods such as those described by Kochi and Gileadi. The study cautions that the method of Nicholson and Shain can overestimate k₀ values.
- Outcome: A set of key kinetic parameters that define the electrode process, essential for understanding and optimizing the electrochemical reaction.
Validation Within the USP/EP Pharmacopeial Framework
Adherence to USP and EP standards is mandatory for methods used in drug development and quality control. These standards provide a framework for ensuring identity, strength, quality, and purity [73].
- Method Validation (USP <1225>): The USP provides clear guidelines for the validation of analytical procedures. Parameters such as accuracy, precision, specificity, detection limit (LOD), quantitation limit (LOQ), linearity, and range must be established [73]. For example, the LOD and sensitivity reported for the NGAL immunosensor fall under this requirement [70].
- Instrument Qualification (USP <1058>): Analytical instruments must be properly qualified and calibrated to ensure their performance is fit for purpose [73].
- System Suitability (USP <621>): Tests such as chromatographic system suitability are required to confirm that the instrument and method are performing as expected at the time of analysis [73]. While from a chromatographic context, the principle applies to electrochemical systems to ensure day-to-day validity.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Electrochemical Method Development
| Item | Function/Benefit | Example Application in Context |
|---|---|---|
| Screen-Printed Carbon Electrodes (SPCEs) | Disposable, cost-effective, portable platforms ideal for decentralized sensing. | Base platform for gold electrodeposition in immunosensor development [70]. |
| Carbon-Fiber Microelectrodes | Small size (micrometers) suitable for in vivo sensing with high temporal resolution. | Used as the base sensor for mPD coating in H2O2 detection in the brain [71]. |
| USP/EP Reference Standards | Highly purified substances with certified properties for method validation, calibration, and quality control. | Essential for complying with pharmacopeial guidelines during analytical method development and transfer [73]. |
| Supporting Electrolytes (e.g., LiClO₄, PBS) | Carry current and control ionic strength, minimizing ohmic resistance and ensuring the migration of analyte is not the driving force. | Used in virtually all electrochemical experiments, including paracetamol CV [23] and H2O2 sensor calibration [71]. |
| Coupling Agents (e.g., EDC/NHS) | Facilitate the covalent immobilization of biomolecules (like antibodies) onto functionalized electrode surfaces. | Used to immobilize NGAL antibody onto the MPA SAM on the gold platform [70]. |
Workflow and Signaling Pathway Diagrams
Electrochemical Immunosensor Development Workflow
USP/EP Method Validation Pathway
The Critical Role of System Suitability Testing
System Suitability Testing (SST) serves as a critical quality control measure within analytical laboratories, ensuring that chromatographic or mass spectrometric systems operate within specified parameters for a given analytical method before, during, and after sample analysis. The core purpose of SST is to verify that the total analytical system—comprising the instrument, reagents, and operator—is capable of producing data of acceptable accuracy and precision for its intended application. In regulated environments, such as pharmaceutical development and quality control, SST is not merely a best practice but a mandatory requirement as outlined in various pharmacopoeias, including the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP) [30]. The fundamental principle underpinning SST is that a method, even after thorough validation, must be monitored during routine use to guarantee reproducible and reliable results, thereby forming a cornerstone of sound scientific publication and experimental repeatability [74].
The transition from older analytical techniques, such as thin-layer chromatography (TLC), to more sophisticated methods like high-performance liquid chromatography (HPLC) and capillary electrophoresis (CE) within pharmacopoeial monographs underscores the continuous pursuit of higher selectivity, sensitivity, and efficiency in quality assessment [30]. This evolution places greater emphasis on robust system suitability protocols. For instance, manual processing of data is not only time-consuming but also prone to subjective interpretation, where small fluctuations in parameters like peak retention times can be difficult to detect visually yet significantly impact reproducibility [75]. SST provides an objective, data-driven framework to mitigate these risks, ensuring the integrity of data generated for drug substance analysis, impurity profiling, and other critical analyses.
Key System Suitability Parameters and Experimental Protocols
System suitability evaluates a set of chromatographic or spectrometric parameters that collectively demonstrate the performance of the analytical system. These parameters are derived from the analysis of a standard reference material prepared and analyzed under the exact conditions specified by the analytical method.
Core SST Parameters and Acceptance Criteria
The following parameters are universally recognized as critical for assessing system performance in chromatographic methods:
- Retention Time (RT) and Retention Factor (k): SST confirms that the analyte of interest elutes at a consistent and expected retention time, demonstrating stable chromatographic conditions. Significant drift can indicate issues with the mobile phase composition, column degradation, or temperature fluctuations.
- Theoretical Plates (N): This parameter is a measure of column efficiency—the ability of the column to provide sharp, well-defined peaks. A higher number of theoretical plates indicates better efficiency. A drop in plate count can signal column deterioration or poor connection tubing.
- Tailing Factor (Tf): The tailing factor measures the symmetry of an analyte peak. Asymmetric or tailing peaks can lead to inaccurate integration and quantification. SST sets an acceptable maximum for the tailing factor to ensure peak symmetry.
- Resolution (Rs): Resolution measures the degree of separation between two analyte peaks, most critically between the peak of interest and the closest eluting potential interferent. Adequate resolution is fundamental for accurate quantification of individual components in a mixture.
- Precision/Repeatability: This is typically assessed by making multiple replicate injections of a standard solution and calculating the relative standard deviation (RSD%) of the analyte response (peak area or height). This verifies the injection system's precision and the system's overall stability.
- Signal-to-Noise Ratio (S/N): For methods requiring high sensitivity, the signal-to-noise ratio is evaluated to ensure the system can reliably detect and quantify the analyte at the levels of interest.
SST in High-Resolution Mass Spectrometry (HRMS)
For HRMS instruments, mass accuracy becomes a paramount SST parameter. High mass accuracy is essential for the reliable identification and characterization of compounds, particularly in non-targeted screening. A practical SST approach involves analyzing a set of reference standards encompassing a range of polarities and chemical families before and after sample analysis batches [76]. This protocol assesses the impact of factors like time since last calibration and batch length on instrumental performance. The SST ensures mass accuracy errors remain below a critical threshold (e.g., 3 ppm), preventing false negatives and enabling correct molecular formula assignment [76]. An example HRAM-SST (High-Resolution Accurate Mass-System Suitability Test) mixture might include 13 compounds like acetaminophen, caffeine, carbamazepine, and various perfluorinated compounds to test a diverse chemical space [76].
Detailed Experimental Protocol for HPLC-ECD System Suitability
The following protocol is adapted from a validated method for the simultaneous analysis of nine neurotransmitters in rat brain samples using High-Performance Liquid Chromatography with Electrochemical Detection (HPLC-ECD) [72].
- Instrumentation: HPLC system equipped with a DECADE II electrochemical detector and a 150 mm x 4.6 mm, 2.6 µm Kinetex F5 column (Phenomenex, USA) [72].
- Mobile Phase: 0.07 M KH₂PO₄, 20 mM citric acid, 5.3 mM OSA, 100 mM EDTA, 3.1 mM TEA, 8 mM KCl, and 11% (v/v) methanol in water. The pH is adjusted as required. The mobile phase must be filtered through a 0.22 µM cellulose acetate filter before use [72].
- SST Standard Solution: A mixture of dopamine, homovanillic acid, vanilmandelic acid, serotonin, 5-hydroxyindole-3-acetic acid, 4-hydroxy-3-methoxyphenylglycol, norepinephrine, 3,4-dihydroxyphenylacetic acid, and 3-methoxytyramine is prepared in a stability solution of 0.1 M perchloric acid and 0.1 mM sodium metabisulfite [72].
- Chromatographic Conditions: Isocratic elution at a specified flow rate (e.g., 1.0 mL/min) and column temperature (e.g., 30°C). The electrochemical detector potential is set appropriately for the analytes.
- SST Execution and Evaluation: A minimum of six replicate injections of the SST standard solution are performed. The system is deemed suitable if the calculated parameters—such as retention time variability (RSD < 1%), theoretical plates, tailing factor, and resolution between critical pairs—meet pre-defined acceptance criteria established during method validation [72].
Comparative Performance Data and Visualization
Quantitative SST Parameter Comparison: HPLC-ECD vs. UHPLC-Orbitrap-HRMS
The table below summarizes key SST parameters and typical acceptance criteria for two distinct analytical platforms, illustrating how SST requirements are tailored to the technique's specific strengths and applications.
Table 1: System Suitability Parameters Across Analytical Techniques
| SST Parameter | HPLC-ECD (Neurotransmitter Analysis) [72] | UHPLC-Orbitrap-HRMS (Small Molecule Screening) [76] | Rationale for Parameter | |
|---|---|---|---|---|
| Primary Metric | Chromatographic Resolution & Precision | Mass Accuracy (ppm) | Ensures separation of co-eluting isomers and reliable quantification. | Confirms correct mass assignment for molecular formula determination. |
| Key Acceptance Criterion | Resolution (Rs) > 1.5 between critical pairs | Mass error < 3 ppm | Baseline separation is critical for accurate electrochemical detection of individual analytes. | Essential for reliable identification in suspect and non-target screening. |
| Precision (Repeatability) | RSD of retention times and peak areas < 1-2% | RSD of mass accuracy measurements < specified limit | Verifies injection and detection system stability for quantitative analysis. | Ensures mass measurement consistency across the analytical batch. |
| System Stability Check | Multiple replicate injections of analyte standard | Injections of HRAM-SST mix before and after sample batch | Directly assesses the system's performance for the specific analytes of interest. | Monitors for mass drift over time and throughout the batch sequence. |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for System Suitability Testing
| Item | Function in SST | Example from Literature |
|---|---|---|
| System Suitability Test Mix | A solution of certified reference materials used to verify system performance against predefined criteria. | A mix of 13 compounds for HRAM-SST [76]; a mix of 9 neurotransmitters for HPLC-ECD [72]. |
| Stability Solution | A matrix used to prepare and store standard solutions to prevent analyte degradation prior to analysis. | 0.1 M perchloric acid with 0.1 mM sodium metabisulfite for catecholamines [72]. |
| Mobile Phase Additives | Ion-pairing agents, buffers, and antioxidants that ensure stable baselines, good peak shape, and analyte stability. | Octanesulfonic acid (OSA), EDTA, and citric acid in neurotransmitter HPLC-ECD [72]. |
| Chromatographic Column | The stationary phase that provides the required selectivity and efficiency for the separation. | 150 mm x 4.6 mm, 2.6 µm Kinetex F5 column [72]. |
System Suitability Testing Workflow
The following diagram illustrates the logical workflow and decision process for implementing System Suitability Testing within an analytical run.
System Suitability Testing is an indispensable, non-negotiable component of analytical method application in pharmacopoeial and research contexts. It provides the empirical evidence required to trust the data generated, acting as the final gatekeeper before sample analysis commences. As demonstrated by the protocols for HPLC-ECD and HRMS, SST parameters are specifically chosen to guard against the specific failure modes of each analytical technique, whether related to chromatographic separation or mass measurement accuracy. The implementation of automated SST analysis, as seen in industry case studies, further enhances objectivity, enables long-term trend analysis for preventive maintenance, and minimizes system downtime [75]. Ultimately, a rigorously applied SST protocol is a direct reflection of scientific diligence, ensuring that experimental results are not only valid and reliable but also worthy of inclusion in a sound scientific publication.
Best Practices for Data Integrity and Management of Reference Standards
Reference standards are essential components in the pharmaceutical laboratory, serving as established benchmarks for evaluating the identity, strength, quality, and purity of drug substances and products. Within the framework of United States Pharmacopeia (USP) and European Pharmacopoeia (EP) compliance, these standards provide the foundation for ensuring analytical method validity, particularly for electrochemical and other instrumental techniques. The USP operates as an independent non-profit scientific organization that creates stringent scientific standards for pharmaceutical ingredients, while the EP functions as Europe's authoritative reference for pharmaceutical quality testing under the European Directorate for the Quality of Medicines & HealthCare (EDQM) [77].
Both USP and EP supply Reference Standards that have undergone rigorous purification and verification processes. These standards are indispensable for drug identification testing, impurity analysis, and quality control throughout pharmaceutical development and manufacturing [77]. The effective management of these standards directly impacts data integrity—ensuring data remains accurate, consistent, and reliable throughout its lifecycle—which is crucial for regulatory compliance and patient safety [78]. Maintaining data integrity requires adherence to core principles including accuracy, consistency, completeness, and security [78], all of which are particularly critical when working with reference materials that form the basis of analytical decision-making.
Foundational Principles of Data Integrity for Reference Materials
Data integrity ensures that data remains accurate, consistent, and reliable throughout its entire lifecycle, from creation through storage and ultimate usage [78]. For reference standard management, this encompasses several critical dimensions:
- Entity Integrity: Utilizes unique identifiers for each reference standard batch to prevent duplicates and ensure traceability [78].
- Referential Integrity: Maintains proper relationships between datasets, such as linking calibration results with specific reference standard certificates [78].
- Domain Integrity: Restricts data entry to specific formats or ranges appropriate for reference standard documentation [78].
The core principles supporting data integrity include accuracy (data should reflect true values), consistency (data remains uniform across different systems), completeness (every required data point must be fully recorded), and security (protecting data from unauthorized changes) [78]. These principles form the foundation of trustworthy reference standard management and are essential for regulatory compliance.
Comprehensive Data Management Framework
Data Collection and Documentation Protocols
Effective data management begins with systematic collection and documentation practices. A Case Report Form (CRF) serves as a powerful tool for recording pertinent information, whether using paper or electronic formats [79]. Electronic CRFs offer advanced features like validation rules to minimize errors during data entry [79]. For reference standards, essential documentation includes:
- Certificate of Analysis with complete characterization data
- Batch-specific identification and traceability information
- Storage conditions and stability profiles
- Usage logs and requalification records
Well-defined Standard Operating Procedures (SOPs) must be established to manage experimental plans, sample handling, method validation, data recording, and archiving processes [77]. These procedures ensure all data is complete and traceable, facilitating audits or reviews when required.
Data Cleaning, Transformation, and Storage
Data cleaning involves systematic approaches to identify and rectify errors while maintaining logs of all steps performed [79]. Key aspects include:
- Checking for missing data and implementing appropriate handling strategies
- Running data validation checks to identify implausible values
- Correcting errors when possible or marking them for deletion
Data transformation includes re-categorization and re-codification of collected data, plus derivation of new variables to align with analytical plans [79]. Examples include categorizing continuous variables or recoding free-text values into standardized formats. Maintaining a code-book of definitions for all variables helps analysts understand the data thoroughly [79].
Secure data storage is crucial, particularly when data contains protected health information or proprietary method details [79]. Electronic data should be stored on secure institutional servers, never on unencrypted portable media [79]. Implementing role-based access controls (RBAC) ensures users can only access data necessary for their specific tasks, reducing risks of unauthorized access or manipulation [80] [78].
Experimental Protocols for Reference Standard Qualification
Method Validation Requirements
The validation of analytical methods using reference standards must assess multiple critical parameters as outlined in USP <1225>, including specificity, precision, accuracy, linearity, detection limit, and quantitation limit [77]. The validation process follows specific steps:
- Method Development: Analytical methods should adhere to USP/EP guidelines [77].
- Method Validation: Demonstration of method performance through experimental data [77].
- Method Transfer: Ensuring method uniformity across various laboratory settings [77].
- Method Revalidation: Required when significant changes in conditions occur [77].
System suitability testing functions as a fundamental validation component to assess the appropriateness of both instrument and method for the intended analysis [77]. For chromatographic systems, USP <621> requirements must be followed when conducting these tests [77].
Instrument Qualification and Calibration
USP <1058> requires laboratories to conduct both qualification and calibration of analytical instruments to validate their performance meets required expectations [77]. This includes:
- Regular calibration following established schedules
- Performance verification using traceable reference materials
- Comprehensive documentation of all qualification activities
Table 1: Essential Research Reagent Solutions for Pharmacopeial Analysis
| Reagent/Material | Function | Pharmacopeial Reference |
|---|---|---|
| USP Reference Standards | Calibration and qualification of analytical methods | USP-NF [77] |
| EP Reference Standards | Quality control testing for European markets | EP Monographs [77] |
| System Suitability Test Materials | Verify chromatographic system performance | USP <621> [77] |
| Qualified Analytical Instruments | Generate valid analytical data | USP <1058> [77] |
| pH Calibration Buffers | Calibrate pH measurement systems | USP <791> [77] |
Best Practices for Implementing Data Quality Checks
Data Validation Techniques
Robust data validation and verification techniques help ensure data meets required standards at every lifecycle stage [78]. For reference standard management, key validation checks include:
- Validity Checks (Schema Validation): Ensure data conforms to specified formats, schemas, or business rules through data type validation, pattern matching, range checks, and null checks [81].
- Uniqueness Checks: Verify that certain fields or sets of fields contain unique values without duplication, particularly important for reference standard batch numbers and certificates [81].
- Completeness Checks: Validate that no required data is missing, ensuring all fields critical for business logic, reporting, or analysis are populated [81].
- Accuracy Checks: Ensure data values are correct and reflect real-world entities by comparing data against trusted sources or known values [81].
Audit Trails and Monitoring
Maintaining detailed audit trails captures a record of data access, modifications, and user activities, helping detect suspicious behavior and ensuring data accountability [80]. Regular review of audit logs allows organizations to identify potential data breaches, security incidents, or unauthorized data access [80]. Implementation should include:
- Detailed logs of data changes, access activities, and system events
- Regular reviews to detect unusual or unauthorized activities
- Automated alerting for critical data modifications
- Data lineage tracking to ensure traceability from source to destination
Diagram: Integrated Workflow for Reference Standard and Data Management. This diagram illustrates the parallel processes of physical reference standard management and corresponding data integrity activities.
Laboratory Implementation of USP/EP Analytical Standards
Selection and Application of Reference Standards
Laboratories must use analytical methods that align with requirements established by USP and EP [77]. Key considerations include:
- Selection of Appropriate Analytical Methods: Choosing methods specified in relevant monographs and general chapters
- Use of Official Reference Standards: Applying USP or EP reference standards to maintain analytical result reliability
- Storage and Handling: Following established regulatory guidelines for storage conditions and usage procedures
The application of official pharmacopeial reference standards represents an essential procedure to maintain analytical result reliability, as these materials demonstrate high reliability and stability when obtained from authorized agencies [77].
Data Quality Management for Reference Standards
Implementing data quality management practices is essential to maintain accurate and actionable data for reference standard qualification [78]. Key techniques include:
- Data Cleansing: Regular updates to remove outdated information and ensure accuracy
- Deduplication: Preventing repeated entries in reference standard databases
- Routine Audits: Frequent audits to detect discrepancies and ensure regulatory compliance
Table 2: Data Quality Checkpoints for Reference Standard Management
| Checkpoint Stage | Quality Checks | Validation Techniques |
|---|---|---|
| Data Ingestion | Source system integrity, schema validation, missing data checks | Verify records match expected values, ensure conformity to predefined schema [81] |
| Data Staging | Field-level validation, business rule compliance, metadata validation | Check email formats, date formats, numeric ranges; validate timestamps and source identifiers [81] |
| Data Transformation | Transformation validation, referential integrity, data consistency | Verify transformations applied correctly, maintain foreign key relationships [81] |
| Data Loading | Load validation, data reconciliation, target schema validation | Check row counts before/after loading, compare source and destination systems [81] |
Tools and Technologies for Maintaining Data Integrity
Specialized tools simplify data integrity management by enforcing validation, access control, and quality checks [78]. For reference standard management, key tool categories include:
- Data Validation Tools: Open-source tools like Great Expectations allow users to define expectations for datasets and validate them at each process stage [81]. These tools support data profiling, schema validation, and custom rule enforcement.
- Electronic Data Capture Systems: Tools like REDCap (Research Electronic Data Capture) provide electronic case report forms with advanced features like setting validation rules to minimize errors during data collection [79].
- Data Quality Monitoring: Automated systems for continuous data quality assessment with alerting mechanisms for anomalies [78].
These tools enhance data integrity by ensuring reference standard data remains accurate, accessible, and compliant across different stages of the data lifecycle [78].
Regulatory Compliance and Consequences of Non-Compliance
Compliance Framework
Data integrity is not merely a best practice but a legal requirement with severe penalties for non-compliance [78]. Key regulatory frameworks impacting reference standard management include:
- FDA Regulations: Require pharmaceutical and food companies to maintain accurate, traceable data, with non-compliance potentially halting product approvals and incurring multi-million dollar fines [78].
- GDPR: Requires accurate, accessible data and the right for individuals to correct personal information, with violations incurring fines up to €20 million or 4% of annual revenue [78].
- HIPAA: Mandates accuracy and protection of patient data, with fines up to $1.5 million per year for non-compliance [78].
Consequences of Poor Data Quality
Compromised data integrity can lead to significant business impacts:
- Financial Losses: Bad data costs U.S. companies approximately $3.1 trillion annually [78], with poor data quality costing an average of $12.9 million per year for individual organizations [78].
- Operational Inefficiency: High data integrity minimizes costly errors and improves accuracy across workflows [78].
- Reputation Damage: Compromised data can severely damage organizational reputation and stakeholder trust [78].
Diagram: Method Validation Workflow for Reference Standard Qualification. This diagram outlines the comprehensive process for validating analytical methods using reference standards according to USP/EP requirements.
Effective data integrity practices for reference standard management require a comprehensive approach spanning people, processes, and technologies. By implementing robust data collection protocols, systematic quality checks, secure storage solutions, and continuous monitoring, organizations can ensure the accuracy, consistency, and reliability of reference standard data throughout its lifecycle. Adherence to USP and EP standards not only facilitates regulatory compliance but also enhances research efficiency, supports data-driven decision making, and ultimately safeguards product quality and patient safety.
The integration of these practices creates a foundation of trust in analytical results, enabling researchers and drug development professionals to generate reliable data that meets stringent regulatory requirements across global markets. As the pharmaceutical landscape continues to evolve with increasing data complexity and regulatory scrutiny, maintaining vigilant data integrity practices for reference standards remains paramount for successful drug development and commercialization.
Executing Validation Protocols and Navigating USP vs. EP Requirements
A Step-by-Step Guide to Full Method Validation per USP <1225> and ICH Guidelines
Analytical method validation is a critical process in pharmaceutical development, providing documented evidence that an analytical procedure is suitable for its intended purpose. For researchers and drug development professionals, validation is not a mere regulatory formality but a fundamental scientific activity that ensures the reliability, accuracy, and precision of the data used to make critical decisions about drug quality, safety, and efficacy. The United States Pharmacopeia (USP) General Chapter <1225> and the International Council for Harmonisation (ICH) guidelines provide the foundational framework for validation activities, with a recently proposed revision of USP <1225> aiming to create closer alignment with ICH Q2(R2) and integrate the procedure into the Analytical Procedure Life Cycle described in USP <1220> [10] [11].
This guide focuses specifically on the validation of electrochemical methods within this regulatory context. The principles outlined herein ensure that methods developed for assessing the quality of pharmaceutical products meet the proper standards of accuracy and reliability as required by current Good Manufacturing Practice (cGMP) regulations [21 CFR 211.194(a)] [9]. The evolving validation paradigm emphasized in the revised USP <1225> shifts from a static, compliance-focused event to a dynamic, lifecycle management process centered on the critical concept of fitness for purpose [11].
Regulatory Framework and the Lifecycle Approach
Harmonization of USP <1225> and ICH Q2(R2)
The regulatory landscape for analytical method validation is undergoing significant modernization. The proposed revision of USP <1225>, published in the Pharmacopeial Forum (PF 51(6), adapts the chapter for validation of both non-compendial and compendial analytical procedures and provides enhanced connectivity to related USP chapters, particularly USP <1220> Analytical Procedure Life Cycle [10]. This revision deliberately aligns with the principles of ICH Q2(R2) and ICH Q14 (Analytical Procedure Development), creating a unified, interconnected framework that demands a departure from treating validation as a discrete event toward embracing it as an ongoing commitment to analytical quality [11].
Key Concepts in Modern Method Validation
The revised approach introduces several pivotal concepts that reshape traditional validation practices:
- Reportable Result: The definitive analytical result that is reported and used for quality decisions (e.g., on a Certificate of Analysis). This concept forces validation to focus on the actual output used for decision-making, not just individual measurements [11].
- Fitness for Purpose: This principle positions validation as a process to demonstrate that a method is capable of producing reportable results that support confident decision-making for their intended use [10].
- Replication Strategy: The approach to replication during validation should reflect how the method will be used routinely to generate reportable results, ensuring that validation studies capture the same sources of variability encountered during routine use [10] [11].
- Validation Strategy and Study Design: The revised chapter continues to allow both minimal (traditional) and enhanced (Analytical Target Profile-based) validation approaches but clarifies that acceptance criteria should be linked to the performance requirements of the reportable result [10].
Validation Performance Characteristics: Definitions and Experimental Protocols
A thorough validation study for an electrochemical method must systematically evaluate specific performance characteristics. The following sections provide detailed definitions, experimental protocols, and acceptance criteria for each parameter.
Accuracy
Accuracy represents the closeness of agreement between the value found and the value accepted as a true or conventional reference value [9].
Experimental Protocol for Electrochemical Methods:
- Sample Preparation: Prepare a minimum of nine determinations across a minimum of three concentration levels (e.g., 80%, 100%, 120% of target concentration), with three replicates at each level [9].
- Reference Standard: Use analyte of known purity (e.g., a Reference Standard) for drug substance assay. For drug product assay, use synthetic mixtures of the drug product components to which known amounts of analyte have been added.
- Analysis: Analyze all samples using the validated electrochemical method.
- Calculation: Calculate recovery as a percentage of the known added amount:
Recovery % = (Measured Concentration / Known Concentration) × 100.
Precision
Precision expresses the closeness of agreement between a series of measurements obtained from multiple sampling of the same homogeneous sample under prescribed conditions [9].
Experimental Protocol for Electrochemical Methods: Precision should be evaluated at three levels:
- Repeatability: Assess under the same operating conditions over a short time interval using the same analyst and equipment. Perform a minimum of nine determinations covering the specified range (three concentrations/three replicates) or six determinations at 100% of test concentration [9].
- Intermediate Precision: Evaluate the method's robustness to variations within a laboratory (different days, different analysts, different equipment).
- Reproducibility: Assess precision between different laboratories (typically assessed during method transfer).
Table 1: Acceptance Criteria for Precision and Accuracy
| Analytical Characteristic | Validation Level | Recommended Acceptance Criteria |
|---|---|---|
| Precision (Repeatability) | Active Ingredient Assay | RSD ≤ 1.0% for HPLC/Electrochemical |
| Precision (Intermediate Precision) | Active Ingredient Assay | RSD ≤ 1.5-2.0% for HPLC/Electrochemical |
| Accuracy | Active Ingredient Assay | Mean Recovery 98.0-102.0% |
Specificity and Selectivity
Specificity is the ability to assess unequivocally the analyte in the presence of components that may be expected to be present, such as impurities, degradation products, and matrix components [9]. For electrochemical methods, this typically involves demonstrating that the method can distinguish the analyte from interfering species.
Experimental Protocol for Electrochemical Methods:
- Forced Degradation Studies: Subject the drug substance and product to various stress conditions: acid/base hydrolysis, oxidation, thermal degradation, and photolysis.
- Analysis of Interferences: Analyze samples containing potential interfering substances (excipients, impurities) to demonstrate separation from the analyte peak in voltammetric measurements.
- Peak Purity Assessment: For electrochemical techniques coupled with chromatographic separation, use appropriate techniques to demonstrate the analyte peak is attributable to a single component.
Detection Limit (DL) and Quantitation Limit (QL)
The Detection Limit is the lowest amount of analyte in a sample that can be detected but not necessarily quantitated. The Quantitation Limit is the lowest amount of analyte in a sample that can be quantitatively determined with suitable precision and accuracy [9].
Experimental Protocol for Electrochemical Methods:
- Visual Evaluation: For non-instrumental methods, analyze samples with known concentrations and establish the minimum level at which the analyte can be reliably detected.
- Signal-to-Noise Ratio: For instrumental procedures, typically a signal-to-noise ratio of 3:1 is acceptable for DL and 10:1 for QL.
- Standard Deviation of Response: Based on the standard deviation of the response and the slope:
DL = 3.3σ/SandQL = 10σ/S, where σ is the standard deviation of the response and S is the slope of the calibration curve.
Linearity and Range
Linearity is the ability of the method to obtain test results proportional to the concentration of analyte. Range is the interval between the upper and lower concentrations of analyte for which suitability has been demonstrated [9].
Experimental Protocol for Electrochemical Methods:
- Calibration Curve: Prepare a minimum of five concentration levels across the specified range.
- Analysis: Analyze each concentration level in triplicate.
- Statistical Analysis: Perform linear regression analysis on the results. Calculate correlation coefficient, y-intercept, slope, and residual sum of squares.
Table 2: Validation Characteristics for Different Types of Analytical Procedures
| Validation Characteristic | Identification | Testing for Impurities | Assay |
|---|---|---|---|
| Accuracy | - | Required | Required |
| Precision | - | Required | Required |
| Specificity | Required | Required | Required |
| Detection Limit | - | Required | - |
| Quantitation Limit | - | Required | - |
| Linearity | - | Required | Required |
| Range | - | Required | Required |
The Validation Workflow: A Step-by-Step Approach
The following diagram illustrates the complete method validation workflow, integrating all key activities from initial planning through final reporting.
Method Validation Workflow
Pre-Validation Planning
Before initiating laboratory work, thorough planning is essential:
- Define Validation Scope and Acceptance Criteria: Clearly identify the method's intended purpose and establish scientifically justified acceptance criteria for each validation parameter based on the analyte's properties and the method's application [10].
- Develop Validation Protocol: Create a comprehensive protocol detailing the experimental design, materials, responsibilities, and acceptance criteria. The protocol should be approved before study initiation.
Laboratory Execution and Evaluation
- Execute Experimental Studies: Conduct studies according to the approved protocol, following the specific experimental designs outlined in Section 3 of this guide.
- Document Results and Statistical Analysis: Record all raw data and perform appropriate statistical analysis. The revised USP <1225> emphasizes the use of Statistical Intervals (confidence, prediction, tolerance) as tools for evaluating precision and accuracy in relation to decision risk [10].
- Compare Results Against Acceptance Criteria: Evaluate all data against pre-defined acceptance criteria. Any deviations should be investigated and documented.
Post-Validation Activities
- Prepare Final Validation Report: Summarize all validation activities, results, and conclusions. The report should provide documented evidence that the method meets all pre-defined acceptance criteria and is fit for its intended purpose.
- Implement Method in Routine Use: Transfer the validated method to quality control laboratories following established technology transfer protocols.
- Ongoing Performance Verification (Stage 3): Implement procedures for continuous monitoring of method performance throughout its operational life, as emphasized in the lifecycle approach of USP <1220> [11].
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful validation of electrochemical methods requires specific reagents, materials, and equipment to ensure accurate and reproducible results.
Table 3: Essential Research Reagents and Materials for Electrochemical Method Validation
| Item | Function/Purpose | Key Considerations |
|---|---|---|
| Pharmacopeial Reference Standards | Provides verified analyte of known purity and composition for accuracy studies. | Must be obtained from official sources (USP, EP) with valid Certificate of Analysis. |
| High-Purity Supporting Electrolyte | Provides conductive medium while minimizing background current/interference. | Low metal ion content, electrochemical grade, appropriate buffering capacity. |
| Ultra-Pure Water Systems | Produces reagent water free of ionic contaminants that interfere with electrochemical signals. | Minimum 18.2 MΩ·cm resistivity, low TOC, regular system maintenance. |
| Verified Working Electrodes | Surface for electron transfer reactions; critical for signal reproducibility. | Material-specific pretreatment protocols, regular polishing/cleaning, established lifetime. |
| HPLC-Grade Solvents | Used for preparation of standard and sample solutions with minimal UV-absorbing impurities. | Low background signal, appropriate for electrochemical cell configuration. |
| Standardized Buffer Solutions | Controls solution pH which critically affects electrochemical behavior of many analytes. | Certified pH values, low conductivity, compatible with electrode materials. |
Advanced Concepts: Combined Evaluation and Lifecycle Management
The revised USP <1225> introduces more sophisticated approaches to validation that move beyond traditional parameter-checking:
- Combined Evaluation of Accuracy and Precision: The revised chapter describes approaches for evaluating accuracy and precision together using statistical intervals that account for both bias and variability simultaneously. This combined evaluation provides a more scientifically rigorous assessment of total error [10] [11].
- Knowledge Management: The revised chapter explicitly emphasizes knowledge management, acknowledging that validation should build upon data generated during method development, platform knowledge from similar methods, and experience with related products [11].
- Analytical Procedure Lifecycle Management: As outlined in USP <1220>, validation represents Stage 2 in the analytical procedure lifecycle, preceded by procedure development (Stage 1) and followed by ongoing procedure performance verification (Stage 3) [10] [11].
Full method validation per USP <1225> and ICH guidelines requires a systematic, scientifically rigorous approach that demonstrates fitness for purpose. The proposed revision of USP <1225> represents a significant evolution in validation philosophy, aligning with ICH Q2(R2) and emphasizing lifecycle management through concepts like the reportable result and risk-based replication strategy. For scientists validating electrochemical methods, this guide provides a comprehensive framework for designing, executing, and documenting validation studies that meet current regulatory expectations and ensure the generation of reliable, meaningful analytical data throughout the method's lifecycle.
Analytical method validation provides documented evidence that a laboratory procedure is robust, reliable, and reproducible for its intended purpose, forming the cornerstone of pharmaceutical quality control. Within drug development, validation parameters such as the Limit of Detection (LOD), Limit of Quantitation (LOQ), range, and robustness are not merely scientific benchmarks—they are enforceable requirements under pharmacopeial standards [82] [1]. The United States Pharmacopeia (USP) and European Pharmacopoeia (EP) establish legally-recognized standards for the identity, strength, quality, and purity of medicinal substances [1]. Compliance with these standards is mandatory for market approval, and regulatory bodies like the FDA and EMA rigorously assess these validation parameters during inspections [1].
The International Council for Harmonisation (ICH) Q2(R1) guideline provides the foundational framework for validating analytical procedures, a framework adopted by both USP and EP [9] [83]. For researchers developing electrochemical or other analytical methods, demonstrating compliance with these guidelines is critical. This process ensures that methods are "fit for purpose," meaning they can consistently produce reliable results that can be legally defended in the context of product release and regulatory submissions [84].
Core Definitions and Regulatory Importance
Critical Validation Parameters
- Limit of Detection (LOD): The lowest amount of analyte in a sample that can be detected—but not necessarily quantified as an exact value—under the stated experimental conditions [9]. It represents the point at which a measurand can be reliably distinguished from the analytical noise or blank [84] [85].
- Limit of Quantitation (LOQ): The lowest concentration of an analyte that can be quantitatively determined with suitable precision and accuracy [9] [86]. The LOQ is the level at which the method transitions from mere detection to reliable measurement [85].
- Range: The interval between the upper and lower concentrations of analyte in the sample for which it has been demonstrated that the analytical procedure has a suitable level of precision, accuracy, and linearity [9].
- Robustness: A measure of the analytical procedure's capacity to remain unaffected by small, but deliberate, variations in method parameters, such as pH, temperature, or mobile phase composition. It provides an indication of the method's reliability during normal usage and is closely related to ruggedness, which refers to the degree of reproducibility of results under varied conditions, such as different laboratories, analysts, or instruments [9].
Regulatory Context and Impact
Adherence to USP and EP standards is a legal requirement, not an optional scientific exercise [1]. As highlighted in USP General Chapter <1225>, test methods used to assess compliance with established specifications "must meet proper standards of accuracy and reliability" [9]. The validation data supporting these methods form the evidence base that inspectors scrutinize. A failure to adequately define and document parameters like LOD and LOQ can result in regulatory actions, including Form 483 observations or warning letters, ultimately jeopardizing product approval [1].
Comparative Analysis of LOD and LOQ Determination Methods
The ICH Q2(R1) guideline and pharmacopeial standards describe several accepted approaches for determining LOD and LOQ, each with distinct applications, advantages, and limitations [9] [86].
Table 1: Comparison of LOD and LOQ Determination Methods
| Method | Basis of Calculation | Typical Applications | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Standard Deviation of Blank and Slope [85] [86] | LOD = 3.3σ/S; LOQ = 10σ/SWhere σ = standard deviation of response, S = slope of the calibration curve | Instrumental techniques with low background noise (e.g., HPLC, electrochemical methods) | Scientifically rigorous; uses statistical properties of the calibration curve; does not require arbitrary S/N thresholds [86]. | Requires a linear calibration curve in the low concentration range; the estimate of σ (as standard error or y-intercept SD) is critical [86]. |
| Signal-to-Noise (S/N) [85] | LOD: S/N of 2:1 or 3:1LOQ: S/N of 10:1 | Chromatographic procedures that exhibit background noise [9]. | Simple, intuitive, and widely understood; particularly useful for chromatographic methods with baseline noise [85]. | Can be subjective; depends on how noise is measured; may not be suitable for techniques without a stable baseline [85] [86]. |
| Visual Evaluation [85] [9] | Determination by analysis of samples with known concentrations; establishing the minimum level reliably detected by an analyst or instrument. | Non-instrumental methods (e.g., turbidity) or impurity tests [9]. | Practical for non-instrumental limit tests; does not require complex calculations. | Subjective and dependent on analyst perception; provides less statistical defensibility [85]. |
The selection of the appropriate method must be justified based on the nature of the analytical technique. For instance, the calibration curve method is often preferred for its statistical foundation, whereas the signal-to-noise approach is a practical mainstay in chromatography [86]. Ultimately, whichever technique is used, the ICH requires that the calculated LOD and LOQ be confirmed by analyzing a suitable number of samples prepared at or near those limits to demonstrate that the proposed levels are practically achievable [86].
Experimental Protocols and Calculations
Protocol for LOD/LOQ via Calibration Curve
This protocol is widely applicable for techniques like HPLC and electrochemical methods, aligning with ICH Q2(R1) recommendations [86] [83].
- Preparation of Standard Solutions: Prepare a series of at least five standard solutions at concentrations spanning the expected low end of the working range, including the anticipated LOQ.
- Analysis and Calibration: Analyze each standard in duplicate or triplicate. Plot the analyte response (e.g., peak area, current) against the nominal concentration and perform a linear regression analysis.
- Data Calculation:
- From the linear regression output, record the slope (S) of the calibration curve and the standard error (SE) of the regression (or the standard deviation of the y-intercept) [86]. This value serves as the estimate for the standard deviation of the response (σ).
- Apply the ICH formulas:
- Experimental Verification: Prepare and analyze a minimum of six independent samples at the calculated LOQ concentration. The method is considered validated for LOQ if the precision (relative standard deviation) and accuracy (percentage recovery) meet pre-defined criteria (e.g., ±20% for precision and 80-120% for accuracy at the LOQ level) [86].
Table 2: Exemplary LOD and LOQ Calculation from HPLC Data [86]
| Parameter | Value | Source / Calculation |
|---|---|---|
| Calibration Curve Slope (S) | 1.9303 | Linear regression output |
| Standard Error (σ) | 0.4328 | Linear regression output |
| Calculated LOD | 0.74 ng/mL | 3.3 × 0.4328 / 1.9303 |
| Calculated LOQ | 2.24 ng/mL | 10 × 0.4328 / 1.9303 |
| Rounded LOQ for Verification | 3.0 ng/mL | Practical, conservative rounding |
Establishing the Range and Linearity
The validated range is established by demonstrating that the analytical procedure delivers acceptable levels of linearity, accuracy, and precision across the entire interval [9].
- Experiment Design: Prepare and analyze standard solutions or spiked samples at a minimum of five concentration levels covering the entire proposed range (e.g., 50-150% of the test concentration) [9].
- Linearity Assessment: Plot the analytical response against the concentration. Calculate the correlation coefficient (R), y-intercept, slope, and residual sum of squares. While a high R² (>0.998) is often achieved, visual inspection of the residual plot is crucial to confirm the assumption of linearity [83].
- Precision and Accuracy within Range: Accuracy (measured as percentage recovery) and precision (relative standard deviation) must be established across the range. For assay methods, accuracy should typically be 98-102%, with an RSD of less than 2% [9]. Data from an antiretroviral HPLC method validation exemplifies this, where linearity with R² >0.998 was demonstrated alongside precision and accuracy confirmations across the range [83].
Assessing Method Robustness
Robustness testing evaluates the method's resilience to deliberate, small variations in operational parameters.
- Identify Critical Parameters: Define the key method variables that could plausibly vary, such as mobile phase pH (±0.2 units), column temperature (±2°C), flow rate (±5%), or detection wavelength in HPLC [83]. For electrochemical methods, this could include electrode preconditioning time, buffer concentration, or applied potential.
- Design of Experiments (DoE): Use a structured approach, like a Plackett-Burman or factorial design, to efficiently study multiple factors simultaneously.
- Execute and Analyze: Perform analyses while varying the identified parameters within their predefined ranges. Monitor the impact on critical performance attributes, such as retention time, resolution, tailing factor, and assay result.
- Establish System Suitability: Based on the robustness data, set definitive system suitability criteria to ensure the method performs as validated during routine use. USP
<1225>and other general chapters provide guidance on these tests [82] [9].
The following workflow summarizes the strategic process for validating these key parameters, from preparation to final specification inclusion.
Essential Reagents and Research Solutions
Successful method validation relies on high-quality materials and reagents that conform to pharmacopeial standards.
Table 3: Research Reagent Solutions for Validation Studies
| Reagent / Material | Function in Validation | Pharmacopeial Consideration |
|---|---|---|
| Pharmacopeial Reference Standards [82] | To establish identity, potency, and purity; essential for calibrating instruments and confirming method accuracy. | USP and EP Reference Standards are highly purified and characterized; their use is critical for generating defensible data. |
| HPLC-Grade Solvents & Reagents [83] | To prepare mobile phases and sample solutions, minimizing background interference and ensuring reproducibility. | Reagents should meet or exceed the specifications described in general chapters (e.g., USP <1>). |
| Characterized Drug Substance [9] [83] | To prepare samples for accuracy (recovery) studies, precision testing, and forced degradation studies. | The certified purity of the drug substance is used to calculate theoretical concentrations and validate accuracy. |
| Specified Impurities/Degradation Products [9] | To validate specificity, LOD, and LOQ for impurity tests, demonstrating the ability to detect and quantify known impurities. | When available, official impurity standards should be used. Otherwise, well-characterized in-house standards are required. |
The rigorous quantification of LOD, LOQ, range, and robustness is a non-negotiable pillar of analytical method validation, directly underpinning data integrity and regulatory compliance. As detailed in this guide, multiple established pathways exist for determining these parameters, with the choice of method depending on the analytical technique and its intended application. The experimental protocols and calculations outlined here, rooted in ICH, USP, and EP standards, provide a actionable framework for researchers. By systematically implementing these practices and utilizing high-quality research reagents, scientists can ensure their methods—whether electrochemical, chromatographic, or other—are not only scientifically sound but also stand up to rigorous regulatory scrutiny, thereby safeguarding public health and ensuring product quality.
The United States Pharmacopeia (USP) and the European Pharmacopoeia (EP) establish legally enforceable quality standards for pharmaceutical products, serving as essential compendia for drug development, manufacturing, and quality control worldwide [1]. The USP is an independent non-profit scientific organization founded in 1820, publishing standards in the United States Pharmacopeia–National Formulary (USP–NF) [87]. These standards define requirements for pharmaceutical identity, strength, quality, purity, and performance [88]. The European Pharmacopoeia is published by the European Directorate for the Quality of Medicines & HealthCare (EDQM) and constitutes the official quality standard for medicines across Europe, legally binding in 39 European countries and applied in over 130 countries worldwide [89]. Both pharmacopeias provide comprehensive frameworks including monographs, general chapters, and reference standards that form the scientific basis for quality control throughout a product's lifecycle [87] [89].
Table 1: Fundamental Characteristics of USP and EP
| Characteristic | United States Pharmacopeia (USP) | European Pharmacopoeia (EP) |
|---|---|---|
| Governing Body | United States Pharmacopeia (Independent non-profit) | European Directorate for the Quality of Medicines & HealthCare (EDQM) |
| Legal Status | Enforceable by FDA under U.S. federal law [1] | Legally binding in 39 member states; mandatory for product approval in EU [1] [89] |
| Primary Documents | USP-NF (United States Pharmacopeia–National Formulary) [87] | Ph. Eur. (European Pharmacopoeia) [89] |
| Content Structure | Monographs, General Chapters, Reference Standards [88] | Monographs, General Texts (including general monographs and methods of analysis), Reagents [89] |
| Update Frequency | Regularly updated to reflect new science and technology [1] | Continually updated and modernized; 11th Edition effective January 2023 [89] |
Comparative Analysis of Structural Approaches
Similarities in Validation Philosophies
USP and EP share fundamental similarities in their overall approach to analytical method validation. Both pharmacopeias emphasize scientifically rigorous standards to ensure medicine safety and efficacy throughout the development lifecycle [87]. They function as essential quality benchmarks during drug discovery, preclinical research, clinical trials, regulatory approval, and post-marketing surveillance [87]. Compliance with either pharmacopeia provides regulatory predictability, improves research and development efficiency, facilitates international collaboration, and enhances data integrity [87]. Both systems require method validation that encompasses parameters such as specificity, accuracy, precision, detection limit, quantitation limit, linearity, and range [87]. Furthermore, both stress the importance of system suitability testing to verify that analytical systems operate correctly before sample analysis [87].
Differences in Regulatory Implementation
Despite shared scientific principles, USP and EP differ in specific regulatory implementation and methodological requirements. These differences manifest in terminology, validation protocols, and specific analytical procedures.
Table 2: Key Differences in Validation Approaches Between USP and EP
| Validation Aspect | USP Approach | EP Approach | Practical Implications |
|---|---|---|---|
| Terminology | Uses term "verification" for microbial ID methods [90] | Uses term "validation" for the same processes [90] | Conceptual difference in approach to method qualification |
| Microbial ID Method Comparison | Accepts parallel testing of approximately 50 isolates with existing system as verification approach [90] | Cautions against comparing one system with another due to different databases and methodologies [90] | Direct comparison studies acceptable for USP but problematic for EP compliance |
| Electroanalytical Methods | General chapters provide framework for electrochemical instrumentation qualification [87] | Includes specific electroanalytical test methods in monographs with measures to reduce systematic and random error [7] | EP may offer more specific guidance for electrochemical technique implementation |
| Microbial ID Validation Steps | Three verification options: 1) Parallel testing (~50 isolates), 2) 12-15 type strains, 3) Reference laboratory comparison [90] | Three-step validation: 1) Equipment qualification, 2) Primary method validation (by vendor), 3) Validation for intended use (by user) assessing accuracy, precision, robustness [90] | Structural difference in validation workflow with EP requiring more formalized stages |
A notable example of methodological differences appears in microbial identification, where the USP explicitly accepts parallel testing with existing systems as a verification approach, while the EP specifically cautions against this method due to variations in database coverage and technological principles between different systems [90]. This distinction necessitates careful planning for laboratories seeking compliance with both standards, particularly for global pharmaceutical companies facing inspections from different national regulators.
Experimental Protocols for Method Validation
General Method Validation Framework
The validation of analytical methods according to USP and EP guidelines follows a systematic approach to establish that the method is suitable for its intended purpose. Both pharmacopeias require laboratories to develop detailed Standard Operating Procedures (SOPs) for method selection, sample handling, method validation, data recording, and archiving [87]. The core validation parameters include specificity (ability to measure analyte accurately in presence of potential interferents), precision (degree of scatter in results under normal conditions), accuracy (closeness to true value), linearity (ability to obtain proportional results to analyte concentration), range (interval between upper and lower concentration with suitable precision, accuracy, and linearity), detection limit (lowest detectable amount), and quantitation limit (lowest quantifiable amount) [87].
For instrument qualification, both pharmacopeias require laboratories to conduct proper qualification and calibration of analytical instruments. USP <1058> specifically outlines requirements for Analytical Instrument Qualification, while USP <791> mandates calibration of pH measurement systems [87]. The use of official reference standards is essential in both frameworks to maintain analytical reliability, with requirements for proper storage, handling, and documentation [87]. System suitability testing forms a critical component, with USP <621> providing specific requirements for chromatographic system suitability tests [87].
Comparison of Methods Experiment Protocol
The comparison of methods experiment represents a critical validation component for assessing systematic error when implementing new methods. The following protocol outlines the standardized approach:
Method Comparison Protocol Flow
Define Comparative Method: Select an appropriate reference or comparative method. Reference methods with documented correctness through definitive method comparison or traceable reference materials are preferred [91]. When using routine methods as comparators, additional experiments may be needed to identify which method is inaccurate if differences are medically unacceptable [91].
Select Patient Specimens: A minimum of 40 different patient specimens should be tested, selected to cover the entire working range of the method and represent the spectrum of diseases expected in routine application [91]. Specimen quality and range distribution are more critical than total number, though 100-200 specimens help assess method specificity with different sample matrices [91].
Establish Testing Protocol: Analyze specimens within two hours between test and comparative methods to minimize stability issues [91]. Include several analytical runs on different days (minimum 5 days recommended) to minimize run-specific systematic errors [91]. Duplicate measurements are advantageous to identify sample mix-ups, transposition errors, and confirm discrepant results [91].
Execute Analysis & Initial Data Review: Analyze specimens according to established protocol. Perform graphical data inspection during collection using difference plots (test minus comparative results versus comparative result) or comparison plots (test result versus comparative result) to identify discrepant results needing confirmation [91].
Statistical Analysis & Error Estimation: For data covering a wide analytical range, calculate linear regression statistics (slope, y-intercept, standard deviation of points about the line) to estimate systematic error at medically important decision concentrations [91]. For narrow concentration ranges, calculate average difference (bias) between methods using paired t-test calculations [91].
Acceptability Assessment: Determine if estimated systematic errors at critical medical decision concentrations are within clinically acceptable limits based on method intended use and regulatory requirements [91].
Electrochemical Method Validation Case Study
Electrochemical methods represent an emerging area where pharmacopeial validation principles are applied. The development and validation of an electrochemical sensor for hydrochlorothiazide determination in urine demonstrates the application of these principles [92]. The validation included optimization of sensor components (glassy carbon electrode modified with multiwall carbon nanotubes and gold nanoparticles), calibration using cumulative standard addition method to address matrix effects, and comprehensive measurement uncertainty evaluation using the Monte Carlo Method (MCM) [92]. This approach allowed flexible combination of uncertainty components without restrictions regarding component uncertainty value or measurement function linearity [92].
Another validation study for manganese determination in drinking water using a cathodic stripping voltammetry electrochemical sensor demonstrated performance characteristics including 100% agreement with reference ICP-MS method, approximately 70% accuracy, and 91% precision [93]. The calculated limit of detection of 0.56 ppb represented nearly 2× improvement over previous sensor configurations [93].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Pharmacopeial Validation Studies
| Item Category | Specific Examples | Function in Validation |
|---|---|---|
| Reference Standards | USP Reference Standards, EP Reference Standards [87] | Verify identity, purity, strength; essential for quality control testing and method calibration |
| Electrochemical Materials | Multiwall carbon nanotubes (MWCNT), Gold nanoparticles, Glassy carbon electrode, Palladium/Pt electrodes [92] [93] | Sensor modification to enhance sensitivity, selectivity, and detection limits for electrochemical methods |
| Buffer Systems | Sodium acetate buffer (0.1 M, pH 5.2), Other pharmacopeial buffers [93] | Maintain consistent pH and ionic strength for reliable electrochemical measurements and method robustness |
| Calibration Standards | Atomic absorption standard solutions, Certified reference materials [93] | Establish calibration curves, determine method linearity, accuracy, and range |
| Mobile Phase Components | HPLC-grade solvents, Mass spectrometry-compatible additives | Achieve chromatographic separation as specified in monographs; ensure system suitability parameters |
| Microbiological Media | API test strips, Selective agars, Biochemical substrates [90] | Support microbial growth and characterization for identification systems validation |
Successful implementation of USP and EP validation approaches requires not only understanding regulatory requirements but also proper selection and qualification of research materials. Official pharmacopeial reference standards must be obtained from authorized agencies and stored according to regulatory guidelines to maintain reliability and stability [87]. For electrochemical methods specifically, nanomaterials such as multiwall carbon nanotubes and metal nanoparticles have demonstrated significant improvements in sensor performance characteristics including detection limits and sensitivity [92] [93].
The United States Pharmacopeia and European Pharmacopoeia provide complementary yet distinct frameworks for analytical method validation in pharmaceutical development and quality control. While sharing fundamental scientific principles focused on ensuring drug safety, efficacy, and quality, important differences exist in terminology, specific validation protocols, and acceptance criteria. For electrochemical methods specifically, both pharmacopeias provide general validation principles, with EP offering more specific electroanalytical methodologies in some areas. Successful global drug development requires understanding both systems' similarities and differences, implementing rigorous comparison protocols, and maintaining current knowledge of regularly updated standards. Harmonization efforts continue to reduce unnecessary differences while maintaining appropriate region-specific requirements, ultimately strengthening the global pharmaceutical quality framework.
The pharmaceutical industry is undergoing a significant paradigm shift, moving from traditional, document-centric validation processes to dynamic, data-driven approaches known as Validation 4.0 [94]. This evolution is an integral part of the broader Pharma 4.0 framework, leveraging digitalization to enhance competitiveness and deliver faster innovations for patient benefit [95] [94]. At the heart of Validation 4.0 lies Continuous Process Verification (CPV 4.0), which represents the final stage of Process Validation, ensuring ongoing quality assurance during routine production through real-time data monitoring [96].
For researchers and scientists, particularly those focused on the validation of electrochemical methods per USP/EP pharmacopeia research, understanding and implementing CPV 4.0 is crucial. This approach marks a fundamental departure from static, batch-centric verification to a holistic system where processes are continuously monitored and controlled using technologies such as the cloud, Internet of Things (IoT), edge computing, and Artificial Intelligence (AI) [96]. This article provides a comprehensive comparison of traditional versus 4.0 validation approaches, supported by experimental data and detailed methodologies relevant to modern pharmaceutical research and development.
Core Concepts: Traditional Validation vs. Validation 4.0
The Traditional Validation Approach
Historically, validation in the life sciences has been a static, document-centric process, relying on standalone paperwork to ensure proper operation [94]. This method is often time-consuming, rigid, inefficient, and difficult to maintain, impeding the adoption of new technologies and processes [94]. Process validation followed a linear path with distinct stages, but with limited integration and continuous monitoring capabilities.
The Validation 4.0 Framework
Validation 4.0 represents a dynamic, data-driven approach that incorporates several key elements: risk management, Quality by Design (QbD), data integrity by design, integrated environments, and integrated tools [94]. According to ISPE, process validation under Validation 4.0 is divided into three main stages:
- Stage 1: Holistic Planning and Design: Involves developing and understanding the manufacturing process, defining process and data flow maps, process parameters, and establishing the control strategy [94].
- Stage 2: Verification of Controls: Gathers evidence that the controls defined in Stage 1 have been implemented and are effectively reducing risks [94].
- Stage 3: Continuous Verification of Implemented Controls: Provides ongoing assurance that the process remains in a state of control during routine production [94].
The key difference from traditional process validation is the increased focus on a holistic, data-centric approach that incorporates risk assessment and control strategies from the design stage, rather than just qualifying the process in later stages [94].
Experimental Comparison of Control Strategies in Upstream Bioprocessing
Experimental Design and Methodology
An experimental approach developed in 2024 demonstrated the application of CPV 4.0 technologies in upstream bioprocessing using Pichia pastoris producing Candida rugosa lipase 1 under the regulation of the constitutive GAP promoter [96]. The bioprocess operated under hypoxic conditions in carbon-limited fed-batch cultures through physiological control based on the respiratory quotient (RQ) [96].
Core Experimental Components:
- Digital Twin Implementation: A virtual replica of the physical system was built, utilizing real-time data processed in cloud systems to emulate and mirror the behaviour of the actual bioprocess [96].
- Online Sensor Integration: Served as a bridge between the microorganism and AI models to provide predictions from both the edge and the cloud [96].
- AI Model Development: AI models emulated the metabolism of Pichia based on Critical Process Parameters (CPPs) and actionable factors to achieve expected Critical Quality Attributes (CQAs) [96].
The study compared three distinct control strategies:
- Manual-Heuristic Control (MHC)
- Boolean-Logic Control (BLC)
- AI-aided Adaptive-Proportional Control (AI-APC)
Comparative Performance Data
The table below summarizes the quantitative performance data for the three control strategies based on the experimental results:
Table 1: Performance Comparison of Control Strategies for RQ Maintenance
| Control Strategy | Accuracy (Mean Relative Error - MRE) | Precision (Root Mean Square Deviation - RMSD) | Reproducibility | Implementation Complexity |
|---|---|---|---|---|
| Manual-Heuristic Control (MHC) | 10% | Highest value | Lowest | Low |
| Boolean-Logic Control (BLC) | 5% | Medium value | Medium | Medium |
| AI-aided Adaptive-Proportional Control (AI-APC) | <4% | Lowest value | Highest | High |
Methodology for Method Comparison Studies
For researchers conducting comparison studies as part of method validation, proper experimental design is crucial. The following methodology is recommended based on established guidelines:
Table 2: Key Elements of Method Comparison Study Design
| Element | Recommendation | Rationale |
|---|---|---|
| Sample Size | Minimum of 40 patient specimens; preferably 100 or more [91] [97] | Larger sample size helps identify unexpected errors due to interferences or sample matrix effects [97] |
| Sample Selection | Cover entire clinically meaningful measurement range; represent spectrum of diseases [91] | Ensures evaluation across all relevant conditions |
| Measurement Replication | Duplicate measurements for both methods; analyze in different runs or different order [91] | Minimizes random variation effects; checks measurement validity |
| Time Period | Minimum of 5 days; extend to 20 days if possible [91] | Minimizes systematic errors that might occur in a single run |
| Specimen Stability | Analyze within 2 hours of each other by both methods [91] | Prevents differences due to specimen handling variables |
| Statistical Analysis | Use difference plots (Bland-Altman) and regression analysis; avoid correlation analysis and t-test alone [97] | Provides proper estimates of systematic error; correlation and t-test are inadequate for method comparison [97] |
Data Analysis and Statistical Considerations
For the statistical comparison of methods, researchers should avoid common pitfalls:
- Inadequate Statistical Methods: Correlation analysis only measures the degree of association between methods but cannot detect constant or proportional bias [97]. Similarly, t-tests may fail to detect clinically meaningful differences or may detect statistically significant but clinically irrelevant differences [97].
- Appropriate Graphical Methods: Difference plots (Bland-Altman plots) should be used to visualize the agreement between methods [97]. These plots display the differences between methods against the average of the methods, helping identify bias across the measurement range.
- Proper Regression Techniques: For data covering a wide analytical range, linear regression statistics are preferable as they allow estimation of systematic error at multiple decision concentrations and provide information about the proportional or constant nature of the error [91].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for CPV 4.0 Implementation
| Reagent/Material | Function/Application | Experimental Consideration |
|---|---|---|
| Pichia pastoris Cell Factory | Microbial host for recombinant protein production [96] | Selected due to advantages over other yeast cell factories for biopharmaceutical applications [96] |
| Constitutive GAP Promoter | Regulates expression of recombinant proteins [96] | Provides constitutive expression without need for induction |
| Glucose Carbon Source | Substrate for fed-batch cultivation [96] | Enables implementation of hypoxic conditions through RQ control |
| Respiratory Quotient (RQ) Control Reagents | Maintains hypoxic conditions for enhanced production [96] | Optimal RQ set-point of 1.4 determined for balancing production and efficiency [96] |
| Cloud-Based Data Infrastructure | Hosts large volumes of heterogeneous process data [95] | Enables breaking down data silos; provides computational power for AI/ML training [95] |
| AI/ML Algorithms | Pattern recognition, outlier detection, and predictive control [96] | Random forest models used for predicting control actions to maintain optimal RQ set-point [96] |
Technological Architecture for CPV 4.0 Implementation
Data Workflow Components
The most challenging steps in developing a CPV 4.0 technical solution are related to the data workflow, which includes four critical components [95]:
- Data Acquisition: Real-time data acquisition is key to breaking down data silos and avoiding manual data handling, which reduces time and effort, data integrity risk, and non-quality costs [95].
- Data Contextualization: Organizing data in a meaningful manner has advantages for the scalability of a solution, particularly through asset context (physical representation of factory assets) and batch event context (process events categorization) [95].
- Data Modeling: Statistical or ML modeling allows predictions of and alerts for out-of-specification and out-of-trend data, enabling proactive action to bring processes closer to the "golden batch" state [95].
- Data Visualization: Real-time monitoring solutions enable proactive actions with proper security, access controls, and monitoring for risk assessment [95].
Cloud Infrastructure Requirements
A cloud-hosted data store is ideal for the large volume of heterogeneous data required for a CPV 4.0 approach [95]. Key considerations include:
- Scalability: CPV 4.0 needs significant computation power for ML model training and predictive analysis, which cloud environments can provide elastically [95].
- Fault Tolerance: Inherent in cloud environments where operational resources are decoupled from physical servers, switches, and storage [95].
- Compliance: Cloud service providers are increasingly adding ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate + Complete, Consistent, Enduring, Available) considerations to their offerings [95].
- Total Cost of Ownership: The cloud can reduce TCO to approximately one-third that of traditional IT infrastructure through reduced maintenance and elimination of capital expenditure costs [95].
Implementation Workflow for CPV 4.0
The following diagram illustrates the integrated workflow for implementing Continuous Process Verification 4.0 in a pharmaceutical research setting:
Challenges and Implementation Recommendations
Key Implementation Challenges
The transition to Validation 4.0 presents several challenges for organizations:
- Cultural Mindset: The main roadblock for CPV 4.0 rollout is often the cultural mindset, emphasizing the need for willingness to change and adoption of social collaboration [95].
- AI Model Lifecycle: There is currently no established AI model lifecycle strategy for manufacturing, where an AI model is built, qualified, and validated into a specific process [95].
- Data Integrity: Real-time validation and continuous process verification are complex and require significant resources to ensure data integrity [94].
- Regulatory Acceptance: While regulatory bodies advocate for multivariable statistical approaches at Stage 3 of CPV [96], organizations are still looking for official standard procedures to qualify AI algorithms for regulated environments [95].
Strategic Recommendations
For successful implementation of Validation 4.0:
- Begin with Pilot Projects: Implement phased investment plans and start with pilot projects to demonstrate benefits before scaling [94].
- Invest in Robust Data Systems: Deploy robust data management systems and automated monitoring tools to ensure data integrity [94].
- Adopt Flexible Validation Methods: Implement flexible, scalable validation methods that can be easily adjusted as needed [94].
- Break Down Organizational Silos: Use integrated digital platforms and form cross-functional teams to enhance communication [94].
- Focus on Change Management: Creating centers of excellence and applying governance models helps unblock situations within large organizations [95].
The experimental comparison clearly demonstrates the significant advantages of AI-aided control strategies (AI-APC) over traditional approaches, with improved accuracy (MRE <4%), enhanced precision, and superior reproducibility in maintaining optimal process conditions [96]. For researchers and scientists working on validation of electrochemical methods per USP/EP pharmacopeia, embracing Validation 4.0 and CPV represents a fundamental shift from document-centric to data-driven approaches.
The integration of technologies such as cloud computing, IoT, edge computing, and AI enables a holistic, continuous verification approach that enhances product quality, reduces process variability, and accelerates delivery of products to patients [96] [94]. While implementation challenges exist, particularly around cultural mindset and regulatory acceptance of AI algorithms, the proven benefits in experimental settings indicate that Validation 4.0 is set to transform pharmaceutical manufacturing and quality assurance practices.
The future of method validation lies in dynamic, data-driven approaches that leverage digital technologies to ensure robust, reproducible processes while maintaining the highest standards of product quality and patient safety.
This guide provides a structured framework for researchers and drug development professionals to prepare for audits by ensuring analytical methods, particularly electrochemical techniques, are developed and validated in compliance with United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards.
The Evolving Regulatory Landscape for Analytical Validation
The regulatory framework governing analytical procedures is undergoing a significant shift, moving from a static validation model to a dynamic, lifecycle-based approach. This evolution is critical for audit readiness, as it forms the foundation of modern regulatory expectations.
USP General Chapter <1225>, a cornerstone of analytical validation, is currently under revision and slated for a new title: "Validation of Analytical Procedures" [11] [10] [98]. This change underscores its applicability to both compendial and non-compendial methods, ensuring a unified standard across pharmaceutical analysis [10]. The proposed revision, open for comment until January 31, 2026, aims to achieve closer alignment with ICH Q2(R2) on analytical procedure validation and integrate seamlessly with the Analytical Procedure Lifecycle (APL) concepts described in USP <1220> and the newly introduced USP <1221> on Ongoing Procedure Performance Verification [11] [10] [98].
Concurrently, the Pharmacopeial Discussion Group (PDG) works to harmonize general methods across the USP, EP, and Japanese Pharmacopoeia (JP). The table below summarizes the harmonization status of key methodologies, indicating a continuous effort towards global standardization that auditors will expect compliance with [18].
Table: Harmonization Status of Key Pharmacopeial General Methods
| PDG# | Method Name | Coordinating Pharmacopeia | PDG Harmonization Sign-off Status | Stage 4 Web Posting Date |
|---|---|---|---|---|
| Q-02 | Disintegration <701> | USP | S4 (Former S6), Rev. 2 (24-Jun-2025) | 26-Sep-2025 [18] |
| Q-03/04 | Uniformity of Content/Mass <905> | USP | S4 (Former S6), Rev. 2 (04-Nov-2015) | 30-Sep-2022 [18] |
| Q-05b | Microbial Enumeration: <61> | EP | S4 (Former S6), Rev. 1, Corr. 2 (22-Aug-2023) | 26-Apr-2024 [18] |
| Q-09 | Particulate Contamination <788> | EP | S4 (Former S6), Rev. 2 (02-May-2025) | 25-Jul-2025 [18] |
Core Concepts for Modern Method Validation and Documentation
The revised guidelines introduce several pivotal concepts that must be thoroughly understood and documented to demonstrate regulatory readiness during an audit.
- Reportable Result (RR): The revision elevates the "Reportable Result" as the definitive output used for batch release and compliance decisions [11] [10] [98]. Validation must demonstrate that the entire analytical procedure, including all replication steps, produces a reliable RR, not just that individual instrument measurements are precise.
- Fitness for Purpose: This is positioned as the overarching goal of validation [11] [10]. It requires that the analytical procedure's performance characteristics are aligned with its intended use, moving beyond generic, checkbox-style validation [11].
- Replication Strategy: The revised <1225> links replication strategy directly to controlling the uncertainty of the Reportable Result [10] [98]. The strategy should reflect the actual routine testing procedure to ensure validation studies capture real-world variability [11].
- Statistical Intervals and Combined Evaluation: The revision introduces the use of statistical intervals (confidence, prediction, tolerance) for evaluation [10] [98]. It also provides expanded guidance on the combined evaluation of accuracy and precision, offering a more scientifically rigorous approach than assessing these parameters in isolation [11] [10].
Comparative Assessment of Electrochemical Methods: A Case Study
Electrochemical methods are valuable in pharmaceutical analysis. A case study on paracetamol provides a template for how to rigorously compare and validate such methodologies. The study compared different calculation methods for key electrochemical parameters essential for validating a method as "fit for purpose" [23].
Table: Comparison of Methodologies for Calculating Electrochemical Parameters of Paracetamol [23]
| Parameter | Methodology 1 | Methodology 2 | Optimal Methodology per Study | Calculated Value |
|---|---|---|---|---|
| Transfer Coefficient (α) | Ep − Ep/2 equation | Alternative equation | Ep − Ep/2 equation | Effective for calculation [23] |
| Diffusion Coefficient (D₀) | Modified Randles–Ševčík equation | Alternative equation | Modified Randles–Ševčík equation | Effective for calculation [23] |
| Heterogeneous Electron Transfer Rate Constant (k₀) | Nicholson and Shain’s method | Kochi and Gileadi methods | Kochi and Gileadi methods (Reliable alternatives) | ~10⁻² to 10⁻⁵ cm/s (Quasi-reversible) [23] |
Detailed Experimental Protocol for Electrochemical Method Comparison
The following protocol, adapted from the paracetamol case study, outlines the key steps for generating comparative validation data suitable for audit documentation [23].
- Instrumentation and Materials: The experiments were performed using a CHI 760D Electrochemical Workstation. The system utilized a three-electrode cell: a glassy carbon (GC) working electrode (surface area 0.0706 cm²), a platinum counter electrode, and a saturated calomel electrode (SCE) as the reference. The analyte was a 10 mL solution of 1 × 10⁻⁶ M paracetamol with 0.1 M LiClO₄ as the supporting electrolyte in deionized water [23].
- Sample Preparation and System Setup: The supporting electrolyte (LiClO₄) was dissolved in deionized water to create a 0.1 M solution. Paracetamol was then added to achieve a final concentration of 1 × 10⁻⁶ M. Before analysis, the solution was purged with nitrogen gas for 15 minutes to remove dissolved oxygen. The working electrode was polished with 0.2 µm aluminum powder before each experiment to ensure a clean, reproducible surface [23].
- Data Acquisition and Analysis: Cyclic voltammetry was carried out at scan rates from 0.025 V/s to 0.300 V/s with an incremental change of 0.025 V/s. From the resulting voltammograms, direct parameters—anodic peak potential (Epa), cathodic peak potential (Epc), anodic peak current (Ipa), and cathodic peak current (Ipc)—were recorded. Key metrics such as peak separation (ΔEp = |Epc - Epa|) and the peak current ratio (Ipc/Ipa) were calculated to assess the nature of the electron transfer process. The values of α, D₀, and k₀ were then determined using the comparative methodologies listed in the table above [23].
The Scientist's Toolkit: Essential Reagents and Materials
A well-documented and controlled inventory of materials is a fundamental aspect of audit readiness. The following table details key reagents and solutions used in the featured electrochemical experiment and their general functions in pharmacopeial research [99] [23].
Table: Key Research Reagent Solutions for Electrochemical and Pharmacopeial Analysis
| Item | Function in Experiment/Field |
|---|---|
| USP/EP Reference Standards | Highly purified and characterized substances used for method calibration, qualification of instruments, and validation to ensure analytical results are accurate and traceable to official standards [99]. |
| Supporting Electrolyte (e.g., LiClO₄) | A salt added in high concentration to the analyte solution to increase its electrical conductivity, minimize ohmic resistance ("iR drop"), and control the ionic strength [23]. |
| Analytical Grade Solvent (e.g., Deionized Water) | The medium in which the analysis is performed; must be of suitable purity to not introduce interference or contamination [23]. |
| System Suitability Test Materials | Reference materials or test mixtures used to confirm that the chromatographic or analytical system is performing adequately as required by chapters like USP <621> before or during the analysis [99]. |
Strategic Framework for Audit Readiness
Proactive preparation is the most effective strategy for a successful audit. Aligning documentation and practices with the modern lifecycle approach is crucial.
- Implement the Lifecycle Approach: Integrate the principles of USP <1220> Analytical Procedure Lifecycle into your quality system. Move beyond one-time validation and establish robust procedures for ongoing performance verification as outlined in the new USP <1221> [11] [10].
- Document the "Why" Behind Decisions: For each method, maintain clear documentation that articulates its "fitness for purpose" [11] [10]. The experimental design, including the replication strategy, should be justified based on the intended use of the Reportable Result and controlled through a defined Analytical Procedure Control Strategy [11] [10].
- Maintain Rigorous Knowledge Management: Audit-ready documentation includes not just the final validation report, but also data from method development, prior knowledge, and all studies that build the procedure's knowledge base [11] [10]. This demonstrates a deep understanding of the method and its controls.
- Leverage Harmonized Standards: Where available, implement PDG-harmonized methods to streamline validation and demonstrate adherence to global standards [18]. For non-compendial methods, such as the electrochemical case study, a rigorous, well-documented comparative assessment provides the evidence needed to justify the selected methodology [23].
Conclusion
The successful validation of electrochemical methods is paramount for demonstrating pharmaceutical product quality and ensuring regulatory compliance with USP and EP. This synthesis of core concepts demonstrates that a foundational understanding of pharmacopeia guidelines, combined with robust methodological application, proactive troubleshooting, and a meticulous validation strategy, forms the bedrock of reliable analytical procedures. The future of electrochemical method validation is increasingly digital and continuous, embracing Quality by Design principles and Process Analytical Technology for real-time quality assurance. As analytical technologies advance, their integration into a flexible, knowledge-driven framework will be crucial for developing safer and more effective medicines, solidifying the role of validated electrochemical methods as indispensable tools in modern pharmaceutical sciences.
References
- 1. Understanding the Role of USP and EP Standards in ... [https://www.labmanager.com/understanding-the-role-of-usp-and-ep-standards-in-pharma-qc-ensuring-consistency-and-compliance-34061]
- 2. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoprjQA4Y_5l7_uxh3KnjyKIsTyEaEaDgEbUPIpMtt9L0-D6Zonx]
- 3. USP Reference Standards [https://www.usp.org/reference-standards]
- 4. Validation, Verification, and Transfer of Analytical ... [https://www.usp.org/events-training/course/validation-verification-and-transfer-analytical-procedures-demand]
- 5. Development and validation of an advanced ... [https://pubmed.ncbi.nlm.nih.gov/32312432/]
- 6. Overview of USP and Ph. Eur. Allowable Adjustments for ... [https://www.chromatographyonline.com/view/overview-of-usp-and-ph-eur-allowable-adjustments-for-hplc-monograph-methods]
- 7. [Electroanalytical methods in the European pharmacopoeia] [https://pubmed.ncbi.nlm.nih.gov/11961904/]
- 8. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOorMGrvt_Zie8zCdGhncyPdB5JDbN1u4K1VtHfa36Iqcyd1rkDDa]
- 9. <1225> VALIDATION OF COMPENDIAL METHODS [http://www.uspbpep.com/usp29/v29240/usp29nf24s0_c1225.html]
- 10. Proposed Revision of USP <1225> Published in the ... [https://www.gmp-compliance.org/gmp-news/proposed-revision-of-usp-1225-published-in-the-pharmacopeial-forum]
- 11. USP <1225> Revised: Aligning Compendial Validation with ... [https://investigationsquality.com/2025/11/13/usp-1225-revised-aligning-compendial-validation-with-ich-q2r2-and-q14s-lifecycle-vision/]
- 12. Free Download of European Pharmacopoeia 10th Edition [https://www.m-pharmaguide.com/2024/09/european-pharmacopoeia-10th-edition.html]
- 13. European Pharmacopoeia 11th Edition pdf free download [https://www.webofpharma.com/2023/06/european-pharmacopoeia-11th-edition-pdf.html]
- 14. Quality by Design: Principles to Develop Successful Products [https://www.scilife.io/blog/quality-by-design-principles]
- 15. Quality by Design for Analytical Methods: Implications ... [https://www.pharmtech.com/view/quality-design-analytical-methods-implications-method-validation-and-transfer]
- 16. Comparison of colorimetric, spectroscopic and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10910492/]
- 17. Comparison of Chromatographic and Electrochemical ... [https://www.mdpi.com/2076-3417/15/10/5504]
- 18. Harmonization Status for General Methods [https://www.usp.org/harmonized-standards/pdg/general-methods]
- 19. Microbiology Roundtable [https://www.americanpharmaceuticalreview.com/Featured-Articles/620802-Microbiology/]
- 20. Introduction to Analytical Method Validation [https://lab-training.com/introduction-to-analytical-method-validation/]
- 21. Validation of Analytical Methods [https://www.intechopen.com/chapters/57909]
- 22. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoqcO5oGeWhYJUU7dIt7fpKvIaStYjeROJsF94Ed_iDVjm_LxLo1]
- 23. Comparison of Different Electrochemical Methodologies for ... [https://www.mdpi.com/2673-3293/5/1/4]
- 24. A comparative study of different electrochemical methods ... [https://www.sciencedirect.com/science/article/abs/pii/S0378775321002482]
- 25. A Succinct Assessment of the Development, Validation ... [https://www.ashdin.com/articles/a-succinct-assessment-of-the-development-validation-and-method-verification-of-analytical-methods-for-natural-and-synthetic-therap-104701.html]
- 26. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoogZ0sAjj1szInbCSl4gPCMi_PCLrkxkYYyOvsII6pobHKm7DrL]
- 27. A practical guide to validation and verification of analytical ... [https://www.sciencedirect.com/science/article/abs/pii/S006524231930006X]
- 28. Amit Andhale's Post - Analytical Method Validation [https://www.linkedin.com/posts/amitandhale_validation-amv2025-ich-activity-7395567873747808256-4ZKY]
- 29. Electrochemical and chromatographic methods for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814624041414]
- 30. 11 The need for CE methods in pharmacopoeial monographs [https://www.sciencedirect.com/science/article/abs/pii/S0149639507000116]
- 31. Electroanalysis Advances in Pharmaceutical Sciences [https://www.mdpi.com/2673-4532/6/2/12]
- 32. Electrochemical Analysis Methods: Potentiometry, ... [https://www.labmanager.com/electrochemical-analysis-methods-potentiometry-voltammetry-and-more-34242]
- 33. Electro-Oxidation Mechanism and Direct Square-Wave ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4379891/]
- 34. Flow injection amperometry as an alternative to ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468621003649]
- 35. Recognition of endpoints (EP) [https://www.metrohm.com/en/discover/blog/20-21/recognition-of-endpoints--ep-.html]
- 36. 11.4: Voltammetric and Amperometric Methods [https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Analytical_Chemistry_2.1_(Harvey)/11%3A_Electrochemical_Methods/11.04%3A_Voltammetric_and_Amperometric_Methods]
- 37. The development, validation, and optimization of a ... [https://www.sciencedirect.com/science/article/pii/S0026265X23007610]
- 38. Single-Drop Analysis of Epinephrine and Uric Acid on a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8392267/]
- 39. Highly Sensitive Square Wave Adsorptive Stripping ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12163804/]
- 40. Screening method for the simultaneous determination of ... [https://www.sciencedirect.com/science/article/abs/pii/S0731708522006434]
- 41. Neurotransmitter analysis using liquid chromatography ... [https://www.sciencedirect.com/science/article/abs/pii/S0731708525004613]
- 42. The High-Precision Liquid Chromatography with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10818480/]
- 43. Analytical Method Validation for Estimation ... - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC8799795/]
- 44. Are You Sure You Understand USP <621>? [https://www.chromatographyonline.com/view/are-you-sure-you-understand-usp-621-]
- 45. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOorpYkRcrSRFde2N3H4C_RDQJytCVpnMXtxerb1BELYt8BNouHLY]
- 46. Neurotransmitter Analysis: HPLC-ECD vs. LC-MS/MS [https://www.amuzainc.com/blog/hplc-ecd-vs-lc-ms-neurotransmitter-analysis/?srsltid=AfmBOoq_9HB_vaRR9G5SSBTMX_W7JFtg3TeBumUT8g30GWVXMJvTPtp8]
- 47. Efficient and economic HPLC performance qualification [https://www.sciencedirect.com/science/article/abs/pii/S0731708509005421]
- 48. Proposed Update to USP <1058> on Analytical Instrument ... [https://www.gmp-compliance.org/gmp-news/proposed-update-to-usp-1058-on-analytical-instrument-and-system-qualification]
- 49. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoobosnckQEQWsR6207hC65OhJ4AcXxP8fxg7vVGKSgV8Oci6xfr]
- 50. Modeling & Simulation as Decision-Making Tools for Drug ... [https://www.americanpharmaceuticalreview.com/Featured-Articles/585194-Modeling-Simulation-as-Decision-Making-Tools-for-Drug-Substance-Continuous-Manufacturing/]
- 51. In Vitro Studies are Sometimes Better than Conventional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2751377/]
- 52. Validation of Analytical Methods for Biomarkers Employed ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2744124/]
- 53. Product Performance Analysis - A 7-Step Playbook with ... [https://uxcam.com/blog/product-performance-analysis/]
- 54. Implementing Alternative Methods [https://www.fda.gov/science-research/advancing-alternative-methods-fda/implementing-alternative-methods]
- 55. Strategies for Assessing the Limit of Detection in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11300140/]
- 56. How to improve LOD or detection limits in HPLC [https://www.mtc-usa.com/kb-article/aa-01024]
- 57. When lower isn't always better in biosensor research and ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566324006766]
- 58. Detection limit matters. Sensitivity not really [https://www.biolinscientific.com/blog/qcm-sensitivity-vs-limit-of-detection-lod?update_2025=1]
- 59. A tutorial for computing limits of detection and ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267021011685]
- 60. Methods For Improving Sensitivity in Gas Chromatography ... [https://www.alwsci.com/news/methods-for-improving-sensitivity-in-gas-chrom-85272601.html]
- 61. The Limit of Detection, the Uncertainty of Measurement, ... [https://www.mdpi.com/2079-6412/15/6/719]
- 62. Comparison of approaches for assessing detection and ... [https://www.nature.com/articles/s41598-024-83474-5]
- 63. Harmonization Status for General Chapters [https://www.usp.org/harmonized-standards/pdg/general-chapters]
- 64. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoo2wDDxo1-FU581bdQD0kEa2z8gWPH7wcaA60QUaaCvbHRmF8gU]
- 65. Electrochemical sensing of benzodiazepines [https://www.sciencedirect.com/science/article/pii/S2667056925000987]
- 66. emphasizing green, white, and blue analytical approaches [https://www.nature.com/articles/s41598-024-69518-w]
- 67. Comparison of Chemical and Electrochemical Approaches ... [https://www.mdpi.com/1424-8220/23/5/2776]
- 68. Electrochemical Degradation of Venlafaxine on Platinum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12073582/]
- 69. Multivariate Chemometric Comparison of Forced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9920961/]
- 70. Optimization of a gold electrodeposited platform for the ... [https://www.sciencedirect.com/science/article/pii/S0013468625001501]
- 71. Selective and Mechanically Robust Sensors for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5750107/]
- 72. The Development and Full Validation of a Novel Liquid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10631362/]
- 73. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOorczpP1BOYPx8k_eazWNQ7S_HzWcOlilU71LbXfq8WIZTzno28Q]
- 74. Scientific Validation [https://www.sciencedocs.com/validation-the-key-to-experimental-repeatability-and-a-sound-scientific-publication/]
- 75. Case Study: Automating the processing of analytical data ... [https://mestrelab.com/resources/case-study-automating-the-processing-of-analytical-data-for-system-suitability-testing-sst.html]
- 76. Long-Term System Suitability Evaluation for Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12412142/]
- 77. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOoq2LQHzN5VMlAVdwVsbyy8l7iykKFLafaP2KhIAScFTP1xo985S]
- 78. How to Ensure Data Integrity: Best Practices and Tools [https://www.acceldata.io/blog/how-to-ensure-data-integrity-strategies-tools-and-best-practices]
- 79. Essentials of Data Management: An Overview - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8371066/]
- 80. Essential Data Integrity Best Practices for 2025 [https://atlan.com/data-integrity-best-practices/]
- 81. Data Integrity in a Data Pipeline: Best Practices and ... [https://medium.com/@georgemichaeldagogomaynard/data-integrity-in-a-data-pipeline-best-practices-and-strategies-for-data-quality-checks-dim-71af7a3bf21e]
- 82. Navigating the Regulatory Landscape: USP and EP Standards in Drug Development - Analytical Products / Alfa Chemistry [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html]
- 83. The Validation of a Simple, Robust, Stability-Indicating RP ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8837449/]
- 84. Limit of Blank, Limit of Detection and Limit of Quantitation [https://pmc.ncbi.nlm.nih.gov/articles/PMC2556583/]
- 85. Method Validation Essentials, Limit of Blank ... [https://www.biopharminternational.com/view/method-validation-essentials-limit-blank-limit-detection-and-limit-quantitation]
- 86. Determining LOD and LOQ Based on the Calibration Curve [https://www.sepscience.com/hplc-solutions-126-chromatographic-measurements-part-5-determining-lod-and-loq-based-on-the-calibration-curve-6959]
- 87. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOorG2LgicXM1awK42ckRa17zKBOIq_JvXneyVR5oTliqqfvogc7e]
- 88. What is a USP Monograph [https://www.usp.org/about/public-policy/overview-of-monographs]
- 89. European Pharmacopoeia (Ph. Eur.) 11th Edition [https://www.edqm.eu/en/european-pharmacopoeia-ph.-eur.-11th-edition]
- 90. Automated Microbial Identification: A Comparison of USP ... [https://www.americanpharmaceuticalreview.com/Featured-Articles/140520-Automated-Microbial-Identification-A-Comparison-of-USP-and-EP-Approaches/]
- 91. The Comparison of Methods Experiment [https://www.westgard.com/lessons/basic-method-validation/lesson23.html]
- 92. Development and validation of an advanced ... [https://www.sciencedirect.com/science/article/abs/pii/S0039914020301740]
- 93. Validation of electrochemical sensor for determination ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10704915/]
- 94. Transitioning to Process Validation 4.0 in Pharma [https://www.scilife.io/blog/how-to-transition-to-validation-4.0]
- 95. Using Technology for Continuous Process Verification 4.0 [https://ispe.org/pharmaceutical-engineering/january-february-2023/using-technology-continuous-process-verification]
- 96. Continuous Process Verification 4.0 application in upstream [https://pmc.ncbi.nlm.nih.gov/articles/PMC11480048/]
- 97. Statistical analysis in method comparison studies part one [https://acutecaretesting.org/en/articles/statistical-analysis-in-method-comparison-studies-part-one]
- 98. USP <1225> Revision: Changes and Impacts [https://www.linkedin.com/posts/adel-nabil-333b7120_proposed-revision-of-usp-1225-validation-activity-7395793986616999936-MEDl]
- 99. USP and EP Standards in Drug Development [https://reagents.alfa-chemistry.com/resources/navigating-the-regulatory-landscape-usp-and-ep-standards-in-drug-development.html?srsltid=AfmBOooLhAVMUNKOcI8Jul0-FNiG788OwWwnpi_dX1raYTxTh1aCKSON]